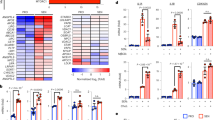
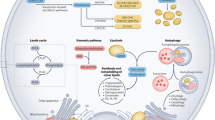
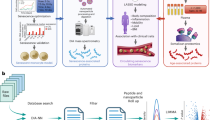
Abstract
Senescent cells (SnCs) are increasingly recognized as key contributors to osteoarthritis, with conventional strategies centered on their elimination. However, senolytic approaches face mounting limitations, driving the need for refined interventions. Here, we exploit SnCs’ lipid metabolic signature to develop a senotherapeutic strategy. Given the universal lipid accumulation in SnCs and the dual role of lipids as both metabolic liabilities and essential lubricants, we engineer an injectable nanoliposuction hydrogel platform designed for “waste recycling”. This system removes excess lipids to mitigate senescence-associated secretory phenotype (SASP) propagation and incorporates the harvested lipids as a pivotal component of the biomimetic lubricating system, thereby alleviating mechanical allodynia and cartilage wear. This strategy circumvents potential side effects associated with direct SnCs clearance and repurposes these traditionally harmful cells as functional resources. Our findings establish a shift in the philosophy of senescence therapy, transitioning the focus from destructive ablation to spatially adaptive reallocation of metabolites.
Similar content being viewed by others
Data availability
The main data supporting the findings of this study are presented in the paper and its supplementary information. The raw mass spectrometry proteomics data generated in this study have been deposited in the iProX database (https://www.iprox.cn/), under accession number IPX0012242000. Source data are provided with this paper.
References
Di Micco, R., Krizhanovsky, V., Baker, D. & d’Adda di Fagagna, F. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 22, 75–95 (2021).
Coryell, P. R., Diekman, B. O. & Loeser, R. F. Mechanisms and therapeutic implications of cellular senescence in osteoarthritis. Nat. Rev. Rheumatol. 17, 47–57 (2021).
Mehdizadeh, M., Aguilar, M., Thorin, E., Ferbeyre, G. & Nattel, S. The role of cellular senescence in cardiac disease: basic biology and clinical relevance. Nat. Rev. Cardiol. 19, 250–264 (2022).
Schmitt, C. A., Wang, B. & Demaria, M. Senescence and cancer—role and therapeutic opportunities. Nat. Rev. Clin. Oncol. 19, 619–636 (2022).
Sanfeliu-Redondo, D., Gibert-Ramos, A. & Gracia-Sancho, J. Cell senescence in liver diseases: pathological mechanism and theranostic opportunity. Nat. Rev. Gastroenterol. Hepatol. 21, 477–492 (2024).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: an expanding universe. Cell 186, 243–278 (2023).
Chaib, S., Tchkonia, T. & Kirkland, J. L. Cellular senescence and senolytics: the path to the clinic. Nat. Med. 28, 1556–1568 (2022).
Colville, A. et al. Death-seq identifies regulators of cell death and senolytic therapies. Cell Metab. 35, 1814–1829 (2023).
Jeon, O. et al. Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nat. Med. 23, 775–781 (2017).
de Magalhães, J. P. Cellular senescence in normal physiology. Science 384, 1300–1301 (2024).
Reyes, N. S. et al. Sentinel p16INK4a+ cells in the basement membrane form a reparative niche in the lung. Science 378, 192–201 (2022).
Wang, X. et al. Signalling by senescent melanocytes hyperactivates hair growth. Nature 618, 808–817 (2023).
Rabinovitch, M. Are senolytic agents guilty of overkill or inappropriate age discrimination?. Circulation 147, 667–668 (2023).
Reimann, M., Lee, S. & Schmitt, C. A. Cellular senescence: neither irreversible nor reversible. J. Exp. Med. 221, e20232136 (2024).
Hornburg, D. et al. Dynamic lipidome alterations associated with human health, disease and ageing. Nat. Metab. 5, 1578–1594 (2023).
Tsugawa, H. et al. A lipidome landscape of aging in mice. Nat. Aging 4, 709–726 (2024).
Byrns, C. N. et al. Senescent glia link mitochondrial dysfunction and lipid accumulation. Nature 630, 475–483 (2024).
Kaffe, E. et al. Bioactive signalling lipids as drivers of chronic liver diseases. J. Hepatol. 80, 140–154 (2024).
Zeng, Q. et al. Lipids and lipid metabolism in cellular senescence: Emerging targets for age-related diseases. Ageing Res. Rev. 97, 102294 (2024).
Jeon, Y. G., Kim, Y. Y., Lee, G. & Kim, J. B. Physiological and pathological roles of lipogenesis. Nat. Metab. 5, 735–759 (2023).
