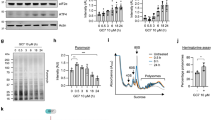
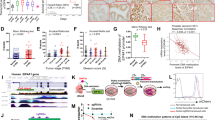
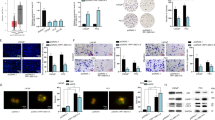
Abstract
Protein synthesis plays a central role in cancer development and progression. eukaryotic initiation factor 5 A (eIF5A), a translation factor activated by hypusination, is implicated in tumorigenesis, however, its mode of action is still unclear. We find that hypusinated eIF5A (eIF5Ahyp) promotes metastasis and tumor growth in prostate cancer (PCa) by supporting mitochondrial metabolism and translation. eIF5Ahyp controls the subcellular localization of Mitochondrial Ribonuclease P Protein 3 (MRPP3) mRNA encoding a protein essential for mitochondrial tRNA (mt-tRNA) maturation. We show that eIF5Ahyp regulates the nuclear export of MRPP3 mRNA, its expression, thereby promoting mt-tRNA maturation. Our findings establish that MRPP3 enhances mitochondrial metabolism and supports PCa metastasis. Importantly, its expression restores mitochondrial translation and tumor growth inhibited by the downregulation of eIF5Ahyp. Together, we uncover a critical role for eIF5Ahyp in mitochondrial protein synthesis and highlight its broader implications in coordinating the expression of nuclear and mitochondrial genomes, linking hypusination to cancer progression.
Similar content being viewed by others
Data availability
All data are available from the corresponding author upon request. The Proteomic data generated in this study have been deposited in the PRIDE database under accession code PXD058627 PRIDE - PRoteomics IDEntifications Database. The Polysome Sequencing data and RNASeq data generated in this study have been deposited in the GEO database under the accession number GSE313170 https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE313170. Raw steady-state metabolomic and isotope tracing data are provided in the Source Data File and Supplementary files. The public databases GSE70770 and GSE193337 were used in Fig. 1 and Supplementary Fig. 4. GSE70770 was analyzed with Phantasus and GSE193337 with Rstudio and Seurat. Source data are provided in this paper.
References
Kovalski, J. R., Kuzuoglu-Ozturk, D. & Ruggero, D. Protein synthesis control in cancer: selectivity and therapeutic targeting. EMBO J. 41, e109823 (2022).
Schuller, A. P., Wu, C. C., Dever, T. E., Buskirk, A. R. & Green, R. eIF5A Functions Globally in Translation Elongation and Termination. Mol. Cell 66, 194–205 (2017).
Clement, P. M., Johansson, H. E., Wolff, E. C. & Park, M. H. Differential expression of eIF5A-1 and eIF5A-2 in human cancer cells. FEBS J. 273, 1102–1114 (2006).
Manjunath, H. et al. Suppression of ribosomal pausing by eIF5A is necessary to maintain the fidelity of start codon selection. Cell Rep. 29, 3134–3146 (2019).
Casero, R. A. Jr., Murray Stewart, T. & Pegg, A. E. Polyamine metabolism and cancer: treatments, challenges and opportunities. Nat. Rev. Cancer 18, 681–695 (2018).
Kaminski, L. et al. PGC1alpha Inhibits polyamine synthesis to suppress prostate cancer aggressiveness. Cancer Res. 79, 3268–3280 (2019).
Zabala-Letona, A. et al. mTORC1-dependent AMD1 regulation sustains polyamine metabolism in prostate cancer. Nature 547, 109–113 (2017).
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Fabbri, L., Chakraborty, A., Robert, C. & Vagner, S. The plasticity of mRNA translation during cancer progression and therapy resistance. Nat. Rev. Cancer 21, 558–577 (2021).
Li, G. W., Burkhardt, D., Gross, C. & Weissman, J. S. Quantifying absolute protein synthesis rates reveals principles underlying allocation of cellular resources. Cell 157, 624–635 (2014).
Puleston, D. J. et al. Polyamines and eIF5A hypusination modulate mitochondrial respiration and macrophage activation. Cell Metab. 30, 352–363 (2019).
Jiang, X. et al. P53-dependent hypusination of eIF5A affects mitochondrial translation and senescence immune surveillance. Nat. Commun. 15, 7458 (2024).
