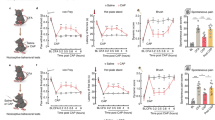
Abstract
Fear and pain are two frequently co-occurring states that mammals need to orchestrate to ensure survival. Nevertheless, how the brain dynamically prioritizes between them remains poorly understood. Here, we demonstrate that innate fear suppresses both acute and chronic pain, whereas pain does not reciprocally modulate fear responses in male mice. Using fiber photometry, virus tracing, and electrophysiological approaches, we show that exposure to a fear-inducing odor activates GABAergic neurons in the anterior piriform cortex (APC), which subsequently attenuates pain-associated hyperactivity in the downstream mediodorsal thalamus (MD). Crucially, inhibiting either APCGABA neurons or the APCGABA-MD circuit enhances pain sensitivity and abolishes fear-induced analgesia. Conversely, activation of APCGABA neurons or the APCGABA-MD circuit induces freezing responses and relieves pain, mimicking fear-induced analgesia. These findings unveil a corticothalamic circuit that bidirectionally regulates pain processing and underlies fear-provoked analgesia, offering potential therapeutic avenues for pain management.
Similar content being viewed by others
Data availability
Source data are provided with this paper. There are no restrictions on data availability in the manuscript. Source data are provided with this paper.
References
Anderson, D. J. Circuit modules linking internal states and social behaviour in flies and mice. Nat. Rev. Neurosci. 17, 692–704 (2016).
Flavell, S. W., Gogolla, N., Lovett-Barron, M. & Zelikowsky, M. The emergence and influence of internal states. Neuron 110, 2545–2570 (2022).
Tinbergen, N. The Study of Instinct (Clarendon Press, 1951).
Fanselow, M. S. Conditioned fear-induced opiate analgesia: a competing motivational state theory of stress analgesia. Ann. N. Y. Acad. Sci. 467, 40–54 (1986).
Alhadeff, A. L. et al. A neural circuit for the suppression of pain by a competing need state. Cell 173, 140–152 e115 (2018).
Farrell, M. J. et al. Unique, common, and interacting cortical correlates of thirst and pain. Proc. Natl. Acad. Sci. USA 103, 2416–2421 (2006).
Hamm, R. J. & Lyeth, B. G. Nociceptive thresholds following food restriction and return to free-feeding. Physiol. Behav. 33, 499–501 (1984).
Hargraves, W. A. & Hentall, I. D. Analgesic effects of dietary caloric restriction in adult mice. Pain 114, 455–461 (2005).
Goldstein, N. et al. A parabrachial hub for need-state control of enduring pain. Nature 647, 689–697 (2025).
LaGraize, S. C., Borzan, J., Rinker, M. M., Kopp, J. L. & Fuchs, P. N. Behavioral evidence for competing motivational drives of nociception and hunger. Neurosci. Lett. 372, 30–34 (2004).
Olango, W. M., Roche, M., Ford, G. K., Harhen, B. & Finn, D. P. The endocannabinoid system in the rat dorsolateral periaqueductal grey mediates fear-conditioned analgesia and controls fear expression in the presence of nociceptive tone. Br. J. Pharmacol. 165, 2549–2560 (2012).
Helmstetter, F. J. & Fanselow, M. S. Effects of naltrexone on learning and performance of conditional fear-induced freezing and opioid analgesia. Physiol. Behav. 39, 501–505 (1987).
Ford, G. K., Kieran, S., Dolan, K., Harhen, B. & Finn, D. P. A role for the ventral hippocampal endocannabinoid system in fear-conditioned analgesia and fear responding in the presence of nociceptive tone in rats. Pain 152, 2495–2504 (2011).
Stegemann, A. et al. Prefrontal engrams of long-term fear memory perpetuate pain perception. Nat. Neurosci. 26, 820–829 (2023).
Bolles, R. C. & Fanselow, M. S. A perceptual-defensive-recuperative model of fear and pain. Behav. Brain Sci. 3, 291–301 (1980).
