Abstract
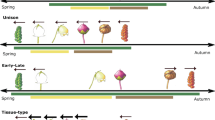
As climate warming accelerates, shifts in plant phenology are reshaping the functioning and stability of terrestrial ecosystems. While the roles of climatic drivers in shaping phenological responses to warming are well established, the influence of intrinsic plant functional traits remains poorly understood. Here, we combine two complementary approaches through a meta-analysis of 124 field warming experiments and an analysis of long-term phenological monitoring networks (CPON and USA‑NPN) to evaluate phenological responses to warming across a spectrum of resource-use strategies in seasonally cold biomes. Our meta-analysis demonstrates that resource-acquisitive plants, characterized by higher nutrient concentrations and thinner leaves, show significantly stronger phenological responses to experimental warming. This pattern is observed consistently across both leaf-out in spring and senescence in autumn. These results from meta-analysis are further supported by two long-term observational datasets, which also show more pronounced phenological shifts in acquisitive species under long-term warming. Our findings present a trait-climate integration framework that extends beyond conventional environmental drivers, providing a mechanistic foundation to enhance the accuracy of forecasts for plant responses to climate change.
Similar content being viewed by others
Data availability
The data generated in this study have been deposited in Figshare at https://doi.org/10.6084/m9.figshare.29917052. Plant traits data were obtained from TRY Plant Trait Database (https://www.try-db.org/), climate data from WorldClim (https://www.worldclim.org/) and Climate Research Unit (https://crudata.uea.ac.uk/cru/data/hrg/cru_ts_4.08/). The long-term ground phenological data of USA National Phenology Network (USA-NPN) are available from the website: https://www.usanpn.org/ results/data. The phenological data from China Phenological Observation Network (CPON) were provided by the Meteorological Information Center of the China Meteorological Administration. Source data are provided with this paper.
Code availability
Codes for analysis are available in the Figshare repository (https://doi.org/10.6084/m9.figshare.29917052).
References
Piao, S. et al. Net carbon dioxide losses of northern ecosystems in response to autumn warming. Nature 451, 49–52 (2008).
Manlick, P. J., Perryman, N. L., Koltz, A. M., Cook, J. A. & Newsome, S. D. Climate warming restructures food webs and carbon flow in high-latitude ecosystems. Nat. Clim. Chang. 14, 184–189 (2024).
Xi, Y., Zhang, W., Wei, F., Fang, Z. & Fensholt, R. Boreal tree species diversity increases with global warming but is reversed by extremes. Nat. Plants 10, 1473–1483 (2024).
Körner, C. & Basler, D. Phenology under global warming. Science 327, 1461–1462 (2010).
Oleksyn, J. et al. A fingerprint of climate change across pine forests of Sweden. Ecol. Lett. 23, 1739–1746 (2020).
Reich, P. B., Rich, R. L., Lu, X., Wang, Y.-P. & Oleksyn, J. Biogeographic variation in evergreen conifer needle longevity and impacts on boreal forest carbon cycle projections. Proc. Natl. Acad. Sci. USA 111, 13703–13708 (2014).
Elmore, A. J., Nelson, D. M. & Craine, J. M. Earlier springs are causing reduced nitrogen availability in North American eastern deciduous forests. Nat. Plants 2, 16133 (2016).
Chuine, I. Why does phenology drive species distribution?. Philos. Trans. R. Soc. B: Biol. Sci. 365, 3149–3160 (2010).
Fridley, J. D. Extended leaf phenology and the autumn niche in deciduous forest invasions. Nature 485, 359–362 (2012).
Yang, X. et al. Plant phenology response to nitrogen addition decreases community biomass stability in an alpine meadow. New Phytol. nph.70132, (2025).
Shen, M. et al. Plant phenology changes and drivers on the Qinghai–Tibetan Plateau. Nat. Rev. Earth Environ. 3, 633–651 (2022).
