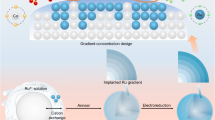
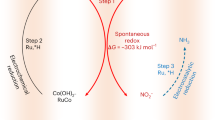
Abstract
Geen ammonia synthesis through electrochemical nitrate reduction (e-NO3R) using cost-effective Co-based catalysts is promising, but the inevitable structural evolution induced by reductive potentials compromises long-term stability and hinders practical implementation. Focusing on β-Co(OH)2, which serves as the active phase in e-NO3R systems, this work combines experimental analysis and computational studies to reveal a dynamic surface *OH evolution process: *OH cleavage under negative potentials and *OH generation by the dissociation of NO3-. Notably, Ru nanoparticles anchored on β-Co(OH)2 nanosheets promote structural evolution by facilitating *OH cleavage and generation, thereby sustaining a highly active and selective OH-terminated surface. Simultaneously, Ru provides moderate *H adsorption, accelerates the conversions from NO3− to NO2− and from NO2− to NH3, and thus enhances ammonia synthesis. The optimized Co(OH)2-Ru catalyst achieves an ammonia yield of 98 ± 0.91 mg·h-1·cm-2 with a Faradaic efficiency (FE) of 97.7 ± 0.90 % at −0.7 V versus reversible hydrogen electrode (vs. RHE), while maintaining NH3 FEs above 95% across a broad potential window. This work elucidates structural evolution dynamics, offering a design principle for robust electrocatalysts.
Similar content being viewed by others
Data availability
All the data supporting the findings of this study are available within the article and its Supplementary Information files. Source data are provided with this paper.
References
Chen, F.-Y. et al. Electrochemical nitrate reduction to ammonia with cation shuttling in a solid electrolyte reactor. Nat. Catal. 7, 1032–1043 (2024).
Yu, Y.-Z., Cheng, Y., Cheng, S. & Wu, Z.-Y. Advanced ruthenium-based electrocatalysts for no reduction to ammonia. Adv. Mater. 37, 2412363 (2025).
MacFarlane, D. R. et al. A roadmap to the ammonia economy. Joule 4, 1186–1205 (2020).
Xia, R. et al. Electrochemical oxidation of nitric oxide to concentrated nitric acid with carbon-based catalysts at near-ambient conditions. Nat. Catalysis, https://doi.org/10.1038/s41929-025-01315-8 (2025).
Liu, D. et al. Recent advances in electrocatalysts for efficient nitrate reduction to ammonia. Adv. Funct. Mater. 33, 2303480 (2023).
Li, J. et al. Efficient ammonia electrosynthesis from nitrate on strained ruthenium nanoclusters. J. Am. Chem. Soc. 142, 7036–7046 (2020).
Chen, F.-Y. et al. Efficient conversion of low-concentration nitrate sources into ammonia on a Ru-dispersed Cu nanowire electrocatalyst. Nat. Nanotechnol. 17, 759–767 (2022).
Wang, Y. et al. Enhanced nitrate-to-ammonia activity on copper–nickel alloys via tuning of intermediate adsorption. J. Am. Chem. Soc. 142, 5702–5708 (2020).
Zhang, S. et al. Fe/Cu diatomic catalysts for electrochemical nitrate reduction to ammonia. Nat. Commun. 14, 3634 (2023).
Liu, D. et al. Electrocatalytic reduction of nitrate to ammonia on low-cost manganese-incorporated Co3O4 nanotubes. Appl. Catal. B: Environ. 324, 122293 (2023).
Qiao, L. et al. Nickel-facilitated in-situ surface reconstruction on spinel Co3O4 for enhanced electrochemical nitrate reduction to ammonia. Appl. Catal. B: Environ. 340, 123219 (2024).
Feng, J., Wang, X. & Pan, H. In-situ Reconstruction of Catalyst in Electrocatalysis. Adv. Mater. 36, 2411688 (2024).
Zhu, A. et al. Facet-dependent evolution of active components on spinel Co3O4 for electrochemical ammonia synthesis. ACS Nano 18, 22344–22355 (2024).
