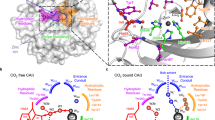
Abstract
Stomata of plant leaves open to enable CO2 entry for photosynthesis and close when CO2 in the leaf is elevated. CO2 is thought to promote stomatal closure in part by activating the SLAC1 anion channel at the guard cell plasma membrane. Carbonic anhydrases (CAs) contribute to this activation, but their contribution as distinct from CO2-H2CO3 catalysis remains controversial. Here we show that the β-carbonic anhydrase CA4 binds selectively with the guard-cell anion channel SLAC1 to enhance channel current. The interaction is CO2-dependent, but binding is mediated by amino acids distal from the CO2-binding site of CA4 and is separable from carbonic-anhydrase activity. CA4 mutants impaired in channel binding eliminate the CO2-sensitivity of SLAC1 in vivo and slow stomatal kinetics with a commensurate loss in water use efficiency. The findings demonstrate that CA4 contributes directly to the CO2-response mechanics regulating SLAC1 at near-ambient CO2 in guard cells and to stomatal kinetics in the plant.
Similar content being viewed by others
Data availability
Data generated and analysed during this study are included in the article and its supplementary files. Unique biological materials are available on reasonable request to the corresponding author. Source data are provided with this paper.
Code availability
The OnGuard3 platform and the model parameter sets are freely available to academic users and may be downloaded from www.plantscienceglasgow.org.
References
Hetherington, A. M. & Woodward, F. I. The role of stomata in sensing and driving environmental change. Nature 424, 901–908 (2003).
Lawson, T. & Blatt, M. R. Stomatal Size, Speed, and Responsiveness Impact on Photosynthesis and Water Use Efficiency. Plant Physiol. 164, 1556–1570 (2014).
Beljaars, A. C. M., Viterbo, P., Miller, M. J. & Betts, A. K. The anomalous rainfall over the United States during July 1993: Sensitivity to land surface parameterization and soil moisture. Mon. Weather Rev. 124, 362–383 (1996).
Berry, J. A., Beerling, D. J. & Franks, P. J. Stomata: key players in the earth system, past and present. Curr. Opin. Plant Biol. 13, 233–240 (2010).
Unesco. Water for a Sustainable World -- UN World Water Development Report. 1 (UNESCO, 2015).
Matthews, J. S. A. & Lawson, T. CLIMATE CHANGE AND STOMATAL PHYSIOLOGY. Annu. Plant Rev. Online 2, 713–751 (2019).
Blatt, M. R., Jezek, M., Lew, V. L. & Hills, A. What can mechanistic models tell us about guard cells, photosynthesis, and water use efficiency? Trends Plant Sci. 27, 166–179 (2022).
Jezek, M. & Blatt, M. R. The Membrane Transport System of the Guard Cell and Its Integration for Stomatal Dynamics. Plant Physiol. 174, 487–519 (2017).
Assmann, S. M. & Jegla, T. Guard cell sensory systems: recent insights on stomatal responses to light, abscisic acid, and CO2. Curr. Opin. Plant Biol. 33, 157–167 (2016).
Nakamura, R. L. et al. Expression of an Arabidopsis potassium channel gene in guard cells. Plant Physiol. 109, 371–374 (1995).
Pilot, G., Gaymard, F., Mouline, K., Cherel, I. & Sentenac, H. Regulated expression of Arabidopsis Shaker K + channel genes involved in K + uptake and distribution in the plant. Plant Mol. Biol. 51, 773–787 (2003).
Lebaudy, A. et al. Plant adaptation to fluctuating environment and biomass production are strongly dependent on guard cell potassium channels. Proc. Natl. Acad. Sci. USA. 105, 5271–5276 (2008).
Clint, G. M. & Blatt, M. R. Mechanisms of fusicoccin action: evidence for concerted modulations of secondary K+ transport in a higher-plant cell. Planta 178, 495–508 (1989).
Blatt, M. R. & Clint, G. M. Mechanisms of fusicoccin action kinetic modification and inactivation of potassium channels in guard cells. Planta 178, 509–523 (1989).
Very, A.-A. et al. Molecular biology of K+ transport across the plant cell membrane: What do we learn from comparison between plant species? J. Plant Physiol. 171, 748–769 (2014).
Vahisalu, T. et al. SLAC1 is required for plant guard cell S-type anion channel function in stomatal signalling. Nature 452, 487–491 (2008).
Negi, J. et al. CO2 regulator SLAC1 and its homologues are essential for anion homeostasis in plant cells. Nature 452, 483–486 (2008).