Fu, Y. H. et al. Declining global warming effects on the phenology of spring leaf unfolding. Nature 526, 104–107 (2015).
Möhl, P., Von Büren, R. S. & Hiltbrunner, E. Growth of alpine grassland will start and stop earlier under climate warming. Nat. Commun. 13, 7398 (2022).
Meng, L. et al. Photoperiod decelerates the advance of spring phenology of six deciduous tree species under climate warming. Glob. Chang. Biol. 27, 2914–2927 (2021).
Wang, H. et al. Divergent phenological responses of soil microorganisms and plants to climate warming. Nat. Geosci. 18, 753–760 (2025).
Peaucelle, M., Peñuelas, J. & Verbeeck, H. Accurate phenology analyses require bud traits and energy budgets. Nat. Plants 8, 915–922 (2022).
Montgomery, R. A., Rice, K. E., Stefanski, A., Rich, R. L. & Reich, P. B. Phenological responses of temperate and boreal trees to warming depend on ambient spring temperatures, leaf habit, and geographic range. Proc. Natl. Acad. Sci. USA 117, 10397–10405 (2020).
Shen, P. et al. Biodiversity buffers the response of spring leaf unfolding to climate warming. Nat. Clim. Chang. 14, 863–868 (2024).
Rauschkolb, R. et al. Spatial variability in herbaceous plant phenology is mostly explained by variability in temperature but also by photoperiod and functional traits. Int J Biometeorol 68, 761–775 (2024).
Joswig, J. S. et al. Climatic and soil factors explain the two-dimensional spectrum of global plant trait variation. Nat. Ecol. Evol. 6, 36–50 (2022).
Reich, P. B., Walters, M. B. & Ellsworth, D. S. From tropics to tundra: global convergence in plant functioning. Proc. Natl. Acad. Sci. USA 94, 13730–13734 (1997).
Wright, I. J. et al. The worldwide leaf economics spectrum. Nature 428, 821–827 (2004).
Quan, Q. et al. Plant height as an indicator for alpine carbon sequestration and ecosystem response to warming. Nat. Plants 10, 890–900 (2024).
Augusto, L. et al. Widespread slow growth of acquisitive tree species. Nature 640, 395–401 (2025).
Yan, P. et al. Plant acquisitive strategies promote resistance and temporal stability of semiarid grasslands. Ecol. Lett. 28, e70110 (2025).
Dorji, T. et al. Plant functional traits mediate reproductive phenology and success in response to experimental warming and snow addition in Tibet. Glob. Chang. Biol. 19, 459–472 (2013).
Bucher, S. F. & Römermann, C. The timing of leaf senescence relates to flowering phenology and functional traits in 17 herbaceous species along elevational gradients. J. Ecol. 109, 1537–1548 (2021).
Chuine, I. A unified model for budburst of trees. J. Theor. Biol. 207, 337–347 (2000).
Herms, D. A. & Mattson, W. J. The dilemma of plants: to grow or defend. Q. Rev. Biol. 67, 283–335 (1992).
de Bello, F. et al. Functional trait effects on ecosystem stability: assembling the jigsaw puzzle. Trends Ecol. Evol. 36, 822–836 (2021).
Ma, Q., Huang, J.-G., Hänninen, H. & Berninger, F. Divergent trends in the risk of spring frost damage to trees in Europe with recent warming. Glob. Chang. Biol. 25, 351–360 (2019).
Guo, Y. et al. Leaf senescence: progression, regulation, and application. Mol. Hortic. 1, 1–25 (2021).
Liu, Q. et al. Extension of the growing season increases vegetation exposure to frost. Nat. Commun. 9, 426 (2018).
Richardson, A. D. et al. Ecosystem warming extends vegetation activity but heightens vulnerability to cold temperatures. Nature 560, 368–371 (2018).
Alexander, J. M. & Levine, J. M. Earlier phenology of a nonnative plant increases impacts on native competitors. Proc. Natl. Acad. Sci. USA 116, 6199–6204 (2019).