Yang, L., Zhao, X., Ma, Z., Ma, S. & Zhou, F. An overview of functional biolubricants. Friction 11, 23–47 (2023).
Gebeshuber, I. C. & Van Aken, G. Editorial: Friction and Lubricants Related to Human Bodies. Lubricants 2017, 4 (2017). 5.
DeMoya, C. D. et al. Advances in viscosupplementation and tribosupplementation for early-stage osteoarthritis therapy. Nat. Rev. Rheumatol. 20, 432–451 (2024).
Robinson, W. et al. Low-grade inflammation as a key mediator of the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 12, 580–592 (2016).
Seror, J. et al. Supramolecular synergy in the boundary lubrication of synovial joints. Nat. Commun. 6, 6497 (2015).
Zhou, Q. et al. Lipidomics unravels lipid changes in osteoarthritis articular cartilage. Ann. Rheum. Dis. S0003-4967(25)00054-8. Advance online publication (2025).
Wei, G. et al. Risk of metabolic abnormalities in osteoarthritis: a new perspective to understand its pathological mechanisms. Bone Res 11, 63 (2023).
Monnaert, V. et al. Behavior of alpha-, beta-, and gamma-cyclodextrins and their derivatives on an in vitro model of blood-brain barrier. J. Pharmacol. Exp. Ther. 310, 745–751 (2004).
Wang, B. et al. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat. Rev. Mol. Cell Biol. 25, 958-978 (2024).
Davies, M. E., Sharma, H. & Pigott, R. ICAM-1 expression on chondrocytes in rheumatoid arthritis: induction by synovial cytokines. Mediators Inflamm. 1, 71–74 (1992).
Suryadevara, V. et al. SenNet recommendations for detecting senescent cells in different tissues. Nat. Rev. Mol. Cell Biol. 25, 1001-1023 (2024).
Gorgoulis, V. et al. p53-Dependent ICAM-1 overexpression in senescent human cells identified in atherosclerotic lesions. Lab. Invest. 85, 502–511 (2005).
Swahn, H. et al. Senescent cell population with ZEB1 transcription factor as its main regulator promotes osteoarthritis in cartilage and meniscus. Ann. Rheum. Dis. 82, 403–415 (2023).
Fan, Y. et al. Unveiling inflammatory and prehypertrophic cell populations as key contributors to knee cartilage degeneration in osteoarthritis using multi-omics data integration. Ann. Rheum. Dis. 83, 926–944 (2024).
Zhang, B. C. et al. Cholesterol-binding motifs in STING that control endoplasmic reticulum retention mediate anti-tumoral activity of cholesterol-lowering compounds. Nat. Commun. 15, 2760 (2024).
Hu, X. et al. Bitter taste TAS2R14 activation by intracellular tastants and cholesterol. Nature 631, 459–466 (2024).
Roh, K. et al. Lysosomal control of senescence and inflammation through cholesterol partitioning. Nat. Metab. 5, 398–413 (2023).
Doktorova, M. et al. Preparation of asymmetric phospholipid vesicles for use as cell membrane models. Nat. Protoc. 13, 2086–2101 (2018).
Bozelli, J. C. Jr, Hou, Y. H. & Epand, R. M. Thermodynamics of methyl-β-cyclodextrin-induced lipid vesicle solubilization: effect of lipid headgroup and backbone. Langmuir 33, 13882–13891 (2017).
Lin, W. & Klein, J. Recent progress in cartilage lubrication. Adv. Mater. 33, e2005513 (2021).
Sett, R., Paul, B. K. & Guchhait, N. Unsaturation of the phospholipid side-chain influences its interaction with cyclodextrins: a spectroscopic exploration using a phenazinium dye. Colloids Surf. B. Biointerfaces 180, 150–158 (2019).
Szente, L. & Fenyvesi, É Cyclodextrin-lipid complexes: cavity size matters. Struct. Chem. 28, 479–492 (2017).
Abe, M. & Kobayashi, T. Imaging cholesterol depletion at the plasma membrane by methyl-β-cyclodextrin. J. Lipid Res. 62, 100077 (2021).
Lei, Y. et al. Injectable hydrogel microspheres with self-renewable hydration layers alleviate osteoarthritis. Sci. Adv. 8, eabl6449 (2022).
Bajpayee, A. & Grodzinsky, A. Cartilage-targeting drug delivery: can electrostatic interactions help?. Nat. Rev. Rheumatol. 13, 183–193 (2017).
Xie, R. et al. Biomimetic cartilage-lubricating polymers regenerate cartilage in rats with early osteoarthritis. Nat. Biomed. Eng. 5, 1189–1201 (2021).
Moiseeva, V. et al. Senescence atlas reveals an aged-like inflamed niche that blunts muscle regeneration. Nature 613, 169–178 (2023).