Wang, Y. et al. Systems dynamic modelling of a guard cell Cl- channel mutant uncovers an emergent homeostatic network regulating stomatal transpiration. Plant Physiol. 160, 1956–1972 (2012).
Jezek, M. et al. Guard cell endomembrane Ca2+-ATPases underpin a ‘carbon memory’ of photosynthetic assimilation that impacts on water use efficiency. Nat. Plants 7, 1301–1307 (2021).
Geiger, D. et al. Guard cell anion channel SLAC1 is regulated by CDPK protein kinases with distinct Ca2+ affinities. Proc. Natl. Acad. Sci. USA. 107, 8023–8028 (2010).
Chen, Z. H., Hills, A., Lim, C. K. & Blatt, M. R. Dynamic regulation of guard cell anion channels by cytosolic free Ca2+ concentration and protein phosphorylation. Plant J. 61, 816–825 (2010).
Geiger, D. et al. Activity of guard cell anion channel SLAC1 is controlled by drought-stress signaling kinase-phosphatase pair. Proc. Natl. Acad. Sci. USA. 106, 21425–21430 (2009).
Maierhofer, T. et al. Site- and kinase-specific phosphorylation-mediated activation of SLAC1, a guard cell anion channel stimulated by abscisic acid. Sci. Signal. 7, https://doi.org/10.1126/scisignal.2005703 (2014).
Li, Y. et al. Structure of the Arabidopsis guard cell anion channel SLAC1 suggests activation mechanism by phosphorylation. Nat. Commun. 13, 2511–2511 (2022).
Deng, Y. N. et al. Structure and activity of SLAC1 channels for stomatal signaling in leaves. Proc. Natl Acad. Sci. USA. 118, https://doi.org/10.1073/pnas.2015151118 (2021).
Takahashi, Y. et al. Stomatal CO2/bicarbonate sensor consists of two interacting protein kinases, Raf-like HT1 and non-kinase-activity activity requiring MPK12/MPK4. Sci. Adv. 8, https://doi.org/10.1126/sciadv.abq6161 (2022).
Horak, H. et al. A Dominant Mutation in the HT1 Kinase Uncovers Roles of MAP Kinases and GHR1 in CO2-Induced Stomatal Closure. Plant Cell 28, 2493–2509 (2016).
Wang, C. et al. Reconstitution of CO2 Regulation of SLAC1 Anion Channel and Function of CO2-Permeable PIP2;1 Aquaporin as CARBONIC ANHYDRASE4 Interactor. Plant Cell 28, 568–582 (2016).
Hu, H. H. et al. Carbonic anhydrases are upstream regulators of CO2 -controlled stomatal movements in guard cells. Nat. Cell Biol. 12, 87–90 (2010).
Fabre, N., Reiter, I. M., Becuwe-Linka, N., Genty, B. & Rumeau, D. Characterization and expression analysis of genes encoding a and b carbonic anhydrases in Arabidopsis. Plant Cell Environ. 30, 617–629 (2007).
Honsbein, A. et al. A Tripartite SNARE-K+ Channel Complex Mediates in Channel-Dependent K+ Nutrition in Arabidopsis. Plant Cell 21, 2859–2877 (2009).
Grefen, C., Obrdlik, P. & Harter, K. The determination of protein-protein interactions byt the mating-based split-ubiquitin system (mbSUS). Methods Mol. Biol. 479, 1–17 (2009).
Grefen, C. et al. A vesicle-trafficking protein commandeers Kv channel voltage sensors for voltage-dependent secretion. Nat. Plants 1, 15108–15119 (2015).
Blatt, M. R. Potassium channel currents in intact stomatal guard cells: rapid enhancement by abscisic acid. Planta 180, 445–455 (1990).
DiMario, R. J. et al. The Cytoplasmic Carbonic Anhydrases βCA2 and βCA4 Are Required for Optimal Plant Growth at Low CO2. Plant Physiol. 171, 280–293 (2016).
Nett, R. S. et al. Plant carbonic anhydrase-like enzymes in neuroactive alkaloid biosynthesis. Nature 624, 182-+ (2023).
Langella, E. et al. α-CAs from Photosynthetic Organisms. Int. J. Mol. Sci. 23, https://doi.org/10.3390/ijms231912045 (2022).
Burén, S. et al. Importance of Post-Translational Modifications for Functionality of a Chloroplast-Localized Carbonic Anhydrase (CAH1) in Arabidopsis thaliana. Plos One 6, https://doi.org/10.1371/journal.pone.0021021 (2011).
Rowlett, R. S. in Carbonic Anhydrase: Mechanism, Regulation, Links to Disease, and Industrial Applications Vol. 75 Subcellular Biochemistry (eds Frost, S. C. & McKenna, R.) 53–76 (2014).