Suomalainen, A. & Nunnari, J. Mitochondria at the crossroads of health and disease. Cell 187, 2601–2627 (2024).
Papier, O., Minor, G., Medini, H. & Mishmar, D. Coordination of mitochondrial and nuclear gene-expression regulation in health, evolution, and disease. Curr. Opin. Physiol. 27, 100554 (2022).
Ross-Adams, H. et al. Integration of copy number and transcriptomics provides risk stratification in prostate cancer: A discovery and validation cohort study. EBioMedicine. 2, 1133–1144 (2015).
Heidegger, I. et al. Comprehensive characterization of the prostate tumor microenvironment identifies CXCR4/CXCL12 crosstalk as a novel antiangiogenic therapeutic target in prostate cancer. Mol. Cancer 21, 132 (2022).
Grasso, C. S. et al. The mutational landscape of lethal castration-resistant prostate cancer. Nature 487, 239–243 (2012).
Lapointe, J. et al. Gene expression profiling identifies clinically relevant subtypes of prostate cancer. Proc. Natl. Acad. Sci. USA 101, 811–816 (2004).
Varambally, S. et al. Integrative genomic and proteomic analysis of prostate cancer reveals signatures of metastatic progression. Cancer Cell 8, 393–406 (2005).
Cortazar, A. R. et al. CANCERTOOL: A visualization and representation interface to exploit cancer datasets. Cancer Res. 78, 6320–6328 (2018).
Beltran, H. et al. Divergent clonal evolution of castration-resistant neuroendocrine prostate cancer. Nat. Med. 22, 298–305 (2016).
Sun, R. et al. Proteomic landscape profiling of primary prostate cancer reveals a 16-protein panel for prognosis prediction. Cell Rep. Med. 5, 101679 (2024).
Gao, D. et al. Organoid cultures derived from patients with advanced prostate cancer. Cell 159, 176–187 (2014).
Newman, R. H. & Zhang, J. Fucci: street lights on the road to mitosis. Chem. Biol. 15, 97–98 (2008).
Dirat, B. et al. Inhibition of the GTPase Rac1 mediates the antimigratory effects of metformin in prostate cancer cells. Mol. Cancer Ther. 14, 586–596 (2015).
Bakht, M. K. et al. Landscape of prostate-specific membrane antigen heterogeneity and regulation in AR-positive and AR-negative metastatic prostate cancer. Nat. Cancer 4, 699–715 (2023).
Loubiere, C. et al. The energy disruptor metformin targets mitochondrial integrity via modification of calcium flux in cancer cells. Sci. Rep. 7, 5040 (2017).
Perdiz, D., Oziol, L. & Poüs, C. Early mitochondrial fragmentation is a potential in vitro biomarker of environmental stress. Chemosphere 223, 577–587 (2019).
Therizols, G. et al. Alteration of ribosome function upon 5-fluorouracil treatment favors cancer cell drug-tolerance. Nat. Commun. 13, 173 (2022).
Lightowlers, R. N. & Chrzanowska-Lightowlers, Z. M. Human pentatricopeptide proteins: only a few and what do they do?. RNA Biol. 10, 1433–1438 (2013).
Münch, C. & Harper, J. W. Mitochondrial unfolded protein response controls matrix pre-RNA processing and translation. Nature 534, 710–713 (2016).
Rackham, O. et al. Hierarchical RNA processing is required for mitochondrial ribosome assembly. Cell Rep. 16, 1874–1890 (2016).
Rosorius, O. et al. Nuclear pore localization and nucleocytoplasmic transport of eIF-5A: evidence for direct interaction with the export receptor CRM1. J. Cell Sci. 112, 2369–2380 (1999).
Maier, B. et al. The unique hypusine modification of eIF5A promotes islet beta cell inflammation and dysfunction in mice. J. Clin. Invest. 120, 2156–2170 (2010).
Xu, A., Jao, D. L. & Chen, K. Y. Identification of mRNA that binds to eukaryotic initiation factor 5A by affinity co-purification and differential display. Biochem. J. 384, 585–590 (2004).