Craven, D. et al. Multiple facets of biodiversity drive the diversity–stability relationship. Nat. Ecol. Evol. 2, 1579–1587 (2018).
Májeková, M., de Bello, F., Doležal, J. & Lepš, J. Plant functional traits as determinants of population stability. Ecology 95, 2369–2374 (2014).
Zhang, X. et al. Resource-acquisitive species have greater plasticity in leaf functional traits than resource-conservative species in response to nitrogen addition in subtropical China. Sci. Total Environ. 903, 166177 (2023).
Li, H.-L. et al. China’s subtropical deciduous plants are more sensitive to climate change than evergreen plants by flowering phenology. Glob. Chang. Biol. 30, e17168 (2024).
Polley, H. W., Isbell, F. I. & Wilsey, B. J. Plant functional traits improve diversity-based predictions of temporal stability of grassland productivity. Oikos 122, 1275–1282 (2013).
Zohner, C. M. et al. Effect of climate warming on the timing of autumn leaf senescence reverses after the summer solstice. Science 381, eadf5098 (2023).
Bigler, C. & Vitasse, Y. Premature leaf discoloration of European deciduous trees is caused by drought and heat in late spring and cold spells in early fall. Agric. For. Meteorol. 307, 108492 (2021).
Vitasse, Y. et al. Impact of microclimatic conditions and resource availability on spring and autumn phenology of temperate tree seedlings. New Phytol 232, 537–550 (2021).
Wu, X. et al. Canopy structure regulates autumn phenology by mediating the microclimate in temperate forests. Nat. Clim. Chang. 14, 1299–1305 (2024).
Bai, Y. et al. Heating up the roof of the world: tracing the impacts of in-situ warming on carbon cycle in alpine grasslands on the Tibetan Plateau. Natl. Sci. Rev. 12, nwae371 (2024).
Zani, D., Crowther, T. W., Mo, L., Renner, S. S. & Zohner, C. M. Increased growing-season productivity drives earlier autumn leaf senescence in temperate trees. Science 370, 1066–1071 (2020).
Wang, H. et al. Alpine grassland plants grow earlier and faster but biomass remains unchanged over 35 years of climate change. Ecol. Lett. 23, 701–710 (2020).
Umaña, M. N. et al. Upscaling the effect of traits in response to drought: the relative importance of safety–efficiency and acquisitive–conservation functional axes. Ecol. Lett. 26, 2098–2109 (2023).
Zhang, W. et al. Seasonal stabilization effects slowed the greening of the Northern Hemisphere over the last two decades. Nat Commun 16, 6287 (2025).
Richardson, A. D. et al. Influence of spring and autumn phenological transitions on forest ecosystem productivity. Philos. Trans. R. Soc. B: Biol. Sci. 365, 3227–3246 (2010).
Gallinat, A. S., Primack, R. B. & Wagner, D. L. Autumn, the neglected season in climate change research. Trends Ecol. Evol. 30, 169–176 (2015).
Lubbe, F. C., Klimešová, J. & Henry, H. A. L. Winter belowground: changing winters and the perennating organs of herbaceous plants. Funct. Ecol. 35, 1627–1639 (2021).
Xu, H. et al. Convergent strategies for leaf traits in tree species from divergent habitats. Glob. Chang. Biol. 31, e70108 (2025).
Harris, T. et al. Capital and income breeders among herbs: how relative biomass allocation into a storage organ relates to clonal traits, phenology and environmental gradients. New Phytol 245, 154–168 (2025).
Jiang, B. et al. Complex interactions of ‘water-light-heat’ climatic conditions on spring phenology in the mid-high latitudes of the Northern Hemisphere. Agric. For. Meteorol. 367, 110520 (2025).
Peaucelle, M. et al. Spatial variance of spring phenology in temperate deciduous forests is constrained by background climatic conditions. Nat Commun 10, 5388 (2019).