Isosaka, T. et al. Htr2a-expressing cells in the central amygdala control the hierarchy between innate and learned fear. Cell 163, 1153–1164 (2015).
Sosulski, D. L., Bloom, M. L., Cutforth, T., Axel, R. & Datta, S. R. Distinct representations of olfactory information in different cortical centres. Nature 472, 213–216 (2011).
Wilson, D. A. & Sullivan, R. M. Cortical processing of odor objects. Neuron. 72, 506–519 (2011).
Bekkers, J. M. & Suzuki, N. Neurons and circuits for odor processing in the piriform cortex. Trends Neurosci. 36, 429–438 (2013).
Choi, G. B. et al. Driving opposing behaviors with ensembles of piriform neurons. Cell 146, 1004–1015 (2011).
Terral, G. et al. CB1 receptors in the anterior piriform cortex control odor preference memory. Curr. Biol. 29, 2455–2464 e2455 (2019).
Meissner-Bernard, C., Dembitskaya, Y., Venance, L. & Fleischmann, A. Encoding of odor fear memories in the mouse olfactory cortex. Curr. Biol. 29, 367–380 e364 (2019).
Kondoh, K. et al. A specific area of olfactory cortex involved in stress hormone responses to predator odours. Nature 532, 103–106 (2016).
Matsukawa, M., Yoshikawa, M., Katsuyama, N., Aizawa, S. & Sato, T. The anterior piriform cortex and predator odor responses: modulation by inhibitory circuits. Front. Behav. Neurosci. 16, 896525 (2022).
Wang, L. et al. Cell-type-specific whole-brain direct inputs to the anterior and posterior piriform cortex. Front. Neural Circuits 14, 4 (2020).
Blanchard, R. J. & Blanchard, D. C. Passive and active reactions to fear-eliciting stimuli. J. Comp. Physiol. Psychol. 68, 129–135 (1969).
Takahashi, L. K., Nakashima, B. R., Hong, H. & Watanabe, K. The smell of danger: a behavioral and neural analysis of predator odor-induced fear. Neurosci. Biobehav. Rev. 29, 1157–1167 (2005).
Guenthner, C. J., Miyamichi, K., Yang, H. H., Heller, H. C. & Luo, L. Permanent genetic access to transiently active neurons via TRAP: targeted recombination in active populations. Neuron 78, 773–784 (2013).
Kurahashi, T. & Menini, A. Mechanism of odorant adaptation in the olfactory receptor cell. Nature 385, 725–729 (1997).
Johansen, J. P. & Fields, H. L. Glutamatergic activation of anterior cingulate cortex produces an aversive teaching signal. Nat. Neurosci. 7, 398–403 (2004).
Benarroch, E. E. Involvement of the nucleus accumbens and dopamine system in chronic pain. Neurology. 87, 1720–1726 (2016).
Massaly, N. et al. Pain-induced negative affect is mediated via recruitment of the nucleus accumbens kappa opioid system. Neuron 102, 564–573.e566 (2019).
Ji, Y. W. et al. Plasticity in ventral pallidal cholinergic neuron-derived circuits contributes to comorbid chronic pain-like and depression-like behaviour in male mice. Nat. Commun. 14, 2182 (2023).
Labrakakis, C. The role of the insular cortex in pain. Int. J. Mol. Sci. https://doi.org/10.3390/ijms24065736 (2023).
Wang, X. Y. et al. The thalamic reticular nucleus-lateral habenula circuit regulates depressive-like behaviors in chronic stress and chronic pain. Cell Rep. 42, 113170 (2023).
Gehrlach, D. A. et al. A whole-brain connectivity map of mouse insular cortex. eLife https://doi.org/10.7554/eLife.55585 (2020).
Kozlowska, K., Walker, P., McLean, L. & Carrive, P. Fear and the defense cascade: clinical implications and management. Harv. Rev. Psychiatry 23, 263–287 (2015).