Coni, S. et al. Blockade of EIF5A hypusination limits colorectal cancer growth by inhibiting MYC elongation. Cell Death Dis. 11, 1045 (2020).
Balasubramanian, S. K., Azmi, A. S. & Maciejewski, J. Selective inhibition of nuclear export: a promising approach in the shifting treatment paradigms for hematological neoplasms. Leukemia 36, 601–612 (2022).
Lee, S. B. et al. The effect of hypusine modification on the intracellular localization of eIF5A. Biochem. Biophys. Res. Commun. 383, 497–502 (2009).
Devens, B. H., Weeks, R. S., Burns, M. R., Carlson, C. L. & Brawer, M. K. Polyamine depletion therapy in prostate cancer. Prostate Cancer Prostatic Dis. 3, 275–279 (2000).
Muramatsu, T. et al. The hypusine cascade promotes cancer progression and metastasis through the regulation of RhoA in squamous cell carcinoma. Oncogene 35, 5304–5316 (2016).
Fujimura, K. et al. A hypusine-eIF5A-PEAK1 switch regulates the pathogenesis of pancreatic cancer. Cancer Res. 74, 6671–6681 (2014).
Güth, R. et al. DHPS-dependent hypusination of eIF5A1/2 is necessary for TGFβ/fibronectin-induced breast cancer metastasis and associates with prognostically unfavorable genomic alterations in TP53. Biochem. Biophys. Res. Commun. 519, 838–845 (2019).
Coni, S. et al. Combined inhibition of polyamine metabolism and eIF5A hypusination suppresses colorectal cancer growth through a converging effect on MYC translation. Cancer Lett. 559, 216120 (2023).
Gobert, A. P. et al. Hypusination maintains intestinal homeostasis and prevents colitis and carcinogenesis by enhancing aldehyde detoxification. Gastroenterology https://doi.org/10.1053/j.gastro.2023.05.041 (2023).
Landau, G., Bercovich, Z., Park, M. H. & Kahana, C. The role of polyamines in supporting growth of mammalian cells is mediated through their requirement for translation initiation and elongation. J. Biol. Chem. 285, 12474–12481 (2010).
Ivanov, I. P. et al. Polyamine control of translation elongation regulates start site selection on antizyme inhibitor mRNA via ribosome queuing. Mol. Cell 70, 254–264.e256 (2018).
Pelechano, V. & Alepuz, P. eIF5A facilitates translation termination globally and promotes the elongation of many non polyproline-specific tripeptide sequences. Nucleic Acids Res. 45, 7326–7338 (2017).
Liu, Y. et al. The androgen receptor regulates a druggable translational regulon in advanced prostate cancer. Sci. Transl. Med. 11, https://doi.org/10.1126/scitranslmed.aaw4993 (2019).
Ishfaq, M. et al. Acetylation regulates subcellular localization of eukaryotic translation initiation factor 5A (eIF5A). FEBS Lett. 586, 3236–3241 (2012).
Kruse, M. et al. Inhibition of CD83 cell surface expression during dendritic cell maturation by interference with nuclear export of CD83 mRNA. J. Exp. Med. 191, 1581–1590 (2000).
Muneretto, G., Plazzi, F. & Passamonti, M. Mitochondrion-to-nucleus communication mediated by RNA export: a survey of potential mechanisms and players across eukaryotes. Biol. Lett. 20, 20240147 (2024).
Carbajosa, G., Ali, A. T. & Hodgkinson, A. Identification of human mitochondrial RNA cleavage sites and candidate RNA processing factors. BMC Biol. 20, 168 (2022).
Rossetti, G. et al. A common genetic variant of a mitochondrial RNA processing enzyme predisposes to insulin resistance. Sci. Adv. 7, eabi7514 (2021).
Hochberg, I. et al. Bi-allelic variants in the mitochondrial RNase P subunit PRORP cause mitochondrial tRNA processing defects and pleiotropic multisystem presentations. Am. J. Hum. Genet. 108, 2195–2204 (2021).
Ben Sahra, I. et al. The antidiabetic drug metformin exerts an antitumoral effect in vitro and in vivo through a decrease of cyclin D1 level. Oncogene 27, 3576–3586 (2008).
