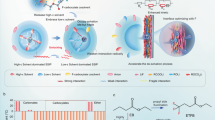
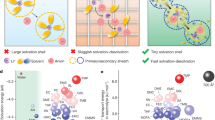
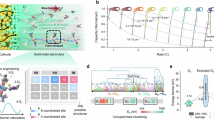
Abstract
The limitations of ion transport kinetics in conventional electrolytes, particularly under extreme operating conditions, arise from suboptimal solvation structures and inefficient charge carrier utilization. Here, we present strategic electrolyte design that reconfigures Li⁺ coordination geometry by modulating intermolecular interactions and solvent molecule volume, fundamentally overcoming these transport constraints. By incorporating an optimized moderator with a low dipole moment and small molecular size, extensive anion aggregation is effectively disrupted into compact ion conduction domains, simultaneously increasing the number of free charge carriers and enhancing ion mobility. Guided by this principle, the designed electrolyte with dichloromethane (85.11 Å, 2.36 Debye) exhibits rapid Li+ hopping between adjacent coordination sites (152.3 ps for acetonitrile and 115.7 ps for FSI-). This electrolyte enables stable cycling of 1.0 Ah 4.5 V graphite (3.13 mAh cm-2)||LiNi0.8Mn0.1Co0.1O2 (2.85 mAh cm-2) pouch cells, delivering 0.87 Ah at −40 °C, surpassing commercial carbonate-based electrolytes, which fail to retain reversible capacity at this temperature. This study establishes fundamental principles for fast ion-transport electrolytes, paving the way for next-generation Li-ion batteries under extreme scenarios.
Similar content being viewed by others
Data availability
All data that support the findings of this study are presented in the manuscript and Supplementary Information, or are available from the corresponding author upon request. Source data are provided with this paper. The atomic coordinates of the optimized geometries, along with related information, can be found in the Supplementary Data. Source data are provided with this paper.
References
Tarascon, J. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 414, 359–367 (2001).
Tian, Y. et al. Promises and challenges of next-generation “beyond Li-ion” batteries for electric vehicles and grid decarbonization. Chem. Rev. 121, 1623–1669 (2021).
Zeng, X. et al. Commercialization of lithium battery technologies for electric vehicles. Adv. Energy Mater. 9, 1900161 (2019).
Zhang, S., Xu, K. & Jow, T. The low temperature performance of Li-ion batteries. J. Power Sources 115, 137–140 (2003).
Liu, Y., Zhu, Y. & Cui, Y. Challenges and opportunities towards fast-charging battery materials. Nat. Energy 4, 540–550 (2019).
Rodrigues, M. et al. A materials perspective on Li-ion batteries at extreme temperatures. Nat. Energy 2, 17108–17121 (2017).
Yu, D. et al. Uncorrelated lithium-ion hopping in a dynamic solvent–anion network. ACS Energy Lett. 8, 1944–1951 (2023).
Chen, X. & Kuroda, D. Molecular motions of acetonitrile molecules in the solvation shell of lithium ions. J. Chem. Phys. 153, 164502 (2020).
Wu, Z. et al. Deciphering and modulating energetics of solvation structure enables aggressive high-voltage chemistry of Li metal batteries. Chem 9, 650–664 (2023).
Ma, B. et al. Molecular-docking electrolytes enable high-voltage lithium battery chemistries. Nat. Chem. 16, 1427–1435 (2024).
Chen, K. et al. Correlating the solvating power of solvents with the strength of ion-dipole interaction in electrolytes of lithium-ion batteries. Angew. Chem. Int. Ed. 62, e202312373 (2023).
Sun, Q. et al. Dipole–dipole interaction induced electrolyte interfacial model to stabilize antimony anode for high-safety lithium-ion batteries. ACS Energy Lett. 7, 3545–3556 (2022).
Hou, S. et al. Solvation sheath reorganization enables divalent metal batteries with fast interfacial charge transfer kinetics. Science 374, 172–178 (2021).
Borodin, O. & Smith, G. D. Li+ transport mechanism in oligo(ethylene oxide)s compared to carbonates. J. Solut. Chem. 36, 803–881 (2007).
Xu, K. Electrolytes and interphases in Li-ion batteries and beyond. Chem. Rev. 114, 11503–11618 (2014).
Valøena, L. & Reimersa, J. Transport properties of LiPF6-based Li-ion battery electrolytes. J. Electrochem. Soc. 152, A882–A891 (2005).
Hayamizu, K. et al. Pulse-gradient spin-echo 1H, 7Li, and 19F NMR diffusion and ionic conductivity measurements of 14 organic electrolytes containing LiN(SO2CF3)2. J. Phys. Chem. B 103, 519–524 (1999).