Fox, A. S., Oler, J. A., Tromp do, P. M., Fudge, J. L. & Kalin, N. H. Extending the amygdala in theories of threat processing. Trends Neurosci. 38, 319–329 (2015).
Helmstetter, F. J. The amygdala is essential for the expression of conditional hypoalgesia. Behav. Neurosci. 106, 518–528 (1992).
Corcoran, L., Mattimoe, D., Roche, M. & Finn, D. P. Attenuation of fear-conditioned analgesia in rats by monoacylglycerol lipase inhibition in the anterior cingulate cortex: potential role for CB(2) receptors. Br. J. Pharmacol. 177, 2240–2255 (2020).
Fanselow, M. S. Odors released by stressed rats produce opioid analgesia in unstressed rats. Behav. Neurosci. 99, 589–592 (1985).
Dahlhamer, J. et al. Prevalence of chronic pain and high-impact chronic pain among adults - United States, 2016. MMWR Morb. Mortal. Wkly. Rep. 67, 1001–1006 (2018).
Chao, C. C. et al. Brain imaging signature of neuropathic pain phenotypes in small-fiber neuropathy: altered thalamic connectome and its associations with skin nerve degeneration. Pain 162, 1387–1399 (2021).
Moisset, X. & Bouhassira, D. Brain imaging of neuropathic pain. NeuroImage 37, S80–S88 (2007).
Kuner, R. Central mechanisms of pathological pain. Nat. Med. 16, 1258–1266 (2010).
Wang, X. Y. et al. A glutamatergic DRN-VTA pathway modulates neuropathic pain and comorbid anhedonia-like behavior in mice. Nat. Commun. 14, 5124 (2023).
Lv, S. S. et al. Corticotropin-releasing hormone neurons control trigeminal neuralgia-induced anxiodepression via a hippocampus-to-prefrontal circuit. Sci. Adv. 10, eadj4196 (2024).
Gardner, W. J., Licklider, J. C. & Weisz, A. Z. Suppression of pain by sound. Science 132, 32–33 (1960).
Noseda, R. et al. Migraine photophobia originating in cone-driven retinal pathways. Brain 139, 1971–1986 (2016).
Peana, A. T. et al. (-)-Linalool produces antinociception in two experimental models of pain. Eur. J. Pharmacol. 460, 37–41 (2003).
Tang, Y. L. et al. Green light analgesia in mice is mediated by visual activation of enkephalinergic neurons in the ventrolateral geniculate nucleus. Sci. Transl. Med. 14, eabq6474 (2022).
Cao, P. et al. Green light induces antinociception via visual-somatosensory circuits. Cell Rep. 42, 112290 (2023).
Zhou, W. et al. Sound induces analgesia through corticothalamic circuits. Science 377, 198–204 (2022).
Miura, K., Mainen, Z. F. & Uchida, N. Odor representations in olfactory cortex: distributed rate coding and decorrelated population activity. Neuron 74, 1087–1098 (2012).
Poo, C. & Isaacson, J. S. Odor representations in olfactory cortex: “sparse” coding, global inhibition, and oscillations. Neuron 62, 850–861 (2009).
Iurilli, G. & Datta, S. R. Population coding in an innately relevant olfactory area. Neuron. 93, 1180–1197.e1187 (2017).
Poo, C., Agarwal, G., Bonacchi, N. & Mainen, Z. F. Spatial maps in piriform cortex during olfactory navigation. Nature 601, 595–599 (2022).
Courtiol, E. & Wilson, D. A. Thalamic olfaction: characterizing odor processing in the mediodorsal thalamus of the rat. J. Neurophysiol. 111, 1274–1285 (2014).
Price, J. L. & Slotnick, B. M. Dual olfactory representation in the rat thalamus: an anatomical and electrophysiological study. J. Comp. Neurol. 215, 63–77 (1983).
