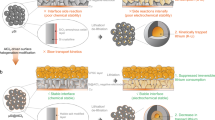
Abstract
O3-phase layered oxides are among the mainstream positive electrode active materials for advanced batteries due to a stable topological lattice framework and potential for tunability. However, surface residual alkali due to sensitivity hinders their large-scale application. Water washing, an industrial surface residual alkali removal method in lithium-based positive electrode materials, brings about severe issues in sodium-based materials, such as lattice collapse and extensive active alkali metal ion leaching. Here, we propose an interaction mechanism between host solid-phase positive electrode materials and guest liquid-phase solvents, which elucidates the dependent structural degradation on the molecular configuration of dispersed solvents and the alkali metal–oxygen bond covalency during dealkalization. For H2O (H3O+), self-propagating molecule intercalation into Na slabs and subsequent protonation induce the leakage of Na ions, leading to lattice destabilization. In contrast, the efficient dealkalizing agent—ethylene glycol—prevents further structural degradation due to the constraint of size effect. Based on the time-dependent deterioration of the extended scope of positive electrode materials, an adaptable analytical framework is established for stability assessment against the liquid phase. Our work provides fundamental theoretical guidance for liquid-phase engineering of O3-phase layered oxides.
Similar content being viewed by others
Data availability
The authors declare that the main data supporting the findings of this study are available within the paper and its Supplementary information. Source data are provided with this paper.
References
Li, W., Erickson, E. M. & Manthiram, A. High-nickel layered oxide cathodes for lithium-based automotive batteries. Nat. Energy 5, 26–34 (2020).
Zuo, W. et al. Engineering Na+-layer spacings to stabilize Mn-based layered cathodes for sodium-ion batteries. Nat. Commun. 12, 4903 (2021).
Yang, Y. et al. Decoupling the air sensitivity of Na-layered oxides. Science 385, 744–752 (2024).
Zuo, W. et al. The stability of P2-layered sodium transition metal oxides in ambient atmospheres. Nat. Commun. 11, 3544 (2020).
Seong, W. M., Kim, Y. & Manthiram, A. Impact of residual lithium on the adoption of high-nickel layered oxide cathodes for lithium-ion batteries. Chem. Mater. 32, 9479–9489 (2020).
Yang, H., Zhang, Q., Chen, M., Yang, Y. & Zhao, J. Unveiling the origin of air stability in polyanion and layered-oxide cathode materials for sodium-ion batteries and their practical application considerations. Adv. Funct. Mater. 34, 2308257 (2023).
Wang, Q. et al. Unlocking anionic redox activity in O3-type sodium 3d layered oxides via Li substitution. Nat. Mater. 20, 353–361 (2021).
Zhao, C. et al. Rational design of layered oxide materials for sodium-ion batteries. Science 370, 708–711 (2020).
Wang, H. et al. In situ plastic-crystal-coated cathode toward high-performance Na-ion batteries. ACS Energy Lett. 8, 1434–1444 (2023).
Zhang, W. et al. Air instability of Ni-rich layered oxides–A roadblock to large scale application. Adv. Energy Mater. 13, 2202993 (2022).
You, L., Chu, B., Li, G., Huang, T. & Yu, A. H3BO3 washed LiNi0.8Co0.1Mn0.1O2 with enhanced electrochemical performance and storage characteristics. J. Power Sources 482, 228940 (2021).
Park, K. et al. Re-construction layer effect of LiNi0.8Co0.15Mn0.05O2 with solvent evaporation process. Sci. Rep. 7, 44557 (2017).
Hamam, I., Zhang, N., Liu, A., Johnson, M. B. & Dahn, J. R. Study of the reactions between Ni-rich positive electrode materials and aqueous solutions and their relation to the failure of Li-ion cells. J. Electrochem. Soc. 167, 130521 (2020).
Hu, B.-R., Yuan, Y.-Y., Wang, Y.-C. & Xiong, X.-H. Simultaneously enhanced electrochemical performance and air stability of Ni-rich cathode with a modified washing process. Rare Met 43, 87–97 (2023).
Li, Y. et al. Enhanced cyclability and reversibility of nickel-rich cathode for lithium-ion batteries via LiH2PO4 assisted saturated Li2CO3 washing. Appl. Surf. Sci. 593, 153409 (2022).
Lee, W. et al. Destabilization of the surface structure of Ni-rich layered materials by water-washing process. Energy Storage Mater. 44, 441–451 (2022).
Pritzl, D. et al. Washing of nickel-rich cathode materials for lithium-ion batteries: Towards a mechanistic understanding. J. Electrochem. Soc. 166, A4056–A4066 (2019).
Zhou, Y., Hu, Z., Huang, Y., Wu, Y. & Hong, Z. Effect of solution wash on the electrochemical performance of LiNi0.8Co0.1Mn0.1O2 cathode materials. J. Alloy. Compd. 888, 161584 (2021).
