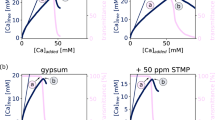
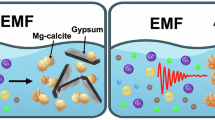
Abstract
Inorganic scaling, governed by complex organic–inorganic interactions, presents a pervasive challenge in aqueous environments with broad implications for engineering systems. Using reverse-osmosis (RO) desalination as a model platform, we investigate how mixed organic foulants influence inorganic gypsum scaling at membrane–water interfaces. Representative proteins, humic substances, and polysaccharides are employed as model foulants to reveal their roles in modulating gypsum crystallization behavior. By integrating advanced in situ, time-resolved synchrotron X-ray scattering within the concentration polarization layer—a region typically inaccessible to conventional characterization techniques—with modelling, spectroscopic, and imaging analyses, we track the evolution of gypsum scaling from nanoscale precursors to mature crystals. Our findings reveal that different classes of organic foulants regulate gypsum crystallization through distinct mechanisms, ranging from inhibiting precursor aggregation in the bulk solution to altering interfacial physicochemical properties that govern the kinetics of heterogeneous nucleation and growth. These findings provide molecular-level insights into the coupled dynamics of organic fouling and inorganic scaling, advancing mechanistic understanding of crystallization at functional interfaces. Such insights offer guidance for the rational design of anti-scaling strategies in engineering systems.
Similar content being viewed by others
Data availability
The data supporting the findings of the study are included in the main text and supplementary information files. Source data are provided with this paper.
References
Nancollas, G. & Reddy, M. The kinetics of crystallization of scale-forming minerals. Soc. Pet. Eng. J. 14, 117–126 (1974).
Bottrell, S. H. & Newton, R. J. Reconstruction of changes in global sulfur cycling from marine sulfate isotopes. Earth-Sci. Rev. 75, 59–83 (2006).
Fouke, B. W. et al. Depositional facies and aqueous-solid geochemistry of travertine-depositing hot springs (Angel Terrace, Mammoth Hot Springs, Yellowstone National Park, USA). J. Sediment. Res. 70, 565–585 (2000).
Elimelech, M. & Phillip, W. A. The future of seawater desalination: energy, technology, and the environment. science 333, 712–717 (2011).
Ahmed, M. A., Amin, S. & Mohamed, A. A. Fouling in reverse osmosis membranes: monitoring, characterization, mitigation strategies and future directions. Heliyon 9, e14908 (2023).
McCutcheon, J. R. & Mauter, M. S. Fixing the desalination membrane pipeline. Science 380, 242–244 (2023).
Løge, I. A., Anabaraonye, B. U. & Fosbøl, P. L. Growth mechanisms of composite fouling: The impact of substrates on detachment processes. Chem. Eng. J. 446, 137008 (2022).
Cai, Y.-H., Burkhardt, C. J. & Schäfer, A. I. Renewable energy powered membrane technology: Impact of osmotic backwash on organic fouling during solar irradiance fluctuation. J. Membr. Sci. 647, 120286 (2022).
Ennaceri, H., Fischer, K., Schulze, A. & Moheimani, N. R. Membrane fouling control for sustainable microalgal biodiesel production: A review. Renew. Sustain. Energy Rev. 161, 112335 (2022).
Cruz, C., Cisternas, L. A. & Kraslawski, A. Scaling problems and control technologies in industrial operations: Technology Assessment. Sep. Purif. Technol. 207, 20–27 (2018).
Meng, J. & Wang, S. Advanced antiscaling interfacial materials toward highly efficient heat energy transfer. Adv. Funct. Mater. 30, 1904796 (2020).
Jafari, M. et al. Cost of fouling in full-scale reverse osmosis and nanofiltration installations in the Netherlands. Desalination 500, 114865 (2021).
Van de Lisdonk, C., Van Paassen, J. & Schippers, J. Monitoring scaling in nanofiltration and reverse osmosis membrane systems. Desalination 132, 101–108 (2000).