Kuroda, M., Lopez-Mascaraque, L. & Price, J. L. Neuronal and synaptic composition of the mediodorsal thalamic nucleus in the rat: a light and electron microscopic Golgi study. J. Comp. Neurol. 326, 61–81 (1992).
Pelzer, P., Horstmann, H. & Kuner, T. Ultrastructural and functional properties of a giant synapse driving the piriform cortex to mediodorsal thalamus projection. Front. Synaptic Neurosci. 9, 3 (2017).
Mazo, C. et al. Long-range GABAergic projections contribute to cortical feedback control of sensory processing. Nat. Commun. 13, 6879 (2022).
Bertero, A., Feyen, P. L. C., Zurita, H. & Apicella, A. J. A non-canonical cortico-amygdala inhibitory loop. J. Neurosci. 39, 8424–8438 (2019).
Dautan, D. et al. Cortico-cortical transfer of socially derived information gates emotion recognition. Nat. Neurosci. 27, 1318–1332 (2024).
Meda, K. S. et al. Microcircuit mechanisms through which mediodorsal thalamic input to anterior cingulate cortex exacerbates pain-related aversion. Neuron. 102, 944–959 e943 (2019).
Whitt, J. L., Masri, R., Pulimood, N. S. & Keller, A. Pathological activity in mediodorsal thalamus of rats with spinal cord injury pain. J. Neurosci. 33, 3915–3926 (2013).
Huang, T. et al. Identifying the pathways required for coping behaviours associated with sustained pain. Nature 565, 86–90 (2019).
You, H. J., Lei, J. & Pertovaara, A. Thalamus: the ‘promoter’ of endogenous modulation of pain and potential therapeutic target in pathological pain. Neurosci. Biobehav. Rev. 139, 104745 (2022).
Lebow, M. A. & Chen, A. Overshadowed by the amygdala: the bed nucleus of the stria terminalis emerges as key to psychiatric disorders. Mol. Psychiatry 21, 450–463 (2016).
Hao, S. et al. The lateral hypothalamic and BNST GABAergic projections to the anterior ventrolateral periaqueductal gray regulate feeding. Cell Rep. 28, 616–624.e615 (2019).
Craig, A. D. Interoception: the sense of the physiological condition of the body. Curr. Opin. Neurobiol. 13, 500–505 (2003).
Adolphs, R. The biology of fear. Curr. Biol. 23, R79–R93 (2013).
Large, A. M., Vogler, N. W., Mielo, S. & Oswald, A. M. Balanced feedforward inhibition and dominant recurrent inhibition in olfactory cortex. Proc. Natl. Acad. Sci. USA 113, 2276–2281 (2016).
Suzuki, N. & Bekkers, J. M. Distinctive classes of GABAergic interneurons provide layer-specific phasic inhibition in the anterior piriform cortex. Cereb. Cortex 20, 2971–2984 (2010).
Chaplan, S. R., Bach, F. W., Pogrel, J. W., Chung, J. M. & Yaksh, T. L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 53, 55–63 (1994).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (grants 32271048, 32571336, 32070999 to Y.Z., grant 82471311 to W.H., and grant 823B2022 to X.Y.W), the Leading Medicine and Advanced Technologies of IHM (grant 2025IHM01100 to Y.Z.), and Anhui Provincial Natural Science Foundation (grant 2008085J16 to Y.Z.).
Author information
Authors and Affiliations
Contributions
W.B.J., X.Y.W., and X.X.X. designed the studies and conducted most of experiments and data analysis. L.T., Y.X.L, and X.L.L. were involved in the revision. X.Q.L. managed the mouse colonies used in this study. Y.Z., W.B.J., and X.Y.W. wrote the first draft. W.H. and Y.Z. were involved in the overall design of the project and editing of the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Mathieu Wolff, Nanci Winke, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jia, WB., Wang, XY., Xia, XX. et al. A corticothalamic circuit modulates pain sensitivity and mediates innate fear-induced analgesia in male mice. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70580-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70580-3