Fan, Z. et al. Interfacial electronic interactions promoted activation for nitrate electroreduction to ammonia over Ag-modified Co3O4. Angew. Chem. Int. Ed. 63, e202410356 (2024).
Han, S. et al. Ultralow overpotential nitrate reduction to ammonia via a three-step relay mechanism. Nat. Catal. 6, 402–414 (2023).
Qiao, L. et al. In situ reconstructed cu/β-Co(OH)2 tandem catalyst for enhanced nitrate electroreduction to ammonia in ampere-level. Adv. Energy Mater. 14, 2402805 (2024).
Wu, S. et al. Ag-Co3O4-CoOOH-nanowires tandem catalyst for efficient electrocatalytic conversion of nitrate to ammonia at low overpotential via triple reactions. Adv. Sci. 10, 2303789 (2023).
Niu, Z. et al. Bifunctional copper-cobalt spinel electrocatalysts for efficient tandem-like nitrate reduction to ammonia. Chem. Eng. J. 450, 138343 (2022).
Gong, Z. et al. Modulating metal-nitrogen coupling in anti-perovskite nitride via cation doping for efficient reduction of nitrate to ammonia. Angew. Chem. Int. Ed. 62, e202308775 (2023).
He, W. et al. Splicing the active phases of copper/cobalt-based catalysts achieves high-rate tandem electroreduction of nitrate to ammonia. Nat. Commun. 13, 1129 (2022).
Zhang, J. et al. Single-entity Electrochemistry Unveils Dynamic Transformation during Tandem Catalysis of Cu2O and Co3O4 for Converting NO3− to NH3. Angew. Chem. Int. Ed. 62, e202214830 (2023).
He, W. et al. Enhanced nitrate-to-ammonia efficiency over linear assemblies of copper-cobalt nanophases stabilized by redox polymers. Adv. Mater. 35, 2303050 (2023).
Zhang, Z. et al. Electrochemical deposition as a universal route for fabricating single-atom catalysts. Nat. Commun. 11, 1215 (2020).
Li, Z. et al. Seed-assisted formation of NiFe anode catalysts for anion exchange membrane water electrolysis at industrial-scale current density. Nat. Catal. 7, 944–952 (2024).
Zhao, S. et al. Constructing regulable supports via non-stoichiometric engineering to stabilize ruthenium nanoparticles for enhanced pH-universal water splitting. Nat. Commun. 15, 2728 (2024).
Gao, P. et al. Understanding the synergistic effects and structural evolution of Co(OH)2 and Co3O4 toward Boosting electrochemical charge storage. Adv. Funct. Mater. 32, 2108644 (2022).
Sanchis-Gual, R. et al. Crystallographic and geometrical dependence of water oxidation activity in Co-based layered hydroxides. ACS Catal. 13, 10351–10363 (2023).
Ze, H. et al. In situ probing the structure change and interaction of interfacial water and hydroxyl intermediates on Ni(OH)2 surface over water splitting. J. Am. Chem. Soc. 146, 12538–12546 (2024).
Chong, L. et al. La- and Mn-doped cobalt spinel oxygen evolution catalyst for proton exchange membrane electrolysis. Science 380, 609–616 (2023).
Persson, K. A., Waldwick, B., Lazic, P. & Ceder, G. Prediction of solid-aqueous equilibria: Scheme to combine first-principles calculations of solids with experimental aqueous states. Phys. Rev. B 85, 235438 (2012).
Chen, J. et al. Reversible hydrogen spillover in Ru-WO3-x enhances hydrogen evolution activity in neutral pH water splitting. Nat. Commun. 13, 5382 (2022).
Li, J., Ma, Y., Ho, J. C. & Qu, Y. Hydrogen spillover phenomenon at the interface of metal-supported electrocatalysts for hydrogen evolution. Acc. Chem. Res. 57, 895–904 (2024).
Liu, R. et al. Defect sites in ultrathin pd nanowires facilitate the highly efficient electrochemical hydrodechlorination of pollutants by H*ads. Environ. Sci. Technol. 52, 9992–10002 (2018).