Zheng, X. et al. Investigation and improvement on the electrochemical performance and storage characteristics of LiNiO2-based materials for lithium ion battery. Electrochim. Acta 191, 832–840 (2016).
Xu, S. et al. A mild surface washing method using protonated polyaniline for Ni-rich LiNi0.8Co0.1Mn0.1O2 material of lithium ion batteries. Electrochim. Acta 248, 534–540 (2017).
You, Y., Song, B., Jarvis, K., Huq, A. & Manthiram, A. Insights into the improved chemical stability against water of LiF-incorporated layered oxide cathodes for sodium-ion batteries. ACS Mater. Lett. 1, 89–95 (2019).
Wang, P. F., You, Y., Yin, Y. X. & Guo, Y. G. Layered oxide cathodes for sodium-ion batteries: phase transition, air stability, and performance. Adv. Energy Mater. 8, 1701912 (2017).
You, Y., Dolocan, A., Li, W. & Manthiram, A. Understanding the air-exposure degradation chemistry at a nanoscale of layered oxide cathodes for sodium-ion batteries. Nano Lett. 19, 182–188 (2018).
Xu, C. et al. Origin of air-stability for transition metal oxide cathodes in sodium-ion batteries. ACS Appl. Mater. Interfaces 14, 5338–5345 (2022).
Yao, H.-R. et al. Designing air-stable O3-type cathode materials by combined structure modulation for Na-ion batteries. J. Am. Chem. Soc. 139, 8440–8443 (2017).
Huang, J. et al. Designing ultrastable P2/O3-type layered oxides for sodium ion batteries by regulating Na distribution and oxygen redox chemistry. J. Energy Chem. 94, 466–476 (2024).
Zhao, Y. et al. Structure evolution of layered transition metal oxide cathode materials for Na-ion batteries: Issues, mechanism and strategies. Mater. Today 62, 271–295 (2023).
Chen, Z. et al. The high-temperature and high-humidity storage behaviors and electrochemical degradation mechanism of LiNi0.6Co0.2Mn0.2O2 cathode material for lithium ion batteries. J. Power Sources 363, 168–176 (2017).
He, Y. et al. Revisiting the electrode manufacturing: A look into electrode rheology and active material microenvironment. J. Energy Chem. 72, 41–55 (2022).
Reynolds, C. D., Hare, S. D., Slater, P. R., Simmons, M. J. H. & Kendrick, E. Rheology and structure of lithium-ion battery electrode slurries. Energy Technol. 10, 2200545 (2022).
Zhang, T. et al. Converting residual alkali into sodium compensation additive for high-energy Na-ion batteries. ACS Energy Lett. 8, 4753–4761 (2023).
Yang, T. et al. Ultrahigh-nickel layered cathode with cycling stability for sustainable lithium-ion batteries. Nat. Sustain. 7, 1204–1214 (2024).
Sun, Y.-K. et al. Nanostructured high-energy cathode materials for advanced lithium batteries. Nat. Mater. 11, 942–947 (2012).
Kumar, B. S., Pradeep, A., Dutta, A. & Mukhopadhyay, A. Water-stable O3-type layered Na transition metal oxides enabling environment friendly ‘aqueous processing’ of electrodes with long-term electrochemical stability. J. Mater. Chem. A 8, 18064–18078 (2020).
Yuan, X. G. et al. A universal strategy toward air-stable and high-rate O3 layered oxide cathodes for Na-ion batteries. Adv. Funct. Mater. 32, 2111466 (2022).
Jia, S., Kumakura, S. & McCalla, E. Unravelling air/moisture stability of cathode materials in sodium ion batteries: characterization, rational design, and perspectives. Energy Environ. Sci. 17, 4343–4389 (2024).
Mortemard de Boisse, B. et al. O3–NaxMn1/3Fe2/3O2 as a positive electrode material for Na-ion batteries: structural evolutions and redox mechanisms upon Na+ (de)intercalation. J. Mater. Chem. A 3, 10976–10989 (2015).
Tang, Y. et al. Sustainable layered cathode with suppressed phase transition for long-life sodium-ion batteries. Nat. Sustain. 7, 348–359 (2024).
Sicklinger, J., Metzger, M., Beyer, H., Pritzl, D. & Gasteiger, H. A. Ambient storage derived surface contamination of NCM811 and NCM111: performance implications and mitigation strategies. J. Electrochem. Soc. 166, A2322–A2335 (2019).
Hartmann, L., Pritzl, D., Beyer, H. & Gasteiger, H. A. Evidence for Li+/H+ exchange during ambient storage of Ni-rich cathode active materials. J. Electrochem. Soc. 168, 070507 (2021).
Sathiya, M. et al. A chemical approach to raise cell voltage and suppress phase transition in O3 sodium layered oxide electrodes. Adv. Energy Mater. 8, 1702599 (2018).