Kimber, M. S. & Pai, E. F. The active site architecture of Pisum sativum β-carbonic anhydrase is a mirror image of that of α-carbonic anhydrases. Embo J. 19, 1407–1418 (2000).
Rowlett, R. S. Structure and catalytic mechanism of the β-carbonic anhydrases. Biochimica Et. Biophysica Acta-Proteins Proteom. 1804, 362–373 (2010).
Rai, A. K. et al. A Rapid Method for Detecting Normal or Modified Plant and Algal Carbonic Anhydrase Activity Using Saccharomyces cerevisiae. Plants-Basel 11, https://doi.org/10.3390/plants11141882 (2022).
Baena, G. et al. Arabidopsis SNARE SYP132 impacts on PIP2;1 trafficking and function in salinity stress. Plant J. 118, 1036–1053 (2024).
Hecker, A. et al. Binary 2in1 vectors improve in planta (co-) localisation and dynamic protein interaction studies. Plant Physiol. 168, 776–787 (2015).
Chen, Y. -h et al. Homologue structure of the SLAC1 anion channel for closing stomata in leaves. Nature 467, 1074–U1157 (2010).
Brearley, J., Venis, M. A. & Blatt, M. R. The effect of elevated CO2 concentrations on K+ and anion channels of Vicia faba L. guard cells. Planta 203, 145–154 (1997).
Zhang, J. et al. Identification of SLAC1 anion channel residues required for CO2/bicarbonate sensing and regulation of stomatal movements. Proc. Natl. Acad. Sci. USA. 115, 11129–11137 (2018).
Wang, Y., Chen, Z. H., Zhang, B., Hills, A. & Blatt, M. R. PYR/PYL/RCAR Abscisic Acid Receptors Regulate K+ and Cl- Channels through Reactive Oxygen Species-Mediated Activation of Ca2+ Channels at the Plasma Membrane of Intact Arabidopsis Guard Cells. Plant Physiol. 163, 566–577 (2013).
Horaruang, W. et al. Engineering a K+ channel ‘sensory antenna’ enhances stomatal kinetics, water use efficiency and photosynthesis. Nat. Plants 8, 1262–1274 (2022).
Blatt, M. R. Electrical characteristics of stomatal guard cells: the ionic basis of the membrane potential and the consequence of potassium chloride leakage from microelectrodes. Planta 170, 272–287 (1987).
Wang, Y. et al. Unexpected Connections between Humidity and Ion Transport Discovered using a Model to Bridge Guard Cell-to-Leaf Scales. Plant Cell 29, 2921–2139 (2017).
Papanatsiou, M. et al. Optogenetic manipulation of stomatal kinetics improves carbon assimilation and water use efficiency. Science 363, 1456–1459 (2019).
Cubero-Font, P. et al. Silent S-Type Anion Channel Subunit SLAH1 Gates SLAH3 Open for Chloride Root-to-Shoot Translocation. Curr. Biol. 26, 2213–2220 (2016).
Birnbaum, K. et al. A gene expression map of the Arabidopsis root. Science 302, 1956–1960 (2003).
Kellermeier, F., Chardon, F. & Amtmann, A. Natural Variation of Arabidopsis Root Architecture Reveals Complementing Adaptive Strategies to Potassium Starvation. Plant Physiol. 161, 1421–1432 (2013).
Hedrich, R. & Geiger, D. Biology of SLAC1-type anion channels - from nutrient uptake to stomatal closure. N. Phytologist 216, 46–61 (2017).
Xue, S. W. et al. Central functions of bicarbonate in S-type anion channel activation and OST1 protein kinase in CO2 signal transduction in guard cells. EMBO Journal 30, 1645–1658; Published online 18 March 2011 Subject Categories: membranes & transport; plant biology (2011).
Christie, J. M. Phototropin blue-light receptors. Annu. Rev. Plant Biol. 58, 21–45 (2007).
Blatt, M. R. A charged existence: A century of transmembrane ion transport in plants. Plant Physiol. 195, 79–110 (2024).
Klejchova, M., Silva-Alvim, F. A. L., Blatt, M. R. & Alvim, J. C. Membrane voltage as a dynamic platform for spatiotemporal signaling, physiological, and developmental regulation. Plant Physiol. 185, 1523–1541 (2021).
Melotto, M., Zhang, L., Oblessuc, P. R. & He, S. Y. Stomatal Defense a Decade Later. Plant Physiol. 174, 561–571 (2017).