Demichev, V., Messner, C. B., Vernardis, S. I., Lilley, K. S. & Ralser, M. DIA-NN: neural networks and interference correction enable deep proteome coverage in high throughput. Nat. Methods 17, 41–44 (2020).
Gerault, M. A., Granjeaud, S., Camoin, L., Nordlund, P. & Dai, L. IMPRINTS.CETSA and IMPRINTS.CETSA.app: an R package and a Shiny application for the analysis and interpretation of IMPRINTS-CETSA data. Brief. Bioinform. 25, https://doi.org/10.1093/bib/bbae128 (2024).
Balukoff, N. C. et al. A translational program that suppresses metabolism to shield the genome. Nat. Commun. 11, 5755 (2020).
Kleverov, M. et al. Phantasus, a web application for visual and interactive gene expression analysis. ELife 13, https://doi.org/10.7554/eLife.85722 (2024).
Acknowledgements
We thank Myriam Bost and Sophie Giorgetti-Peraldi for their assistance with graphic support and Dr. Els Verhoyen for her help. The authors would like to thank Véronique Corcelle and the animal facility team for their essential assistance in carrying out the in vivo experiments. This work was supported by La Ligue Nationale contre le Cancer (Equipe Labellisée 2022), l’Association pour la Recherche sur les Tumeurs de la Prostate (ARTP), ITMO-Cancer and the French Institute of Cancer (INCa, PLBio 25-062 and 22-095). The authors acknowledge MET’CONNECT, a Structuring Action financed by the French National Cancer Institute (INCa), the Région Sud, and the Canceropôle Provence-Alpes-Côte d’Azur. M.K. is supported by the French Ministry of Research and La Ligue Nationale contre le Cancer. AB is supported by a grant from the Foundation Max et Yvonne de Foras. M.C. was supported by the foundation ARC. F.B. and N.M.M. are CNRS investigators. Metabolomics services were performed by the Metabolomics Core Facility at Robert H. Lurie Comprehensive Cancer Center of Northwestern University. IB-S is supported by the National Institutes of Health (NIH) grants R01GM135587 and R01GM143334. This work was supported by the “Fondation ARC pour la Recherche sur le Cancer” PGA for the project TRANSLATOL N°ARCPGA12021020003052_3561 and LYriCAN+ INCa-DGOS-INSERM-ITMO cancer_18003 (for J.-J.D. and A.V.). This article is based upon work from COST Action TRANSLACORE CA21154, supported by COST (European Cooperation in Science and Technology). Cytometry Samples acquisition and data analysis were performed on the C3M Cytometry Core Facility, financed by Conseil Départemental CD06 and Conseil Régional PACA. We sincerely thank the GIS-IBISA multi-sites platform Microscopy Imagery Côte d’Azur (MICA), and particularly the imaging site of C3M (INSERM U1065), supported by INSERM, Cancéropôle PACA, Conseil Régional SUD, Conseil Départemental 06, and IBISA. This work has been supported by the French government, through the France 2030 investment plan managed by the Agence Nationale de la Recherche, as part of the Université Côte d’Azur’s Initiative of Excellence, reference ANR-15-IDEX-01.
Author information
Authors and Affiliations
Contributions
M.K., A.M., L.B., M.P.-M., V.T., A.V., J.M., L.T., P.K., M.I., E.T., C.B., P.B., H.S.C., F.L., E.B., S.A., L.C., M.C., D.A., and A.J. performed experiments and analysis. M.K., A.M., L.B., A.V., F.L., and F.B. designed the experiments. M.K., L.B., V.T., L.T., O.B., A.B., F.L., S.A., D.F.P., S.B., L.C., M.C., M.D., D.A., Y.C., J.J.D., I.B.-S., N.M.M., P.P., and F.B. provided materials and expertise. M.K., N.M.M., P.P., and F.B. wrote the manuscript and designed the figures.
Corresponding author
Ethics declarations
Competing interests
Authors declare no competing interests in this study.
Peer review
Peer review information
Nature Communications thanks Alain Gobert and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kahi, M., Mazzu′, A., Batistic, L. et al. Hypusination of the translation factor eIF5A regulates mitochondrial tRNA processing to promote prostate cancer aggressiveness. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70566-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70566-1