Komaba, S. et al. Study on the reversible electrode reaction of Na1-xNi0.5Mn0.5O2 for a rechargeable sodium-ion battery. Inorg. Chem. 51, 6211–6220 (2012).
Zhang, R., Yang, S., Li, H., Zhai, T. & Li, H. Air sensitivity of electrode materials in Li/Na ion batteries: Issues and strategies. InfoMat 4, e12305 (2022).
Yao, H.-R., Zheng, L., Xin, S. & Guo, Y.-G. Air-stability of sodium-based layered-oxide cathode materials. Sci. China Chem. 65, 1076–1087 (2022).
Huang, J. et al. Thermodynamically spontaneously intercalated H3O+ enables LiMn2O4 with enhanced proton tolerance in aqueous batteries. Nat. Commun. 15, 6666 (2024).
Xu, X.-Q. et al. Origins of high air sensitivity and treatment strategies in O3-type NaMn1/3 Fe1/3Ni1/3O2. J. Am. Chem. Soc. 146, 22374–22386 (2024).
Zou, L. et al. Unlocking the passivation nature of the cathode–air interfacial reactions in lithium ion batteries. Nat. Commun. 11, 3204 (2020).
Sengupta, A. et al. Unleashing the impact of Nb-doped, single crystal, cobalt-free P2-type Na0.67Ni0.33Mn0.67O2 on elevating the cycle life of sodium-ion batteries. Energy Storage Mater. 69, 103435 (2024).
Shin, J., Choi, D. S., Lee, H. J., Jung, Y. & Choi, J. W. Hydrated intercalation for high-performance aqueous zinc ion batteries. Adv. Energy Mater. 9, 1900083 (2019).
Peng, J. et al. The effect of hydration number on the interfacial transport of sodium ions. Nature 557, 701–705 (2018).
Wang, J. L. et al. Design of Cu-substituted O3-type NaFe0.5Mn0.5O2 cathode materials for sodium-ion batteries. Chem. – A Eur. J. 29, e202301014 (2023).
Bresser, D., Buchholz, D., Moretti, A., Varzi, A. & Passerini, S. Alternative binders for sustainable electrochemical energy storage – the transition to aqueous electrode processing and bio-derived polymers. Energy Environ. Sci. 11, 3096–3127 (2018).
Song, M. et al. Interfacial engineering of P2-type Ni/Mn-based layered oxides by a facile water-washing method for superior sodium–ion batteries. ACS Appl. Mater. Interfaces 16, 16120–16131 (2024).
Xiao, P. et al. Insights into the solvation chemistry in liquid electrolytes for lithium-based rechargeable batteries. Chem. Soc. Rev. 52, 5255–5316 (2023).
Wang, J. et al. Green recycling of spent Li-ion battery cathodes via deep-eutectic solvents. Energy Environ. Sci. 17, 867–884 (2024).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Blöchl, P. E., Jepsen, O. & Andersen, O. K. Improved tetrahedron method for Brillouin-zone integrations. Phys. Rev. B 49, 16223–16233 (1994).
Perdew, J. P. et al. Erratum: Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B 48, 4978–4978 (1993).
Dronskowski, R. & Bloechl, P. E. Crystal orbital Hamilton populations (COHP): energy-resolved visualization of chemical bonding in solids based on density-functional calculations. J. Phys. Chem. 97, 8617–8624 (1993).
Acknowledgements
C.S. acknowledges support from the National Natural Science Foundation of China (No. 52374311). K.Y.X. acknowledges support from the NSAF (No. U2330205), National Key R&D Program of China (2023YFE0203000), and the Youth Innovation Team of Shaanxi Universities. C.S. acknowledges support from the National Natural Science Foundation of Shaanxi (2025SYS-SYSZD-035), the Fund of the State Key Laboratory of Solidification Processing in NPU (2025-TS-10), and the Fundamental Research Funds for the Central Universities (D5000250277). Jie Chen was acknowledged for their help on the neutron powder diffraction experiments, which were performed at the general-purpose powder diffractometer (GPPD) of the China Spallation Neutron Source (CSNS), Dongguan, China.
Author information
Authors and Affiliations
Contributions
K.Y.X. and C.S. conceived the project. K.Y.X. supervised the project. W.J.Z., X.K.X., and C.Y.L. synthesized the samples. W.J.Z. and C.S. designed and carried out the experiments, analyzed the results, and wrote the paper. J.F.Z. and A.M.S. provided guidance for the DEMS and ICP-OES experiments analysis. X.Z., M.J.H., and C.H.Y. guided scale-up electrode manufacturing and pouch cell tests. N.L., S.P.Z, and L.D. provided recommendations regarding figure composition and optimization for visualization. Y.R.Y. supported the design and analysis of results for theoretical calculations. All authors discussed the results and co-edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Gui-Liang Xu and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, W., Zhu, J., Song, A. et al. Deciphering the liquid-solid interactions in dealkalization of O3 layered oxides. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70581-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70581-2