Yamada, Y. et al. Unusual stability of acetonitrile-based superconcentrated electrolytes for fast-charging lithium-ion batteries. J. Am. Chem. Soc. 136, 5039–5046 (2014).
Fan, X. et al. Highly fluorinated interphases enable high-voltage Li-metal batteries. Chem 4, 174–185 (2018).
Wang, J. et al. Superconcentrated electrolytes for a high-voltage lithium-ion battery. Nat. Commun. 7, 12032 (2016).
Suo, L. et al. Fluorine-donating electrolytes enable highly reversible 5-V-class Li metal batteries. Proc. Natl. Acad. Sci. USA 115, 1156–1161 (2018).
Yang, Y. et al. Synergy of weakly-solvated electrolyte and optimized interphase enables graphite anode charge at low temperature. Angew. Chem. Int. Ed. 61, e202208345 (2022).
Yu, Z. et al. Molecular design for electrolyte solvents enabling energy-dense and long-cycling lithium metal batteries. Nat. Energy 5, 526–533 (2020).
Yu, Z. et al. Rational solvent molecule tuning for high-performance lithium metal battery electrolytes. Nat. Energy 7, 94–106 (2022).
Yao, Y. et al. Regulating interfacial chemistry in lithium-ion batteries by a weakly solvating electrolyte. Angew. Chem. Int. Ed. 60, 4090–4097 (2021).
Gupta, A. & Manthiram, A. Designing advanced lithium-based batteries for low-temperature conditions. Adv. Energy Mater. 10, 2001972 (2020).
Chen, X. & Zhang, Q. Atomic insights into the fundamental interactions in lithium battery electrolytes. Acc. Chem. Res. 53, 1992–2002 (2020).
Kataoka, H. et al. Ionic mobility of cation and anion of lithium gel electrolytes measured by pulsed gradient spin-echo NMR technique under direct electric field. J. Phys. Chem. B 105, 2546–2550 (2001).
Pfeifer, S. et al. Quantification of cation-cation, anion-anion and cation-anion correlations in Li salt/glyme mixtures by combining very-low-frequency impedance spectroscopy with diffusion and electrophoretic NMR. Phys. Chem. Chem. Phys. 23, 628–640 (2021).
Lu, D. et al. Ligand-channel-enabled ultrafast Li-ion conduction. Nature 627, 101–107 (2024).
Ming, J. et al. Molecular-scale interfacial model for predicting electrode performance in rechargeable batteries. ACS Energy Lett. 4, 1584–1593 (2019).
Zhang, N. et al. Critical review on low-temperature Li-ion/metal batteries. Adv. Mater. 34, e2107899 (2022).
Fan, X. & Wang, C. High-voltage liquid electrolytes for Li batteries: progress and perspectives. Chem. Soc. Rev. 50, 10486–10566 (2021).
Jang, J. et al. Miniature Li+ solvation by symmetric molecular design for practical and safe Li-metal batteries. Nat. Energy 10, 502–512 (2025).
Sun, C. et al. 50C fast-charge Li-ion batteries using a graphite anode. Adv. Mater. 34, 2206020 (2022).
Bergstrom, H. K. & McCloskey, B. D. Ion transport in (localized) high concentration electrolytes for Li-based batteries. ACS Energy Lett. 9, 373–380 (2024).
Zheng, J. et al. Highly stable operation of lithium metal batteries enabled by the formation of a transient high-concentration electrolyte layer. Adv. Energy Mater. 6, 1502151 (2016).
Efaw, C. et al. Localized high-concentration electrolytes get more localized through micelle-like structures. Nat. Mater. 22, 1531–1539 (2023).
Li, X. et al. Understanding steric hindrance effect of solvent molecule in localized high-concentration electrolyte for lithium metal batteries. Carbon Neutrality 2, 34 (2023).
Perez Beltran, S. et al. Localized high concentration electrolytes for high voltage lithium–metal batteries: correlation between the electrolyte composition and its reductive/oxidative Stability. Chem. Mater. 32, 5973–5984 (2020).
Watanabe, Y. et al. Does Li-ion transport occur rapidly in localized high-concentration electrolytes? Phys. Chem. Chem. Phys. 25, 3092–3099 (2023).
Zhang, X. et al. Advanced electrolytes for fast-charging high-voltage lithium-ion batteries in wide-temperature range. Adv. Energy Mater. 10, 2000368 (2020).
Ren, X. et al. Localized high-concentration sulfone electrolytes for high-efficiency lithium-metal batteries. Chem 4, 1877–1892 (2018).