Grabov, A. & Blatt, M. R. Parallel control of the inward-rectifier K+ channel by cytosolic-free Ca2+ and pH in Vicia guard cells. Planta 201, 84–95 (1997).
Min, M. K. et al. Two Clade A Phosphatase 2Cs Expressed in Guard Cells Physically Interact With Abscisic Acid Signaling Components to Induce Stomatal Closure in Rice. Rice 12, https://doi.org/10.1186/s12284-019-0297-7 (2019).
Lefoulon, C., Boxall, S. F., Hartwell, J. & Blatt, M. R. Crassulacean acid metabolism guard cell anion channel activity follows transcript abundance and is suppressed by apoplastic malate. N. Phytologist 227, 1847–1857 (2020).
Kusumi, K., Hirotsuka, S., Kumamaru, T. & Iba, K. Increased leaf photosynthesis caused by elevated stomatal conductance in a rice mutant deficient in SLAC1, a guard cell anion channel protein. J. Exp. Bot. 63, 5635–5644 (2012).
Alvim, F. S. L., Alvim, J., Harvey, A. L. & Blatt, M. R. Speedy stomata of a C4 plant correlate with enhanced K+ channel gating. Plant Cell Environ. 47, 817–831 (2024).
Taylor, S. H. et al. Physiological advantages of C-4 grasses in the field: a comparative experiment demonstrating the importance of drought. Glob. Change Biol. 20, 1992–2003 (2014).
Osborne, C. P. & Sack, L. Evolution of C-4 plants: a new hypothesis for an interaction of CO2 and water relations mediated by plant hydraulics. Philos. Trans. R. Soc. B-Biol. Sci. 367, 583–600 (2012).
Alvim, F., Alvim, J. C., Hibberd, J. M., Harvey, A. R. & Blatt, M. R. A C4 plant K+ channel accelerates stomata to enhance C3 photosynthesis and water use efficiency. Plant Physiol. 197, https://doi.org/10.1093/plphys/kiaf039 (2025).
Doheny-Adams, T., Hunt, L., Franks, P. J., Beerling, D. J. & Gray, J. E. Genetic manipulation of stomatal density influences stomatal size, plant growth and tolerance to restricted water supply across a growth carbon dioxide gradient. Philos. Trans. R. Soc. B-Biol. Sci. 367, 547–555 (2012).
Masle, J., Gilmore, S. R. & Farquhar, G. D. The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature 436, 866–870 (2005).
Wang, Y., Hills, A. & Blatt, M. R. Systems analysis of guard cell membrane transport for enhanced stomatal dynamics and water use efficiency. Plant Physiol. 164, 1593–1599 (2014).
Wang, Y. et al. Overexpression of plasma membrane H+ -ATPase in guard cells promotes light-induced stomatal opening and enhances plant growth. Proc. Nat. Acad. Sci. USA. 111, 533–538 (2014).
Karnik, A., Karnik, R. & Grefen, C. SDM-Assist software to design site-directed mutagenesis primers introducing “silent” restriction sites. Bmc Bioinform. 14 (2013).
Grefen, C. & Blatt, M. R. A 2in1 cloning system enables ratiometric bimolecular fluorescence complementation (rBiFC). Biotechniques 53, 311–314 (2012).
Karimi, M., De Meyer, B. & Hilson, P. Modular cloning in plant cells. Trends Plant Sci. 10, 103–105 (2005).
Lefoulon, C. et al. Voltage-sensor transitions of the inward-rectifying K+ channel KAT1 indicate a closed-to-open latching mechanism that is biased by hydration around the S4 alpha-helix. Plant Physiol. 166, 960–975 (2014).
Xia, L., Marques-Bueno, M. M., Bruce, C. G. & Karnik, R. Unusual Roles of Secretory SNARE SYP132 in Plasma Membrane H+-ATPase Traffic and Vegetative Plant Growth. Plant Physiol. 180, 837–858 (2019).
Waghmare, S. et al. K+ Channel-SEC11 Binding Exchange Regulates SNARE Assembly for Secretory Traffic. Plant Physiol. 181, 1096–1113 (2019).
Tyrrell, M. et al. Selective targeting of plasma membrane and tonoplast traffic by inhibitory (dominant-negative) SNARE fragments. Plant J. 51, 1099–1115 (2007).
Grefen, C. et al. A ubiquitin-10 promoter-based vector set for fluorescent protein tagging facilitates temporal stability and native protein distribution in transient and stable expression studies. Plant J. 64, 355–365 (2010).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Kellermeier, F. et al. Analysis of the Root System Architecture of Arabidopsis Provides a Quantitative Readout of Crosstalk between Nutritional Signals. Plant Cell 26, 1480–1496 (2014).