Shaw, P. X. et al. Complement factor H genotypes impact risk of age-related macular degeneration by interaction with oxidized phospholipids. Proc. Natl. Acad. Sci. USA 109, 13757–13762 (2012).
Liuzzi, R., Gallier, S., Ringler, S., Caserta, S. & Guido, S. Visualization of choline-based phospholipids at the interface of oil/water emulsions with TEPC-15 antibody. RSC Adv. 6, 109960–109968 (2016).
Lin, W. et al. Cartilage-inspired, lipid-based boundary-lubricated hydrogels. Science 370, 335–338 (2020).
Dolgin, E. Send in the senolytics. Nat. Biotechnol. 38, 1371–1377 (2020).
McHugh, D. & Gil, J. Senescence and aging: causes, consequences, and therapeutic avenues. J. Cell. Biol. 217, 65–77 (2018).
Zhao, J. et al. Senolytics cocktail dasatinib and quercetin alleviate chondrocyte senescence and facet joint osteoarthritis in mice. Spine J. 25, 184–198 (2025).
Gosset, M. et al. Primary culture and phenotyping of murine chondrocytes. Nat. Protoc. 3, 1253–1260 (2008).
Arra, M. et al. IκB-ζ signaling promotes chondrocyte inflammatory phenotype, senescence, and erosive joint pathology. Bone Res. 10, 12 (2022).
Ji, X. et al. GSTP1-mediated S-glutathionylation of Pik3r1 is a redox hub that inhibits osteoclastogenesis through regulating autophagic flux. Redox Biol. 61, 102635 (2023).
Kongpracha, P. et al. Simple but efficacious enrichment of integral membrane proteins and their interactions for in-depth membrane proteomics. Mol. Cell Proteom. 21, 100206 (2022).
Amor, C. et al. Senolytic CAR T cells reverse senescence-associated pathologies. Nature 583, 127–132 (2020).
Gu, Z., Eils, R. & Schlesner, M. Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinforma. Oxf. Engl. 32, 2847–2849 (2016).
Wu, T. et al. clusterProfiler 4.0: A universal enrichment tool for interpreting omics data. Innov. Camb. Mass 2, 100141 (2021).
Hao, Y. et al. Dictionary learning for integrative, multimodal and scalable single-cell analysis. Nat. Biotechnol. 42, 293–304 (2024).
Korsunsky, I. et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat. Methods 16, 1289–1296 (2019).
Acknowledgements
This work was supported by grants from the National Nature Science Fund of China (82201729 to Z.Q., 82501891 to J.H., 8251101103 to Safwat Adel Abdo Moqbel, 82072413 and 82101649 to Y.Q.), the Natural Science Foundation of Zhejiang Province (LMS25H060001 to Y.Q.), the Key R&D Program of Zhejiang Province (2021C03108 to G.F.), and the Joint Construction Project of Zhejiang Province and Health Ministry (WKJ-ZJ-2029 to G.F.). We thank Safwat Adel Abdo Moqbel for providing financial support during the revision of this article. We thank Professor Ruikang Tang from Zhejiang University for his invaluable help in clarifying and structuring the research ideas for this paper. We thank Ms. Xiaoye Li and the Center for Basic and Translational Research at the Second Affiliated Hospital of Zhejiang University School of Medicine for their laboratory management and technical support. We thank Xiaoxing Wu from the Small Animal Experiment Center of the Second Affiliated Hospital of Zhejiang University School of Medicine for her valuable assistance with our animal studies. We thank Turtle Tech Ltd. (Shanghai, China) for their technical guidance on PCR and for their assistance with primer design and validation. We thank Shanghai Applied Protein Technology Co., Ltd. for their help with proteomics, Jingyao Chen from the core facility plat form of Zhejiang University School of Medicine for their technical support and Shanghai Bioprofile Technology Co., Ltd. for their help with scRNA-seq analysis. We thank Wendi Chen and Jiawen Sun (from Scientific Compass www.shiyanjia.com) for providing invaluable assistance with the material mechanical property testing and the friction experiment related testing, respectively.
Author information
Authors and Affiliations
Contributions
X.J., X.H., P.T., Y.W., J.Z., K.X., C.L., X.W. and K.W. performed the experimental operations. X.J. and H.C. performed the analysis and visualization of the single-cell data. X.J., Z.Q., J.W. and Z.L. completed the design and preparation of MINH. X.J. and H.Z. completed the related experiments and analysis of p16-3MR mice. Y.Q., Z.Q., G.J., S.Y., W.L. and G.F. supervised this project. X.J. conceived and designed the study. X.J. and Y.Q. completed the conceptual integration and manuscript writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Fernando de la Cuesta and Jing Xie for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ji, X., He, X., Cai, H. et al. Recycling senescent cell lipids for targeted senotherapy. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70486-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70486-0