Delpierre, N. et al. Modelling interannual and spatial variability of leaf senescence for three deciduous tree species in France. Agric. For. Meteorol. 149, 938–948 (2009).
Hänninen, H. et al. Experiments are necessary in process-based tree phenology modelling. Trends Plant Sci 24, 199–209 (2019).
Gu, H. et al. Warming-induced increase in carbon uptake is linked to earlier spring phenology in temperate and boreal forests. Nat. Commun. 13, 3698 (2022).
Li, D., Belitz, M., Campbell, L. & Guralnick, R. Extreme weather events have strong but different impacts on plant and insect phenology. Nat. Clim. Chang. 15, 321–328 (2025).
Ge, Q., Wang, H., Rutishauser, T. & Dai, J. Phenological response to climate change in China: a meta-analysis. Glob. Chang. Biol. 21, 265–274 (2015).
USA National Phenology Network. Plant individual phenometrics data, 2009–2024. USA-NPN https://doi.org/10.5066/F78S4N1V (2024).
Leys, C., Ley, C., Klein, O., Bernard, P. & Licata, L. Detecting outliers: do not use standard deviation around the mean, use absolute deviation around the median. J. Exp. Soc. Psychol. 49, 764–766 (2013).
Kattge, J. et al. TRY – a global database of plant traits. Glob. Chang. Biol. 17, 2905–2935 (2011).
Kembel, S. W. et al. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26, 1463–1464 (2010).
Lu, C. et al. Diminishing warming effects on plant phenology over time. New Phytol 245, 523–533 (2025).
Adams, D. C., Gurevitch, J. & Rosenberg, M. S. Resampling tests for meta-analysis of ecological data. Ecology 78, 1277–1283 (1997).
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 36, 1–48 (2010).
Jin, Y. & Qian, H. V. PhyloMaker: an R package that can generate very large phylogenies for vascular plants. Ecography 42, 1353–1359 (2019).
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinformatics 20, 289–290 (2004).
Blomberg, S. P., Garland, T. & Ives, A. R. Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57, 717–745 (2003).
Lê, S., Josse, J. & Husson, F. FactoMineR: an R package for multivariate analysis. J. Stat. Softw. 25, 1–18 (2008).
Butler, E. E. et al. Mapping local and global variability in plant trait distributions. Proc. Natl. Acad. Sci. USA 114, E10937–E10946 (2017).
R. Core Team. R: A language and environment for statistical computing (R Foundation for Statistical Computing, 2025).
Xiong, K. Dataset and R code for ‘Acquisitive plants exhibit stronger phenological shifts in response to warming: insights from meta-analysis and long-term monitoring’. figshare https://doi.org/10.6084/m9.figshare.29917052 (2025).
Acknowledgements
This research was supported by the National Natural Science Foundation of China (Grant Nos. 32422055 (H.L.), 32130065 (H.L.) and 42125101 (C.W.)) and the National R&D Program of China (Grant No. 2023YFF0806800 (H.L.)). H.L. also acknowledges support from the Shanghai Rising-Star Program (Grant No. 23QA1402900) and Fundamental and Interdisciplinary Disciplines Breakthrough Plan of the Ministry of Education of China (JYB2025XDXM904). We would like to express our gratitude to all the authors of the published papers included in our meta-analysis, as well as to the contributors of the two long-term phenological datasets. We also thank the contributors of the TRY database, as well as the authors who provided the spatial distribution data of the traits used in Fig. 4.
Author information
Authors and Affiliations
Contributions
H.L. and C.W. conceived the study. K.X., H.Z., and H.L. conducted the analysis with inputs from C.L. and X.W. P.R., P.C., J.P., and C.W. provided significant revisions. K.X., C.W., and H.L. wrote the manuscript, with contributions from all co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xiong, K., Reich, P.B., Ciais, P. et al. Acquisitive plants exhibit stronger phenological shifts in response to warming: insights from meta-analysis and long-term monitoring. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70474-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70474-4