Yin, Y. et al. The use of anti-scalants in gypsum scaling mitigation: Comparison with membrane surface modification and efficiency in combined reverse osmosis and membrane distillation. J. Membr. Sci. 643, 120077 (2022).
Hsieh, I.-M. & Malmali, M. Scaling behavior in membrane distillation: Effect of Biopolymers and Antiscalants. Water Res. 255, 121456 (2024).
Li, W. et al. Application of high-dose UV irradiation as nanofiltration pretreatment for drinking water production: Organic fouling mitigation and micropollutant removal. Water Res. 266, 122348 (2024).
Khan, I. A., Lee, Y. S. & Kim, J.-O. Chemically enhanced pretreatment (CEPT) to reduce irreversible fouling during the clean-in-place process for membranes operating under constant flux and constant pressure filtration. Desalination 549, 116313 (2023).
Hoang, T. A. in Water-formed deposits 13-47 (Elsevier, 2022).
Gibbs, J. W. On the equilibrium of heterogeneous substances. Am. J. Sci. 3, 441–458 (1878).
Lee, J., Yang, J., Kwon, S. G. & Hyeon, T. Nonclassical nucleation and growth of inorganic nanoparticles. Nat. Rev. Mater. 1, 1–16 (2016).
Van Driessche, A. E., Kellermeier, M., Benning, L. G. & Gebauer, D. in New Perspectives on Mineral Nucleation and Growth (Springer, 2017).
Gebauer, D. & Cölfen, H. Prenucleation clusters and non-classical nucleation. Nano Today 6, 564–584 (2011).
Anklam, M. R. & Firoozabadi, A. An interfacial energy mechanism for the complete inhibition of crystal growth by inhibitor adsorption. J. Chem. Phys. 123, 144708 (2005).
Lin, D. et al. Nanofiltration scaling influenced by coexisting pollutants considering the interaction between ferric coagulant and natural organic macromolecules. Chem. Eng. J. 413, 127403 (2021).
Jermann, D., Pronk, W. & Boller, M. Mutual influences between natural organic matter and inorganic particles and their combined effect on ultrafiltration membrane fouling. Environ. Sci. Technol. 42, 9129–9136 (2008).
Ji, J., Qiu, J., Wai, N., Wong, F.-S. & Li, Y. Influence of organic and inorganic flocculants on physical–chemical properties of biomass and membrane-fouling rate. Water Res. 44, 1627–1635 (2010).
Quay, A. N. et al. Combined organic fouling and inorganic scaling in reverse osmosis: role of protein–silica interactions. Environ. Sci. Technol. 52, 9145–9153 (2018).
Zhang, Y. et al. Inhibition of crystal nucleation and growth: a review. Cryst. Growth Des. 24, 2645–2665 (2024).
Jin, B., Liu, Z. & Tang, R. Recent experimental explorations of non-classical nucleation. CrystEngComm 22, 4057–4073 (2020).
Zang, S. et al. Direct observation and control of non-classical crystallization pathways in binary colloidal systems. Nat. Commun. 16, 3645 (2025).
Fenter, P. A., Rivers, M. L., Sturchio, N. & Sutton, S. R. Applications of synchrotron radiation in low-temperature geochemistry and environmental science. Vol. 49 (Walter de Gruyter GmbH & Co KG, 2018).
Liu, Y. et al. Progress and challenges in structural, in situ and operando characterization of single-atom catalysts by X-ray based synchrotron radiation techniques. Chem. Soc. Rev. 53, 11850–11887 (2024).
Deng, N. et al. Selenite and selenate sequestration during coprecipitation with barite: insights from mineralization processes of adsorption, nucleation, and growth. Environ. Sci. Technol. 56, 15518–15527 (2022).
Stawski, T. M. et al. Formation of calcium sulfate through the aggregation of sub-3 nanometre primary species. Nat. Commun. 7, 11177 (2016).
Pauw, B. R. Everything SAXS: small-angle scattering pattern collection and correction. J. Phys.: Condens. Matter 25, 383201 (2013).