Watts, T. J., Smart, M. C. & Manthiram, A. Differentiating the Synergistic interactions between Li+ salts and cyclic to linear carbonate ratios to enable wide-temperature performance of lithium-ion batteries. Adv. Funct. Mater. 36, e11694 (2026).
Tu, H. et al. Regulating non-equilibrium solvation structure in locally concentrated ionic liquid electrolytes for wide-temperature and high-voltage lithium metal batteries. Angew. Chem. Int. Ed. 64, e202412896 (2025).
Smart, M. et al. Lithium-ion electrolytes containing ester cosolvents for improved low temperature performance. J. Electrochem. Soc. 157, A1361 (2010).
Nilsson, V. et al. Critical evaluation of the stability of highly concentrated LiTFSI-acetonitrile electrolytes vs. graphite, lithium metal and LiFePO4 electrodes. J. Power Sources 384, 334–341 (2018).
Yao, Y. X. Unlocking charge transfer limitations for extreme fast charging of Li-ion batteries. Angew. Chem. Int. Ed. 62, e202214828 (2023).
Gao, Y. et al. Low-temperature and high-rate-charging lithium metal batteries enabled by an electrochemically active monolayer-regulated interface. Nat. Energy 5, 534–542 (2020).
Chen, S. et al. High-voltage lithium-metal batteries enabled by localized high-concentration electrolytes. Adv. Mater. 30, e1706102 (2018).
Ren, X. et al. Role of inner solvation sheath within salt-solvent complexes in tailoring electrode/electrolyte interphases for lithium metal batteries. Proc. Natl. Acad. Sci. USA 117, 28603–28613 (2020).
Zhi, H., Xing, L., Zheng, X., Xu, K. & Li, W. Understanding how nitriles stabilize electrolyte/electrode interface at high voltage. J. Phys. Chem. Lett. 8, 6048–6052 (2017).
Zhang, S. et al. Tackling realistic Li+ flux for high-energy lithium metal batteries. Nat. Commun. 13, 5431 (2022).
Sun, A. et al. Dual-halide interphase enabling high-performance lithium metal batteries in wide-temperature range. ACS Energy Lett. 9, 2545–2553 (2024).
Wang, H. et al. Dual LiF/LiCl-rich solid electrolyte interphases with robust and Li+-conductive characteristics for 4.8 V lithium metal batteries. Adv. Energy Mater. 14, 2400067 (2024).
Evans, J., Vincent, C. & Bruce, P. Electrochemical measurement of transference numbers in polymer electrolytes. Polymer 28, 2324–2328 (1987).
Grimme, S. et al. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2011).
Lu, T. & Chen, Q. Independent gradient model based on Hirshfeld partition: a new method for visual study of interactions in chemical systems. J. Comput. Chem. 43, 539 (2022).
Dodda, L. et al. LigParGen web server: an automatic OPLS-AA parameter generator for organic ligands. Nucleic Acids Res. 45, W331–W336 (2017).
Brehm, M. & Kirchner, B. TRAVIS—a free analyzer and visualizer for monte carlo and molecular dynamics trajectories. J. Chem. Inf. Model. 51, 2007–2023 (2011).
Brehm, M., Thomas, M., Gehrke, S. & Kirchner, B. TRAVIS—a free analyzer for trajectories from molecular simulation. J. Chem. Phys. 152, 164105 (2020).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal-amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2024YFB3814300), the Key R&D Program of Zhejiang (2023C01128), National Natural Science Foundation of China (T2525005), Natural Science Foundation of Zhejiang Province (LR23B030002, and LMS25B030002), the Fundamental Research Funds for the Central Universities (226-2024-00075), “Hundred Talents Program” of Zhejiang University, the National Postdoctoral Program for Innovative Talents (BX20240310) and China Postdoctoral Science Foundation (2024M762796).
Author information
Authors and Affiliations
Contributions
M.L., D.L., J.W., and X.F. conceived the idea and designed the experiments. M.L. and D.L. conducted the electrochemical experiments and the material characterizations, with the assistance of S.Z., L. Lv, B.M., H.Z., L.Li., J.Q., L.C., and X.F. S.Y. and Y.W. performed DSC under the guidance of L.F. Z.L. performed the viscosity test. J.W. provided the theoretical calculations. M.L., D.L., J.W., S.Z., R.L., T.D., and X.F. prepared the manuscript, with input from all the co-authors. X.F. supervised all the studies.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Karol Fröhlich and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, M., Lu, D., Wang, J. et al. Solvation sheath reorganization enables fast ion transfer kinetics in lithium-ion battery. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70570-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70570-5