Florence, T. M. & Farrar, Y. J. Spectrophotometric Determination of Chloride At Parts-Per-Billion Level By Mercury(II) Thiocyanate Method. Analytica Chim. Acta 54, 373-& (1971).
Cataldo, D. A., Haroon, M., Schrader, L. E. & Youngs, V. L. Rapid Colorimetric Determination of Nitrate In Plant-Tissue By Nitration of Salicylic-Acid. Commun. Soil Sci. Plant Anal. 6, 71–80 (1975).
Rasband, W. S. & Bright, D. S. NIH IMAGE - A public domain image-processing program for the Macintosh. Microbeam Anal. 4, 137–149 (1995).
Warren, S. C. et al. Rapid Global Fitting of Large Fluorescence Lifetime Imaging Microscopy Datasets. Plos One 8, https://doi.org/10.1371/journal.pone.0070687 (2013).
Fässler, F. & Pimpl, P. in Plant Protein Secretion: Methods and Protocols Vol. 1662 Methods in Mol. Biol. (ed L. Jiang) 159-170 (2017).
Szabelski, M. et al. Collisional Quenching of Erythrosine B as a Potential Reference Dye for Impulse Response Function Evaluation. Appl. Spectrosc. 63, 363–368 (2009).
Van den Berg, P. A. W. et al. Exploring the conformational equilibrium of E-coli thioredoxin reductase:: Characterization of two catalytically important states by ultrafast flavin fluorescence spectroscopy. Protein Sci. 10, 2037–2049 (2001).
Lefoulon, C., Waghmare, S., Karnik, R. & Blatt, M. R. Gating control and K+ uptake by the KAT1 K+ channel leaveraged through membrane anchoring of the trafficking protein SYP121. Plant Cell Environ. 41, 2668–2677 (2018).
Eisenach, C., Papanatsiou, M., Hillert, E. K. & Blatt, M. R. Clustering of the K+ channel GORK of Arabidopsis parallels its gating by extracellular. K.+ Plant J. 78, 203–214 (2014).
Grefen, C. et al. A Novel Motif Essential for SNARE Interaction with the K+ Channel KC1 and Channel Gating in Arabidopsis. Plant Cell 22, 3076–3092 (2010).
Blatt, M. R. Electrical characteristics of stomatal guard cells: the contribution of ATP-dependent, “electrogenic” transport revealed by current-voltage and difference-current-voltage analysis. J. Membr. Biol. 98, 257–274 (1987).
Hills, A., Chen, Z. H., Amtmann, A., Blatt, M. R. & Lew, V. L. OnGuard, a Computational Platform for Quantitative Kinetic Modeling of Guard Cell Physiology. Plant Physiol. 159, 1026–1042 (2012).
Chen, Z. H. et al. Systems Dynamic Modeling of the Stomatal Guard Cell Predicts Emergent Behaviors in Transport, Signaling, and Volume Control. Plant Physiol. 159, 1235–1251 (2012).
Jezek, M., Hills, A., Blatt, M. R. & Lew, V. L. A constraint-relaxation-recovery mechanism for stomatal dynamics. Plant, Cell Environ. 42, 2399–2410 (2019).
Acknowledgements
This work was supported by BBSRC grants BB/T013508/1 and BB/T006153/1 to MRB, BB/W001217/1 and BB/X013383/1 to MRB and RK, by Royal Society University Fellowship grant URF1500364 and URF\R\211002 and BBSRC grants BB/Z516788/01 and BB/Y010272/1 to RK. We thank Naomi Donald for support in mbSUS assays and Amparo Ruiz-Pardo for help in plant maintenance.
Author information
Authors and Affiliations
Contributions
M.R.B. and R.K. conceived the work and developed the strategies for analysis with J.C.A., L.X. and C.L.; L.X. and J.C.A. carried out and analyzed the mbSUS assays; C.L. carried out oocyte electrophysiology and, with THN and FALS-A, guard cell electrophysiology; J.C.A., Z.Y. and L.X. carried out confocal, gas exchange and growth studies; M.R.B. and R.K. helped analyse the electrophysiology, gas exchange and growth data; S.W. expressed and purified channel peptides for antibodies and, with S.F., carried out and analysed co-IP experiments; M.R.B. and M.K. carried out the modelling with T.H.N.; M.R.B. wrote the manuscript with L.X., J.C.A. and R.K.; all authors edited and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xia, L., Alvim, J.C., Nguyen, TH. et al. A guard cell carbonic anhydrase binds and regulates SLAC1 separate from its catalytic activity. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70596-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70596-9