Li, Y.-W. et al. BL19U2: Small-angle X-ray scattering beamline for biological macromolecules in solution at SSRF. Nucl. Sci. Tech. 31, 117 (2020).
Bishnoi, A., Kumar, S. & Joshi, N. in Microscopy methods in nanomaterials characterization 313-337 (Elsevier, 2017).
Toso, S., Baranov, D., Giannini, C., Marras, S. & Manna, L. Wide-angle x-ray diffraction evidence of structural coherence in CsPbBr3 nanocrystal superlattices. ACS Mater. Lett. 1, 272–276 (2019).
Yang, Z. et al. Visualization and semiquantitative study of the distribution of major components in wheat straw in mesoscopic scale using fourier transform infrared microspectroscopic imaging. Anal. Chem. 90, 7332–7340 (2018).
Luo, W., Arhatari, B., Gray, S. R. & Xie, M. Seeing is believing: Insights from synchrotron infrared mapping for membrane fouling in osmotic membrane bioreactors. Water Res. 137, 355–361 (2018).
Xie, M., Luo, W. & Gray, S. R. Synchrotron Fourier transform infrared mapping: A novel approach for membrane fouling characterization. Water Res. 111, 375–381 (2017).
Li, S. et al. Synchrotron FTIR mapping of mineralization in a microfluidic device. Lab a Chip 17, 1616–1624 (2017).
He, S., Oddo, J. E. & Tomson, M. B. The inhibition of gypsum and barite nucleation in NaCl brines at temperatures from 25 to 90. C. Appl. Geochem. 9, 561–567 (1994).
Huang, X., Li, C., Zuo, K. & Li, Q. Predominant Effect of Material Surface Hydrophobicity on Gypsum Scale Formation. Environ. Sci. Technol. 54, 15395–15404 (2020).
Dai, Z. et al. Gypsum scale formation and inhibition kinetics with implications in membrane system. Water Res 225, 119166 (2022).
Liu, S. & Nancollas, G. A kinetic and morphological study of the seeded growth of calcium sulfate dihydrate in the presence of additives. J. Colloid Interface Sci. 52, 593–601 (1975).
Cao, T., Rolf, J., Wang, Z., Violet, C. & Elimelech, M. Distinct impacts of natural organic matter and colloidal particles on gypsum crystallization. Water Res. 218, 118500 (2022).
Cody, A. & Cody, R. Evidence for micro-biological induction of {101} montmartre twinning of gypsum (CaSO4⋯ 2H2O). J. Cryst. Growth 98, 721–730 (1989).
Cody, A. & Cody, R. SEM and polarization analyzes updating early light microscope studies related to {101} twin formation in gypsum. J. Cryst. growth 98, 731–738 (1989).
He, S., Oddo, J. E. & Tomson, M. B. The nucleation kinetics of calcium sulfate dihydrate in NaCl solutions up to 6 m and 90 C. J. Colloid Interface Sci. 162, 297–303 (1994).
Choi, J. Y., Lee, T., Cheng, Y. & Cohen, Y. Observed crystallization induction time in seeded gypsum crystallization. Ind. Eng. Chem. Res. 58, 23359–23365 (2019).
Van Driessche, A. et al. The role and implications of bassanite as a stable precursor phase to gypsum precipitation. Science 336, 69–72 (2012).
Wang, Y.-W., Kim, Y.-Y., Christenson, H. K. & Meldrum, F. C. A new precipitation pathway for calcium sulfate dihydrate (gypsum) via amorphous and hemihydrate intermediates. Chem. Commun. 48, 504–506 (2012).
Prosa, T. J., Bauer, B. J., Amis, E. J., Tomalia, D. A. & Scherrenberg, R. A SAXS study of the internal structure of dendritic polymer systems. J. Polym. Sci. Part B: Polym. Phys. 35, 2913–2924 (1997).
Konishi, T. et al. Origin of SAXS intensity in the low-q region during the early stage of polymer crystallization from both the melt and glassy state. Phys. Rev. Mater. 2, 105602 (2018).
de Moor, P.-P. E., Beelen, T. P. & van Santen, R. A. SAXS/WAXS study on the formation of precursors and crystallization of silicalite. Microporous Mater. 9, 117–130 (1997).