Badea, G. E. Electrocatalytic reduction of nitrate on copper electrode in alkaline solution. Electrochim. Acta 54, 996–1001 (2009).
Gong, M. et al. Nanoscale nickel oxide/nickel heterostructures for active hydrogen evolution electrocatalysis. Nat. Commun. 5, 4695 (2014).
Liang, S., Teng, X., Xu, H., Chen, L. & Shi, J. H* Species Regulation by Mn-Co(OH)2 for efficient nitrate electro-reduction in neutral solution. Angew. Chem. 136, e202400206 (2024).
Li, D. et al. Engineering ruthenium species on metal–organic frameworks for water electrolysis at industrial current densities. Adv. Energy Mater. 15, 2404714 (2025).
Wen, Q. et al. Engineering a local free water enriched microenvironment for surpassing platinum hydrogen evolution activity. Angew. Chem. Int. Ed. 61, e202206077 (2022).
Li, Y. et al. Enhancement of nitrate-to-ammonia on amorphous CeOx-modified Cu via tuning of active hydrogen supply. Adv. Energy Mater. 14, 2303863 (2024).
Guo, X. et al. Controlled defective engineering on cuir catalyst promotes nitrate selective reduction to ammonia. ACS Nano 18, 24252–24261 (2024).
Wang, M. et al. Polynuclear cobalt cluster-based coordination polymers for efficient nitrate-to-ammonia electroreduction. J. Am. Chem. Soc. 146, 20439–20448 (2024).
Xu, J. et al. Breaking local charge symmetry of iron single atoms for efficient electrocatalytic nitrate reduction to ammonia. Angew. Chem. Int. Ed. 62, e202308044 (2023).
Hao, R. et al. Elucidation of the electrocatalytic activity origin of Fe3C species and application in the NOx full conversion to valuable ammonia. Chem. Eng. J. 467, 143371 (2023).
Kohn, W. & Sham, L. J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 140, A1133–A1138 (1965).
Hohenberg, P. & Kohn, W. Inhomogeneous electron gas. Phys. Rev.136, B864–B871 (1964).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initiototal-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Li, F., Ai, H., Liu, D., Lo, K. H. & Pan, H. Enhanced oxygen evolution reaction on 2D CoOOH via strain engineering: an insightful view from spin state transition. J. Mater. Chem. A. https://doi.org/10.1039/d1ta03412j (2021).
Friebel, D. et al. Identification of Highly Active Fe Sites in (Ni,Fe)OOH for Electrocatalytic Water Splitting. J. Am. Chem. Soc. 137, 1305–1313 (2015).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
vasp_raman.py. (https://github.com/raman-sc/VASP/, 2013).
Acknowledgements
Dr. H. Pan acknowledges support from the Science and Technology Development Fund (FDCT) from Macau SAR (0050/2023/RIB2, 0023/2023/AFJ, 0002/2024/TFP, and 0111/2022/A2), and Multi-Year Research Grants (MYRG-GRG2025-00007-IAPME and MYRG-GRG2024-00038-IAPME) from the University of Macau. Dr. W. F. Ip acknowledges support from the Science and Technology Development Fund (0087/2024/AFJ). The DFT calculations are performed at the High-Performance Computing Cluster (HPCC) of the Information, Communication Technology Office (ICTO) at the University of Macau.
Author information
Authors and Affiliations
Contributions
D.L., H.B. and M.C. contributed equally to this manuscript. D.L. and H.P. conceived the idea. D.L. and M.C. contributed to the experimental design, material synthesis, electrochemical measurements, data analysis, TEM and XAFS characterization, and drafting of the initial manuscript. H.B. and D.L. designed and performed the theoretical calculations and also contributed to writing the initial draft. S.P., J.K., L.L., Z.F., and C.L. assisted with XRD and XPS characterization and contributed to editing the draft. W.I. and H.P. provided overall supervision of the project and revised the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yijun Yang and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, D., Bai, H., Chen, M. et al. Unravelling the Ru-promoted dynamic evolution of Cobalt hydroxide during nitrate reduction towards ammonia production. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70531-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70531-y