Rabizadeh, T., Stawski, T. M., Morgan, D. J., Peacock, C. L. & Benning, L. G. The effects of inorganic additives on the nucleation and growth kinetics of calcium sulfate dihydrate crystals. Cryst. Growth Des. 17, 582–589 (2017).
Saha, A. et al. New Insights into the transformation of calcium sulfate hemihydrate to gypsum using time-resolved cryogenic transmission electron microscopy. Langmuir 28, 11182–11187 (2012).
Tong, T., Wallace, A. F., Zhao, S. & Wang, Z. Mineral scaling in membrane desalination: Mechanisms, mitigation strategies, and feasibility of scaling-resistant membranes. J. Membr. Sci. 579, 52–69 (2019).
Horseman, T. et al. Wetting, scaling, and fouling in membrane distillation: state-of-the-art insights on fundamental mechanisms and mitigation strategies. ACS EST Eng. 1, 117–140 (2020).
Yin, Y. et al. Which surface is more scaling resistant? A closer look at nucleation theories for heterogeneous gypsum nucleation in aqueous solutions. Environ. Sci. Technol. 56, 16315–16324 (2022).
Van Santen, R. The Ostwald step rule. J. Phys. Chem. 88, 5768–5769 (1984).
Hu, C., Wang, X., Qi, Z. & Li, C. The new infrared beamline at NSRL. Infrared Phys. Technol. 105, 103200 (2020).
Hua, W.-Q. et al. Time-resolved ultra-small-angle X-ray scattering beamline (BL10U1) at SSRF. Nucl. Sci. Tech. 35, 36 (2024).
Zeng, J. et al. Performance on absolute scattering intensity calibration and protein molecular weight determination at BL16B1, a dedicated SAXS beamline at SSRF. J. Synchrotron Radiat. 24, 509–520 (2017).
Basham, M. et al. Data analysis workbench (DAWN). J. Synchrotron Radiat. 22, 853–858 (2015).
Hopkins, J. B., Gillilan, R. E. & Skou, S. BioXTAS RAW: improvements to a free open-source program for small-angle X-ray scattering data reduction and analysis. J. Appl. Crystallogr. 50, 1545–1553 (2017).
Ashiotis, G. et al. The fast azimuthal integration Python library: pyFAI. J. Appl. Crystallogr. 48, 510–519 (2015).
Acknowledgements
We gratefully acknowledge the financial support from the National Science Foundation of China (52170094 and 22576197 to X.L.) and the Fundamental Research Funds for the Central Universities (to X.L.). We thank the National Facility for Protein Science in Shanghai of BL19U2 for providing technical support and assistance in SAXS/WAXS measurements. We also thank the Shanghai Synchrotron Radiation Facility of BL10U1 and BL16B1 for the assistance on USAXS/SAXS/WAXS measurements. We thank the National Synchrotron Radiation Laboratory of BL01B for the assistance on infrared mapping measurements. Some material characterization was carried out at the University of Science and Technology of China (USTC) Center for Micro- and Nanoscale Research and Fabrication, the Instruments Center for Physical Science at USTC, and the First Affiliated Hospital of USTC. We are grateful to Mr. Qingbo Xu at USTC for his assistance with the detector calibration for SAXS and WAXS measurements.
Author information
Authors and Affiliations
Contributions
X.L. conceived the idea. Z.F. and X.L. designed experiments. Z.F., S.X., and Z.R. performed experiments. J.C. conducted crystallographic analysis. Z.F., S.X., J.C., Y.Y., J.J., X.F., and X.L. contributed to the data interpretation. Z.F., Z.R., and X.L. wrote the first draft. X.L., Z.F., S.X., and Y.Y. revised the manuscript. All the authors discussed the results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Wenhai Luo, Yuelian Peng, Zhengyang Huo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Feng, Z., Xu, S., Cao, J. et al. In situ synchrotron X-ray scattering reveals organic-mediated scaling mechanisms on desalination membranes. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70508-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70508-x