Abstract
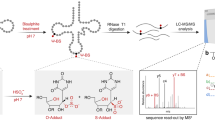
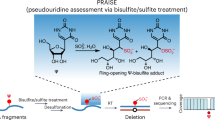
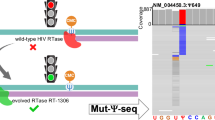
Pseudouridine (Ψ) is one of the most abundant chemical modifications and plays important roles in RNA function. Advances in our understanding of Ψ have been hindered by a limit of robust methods to precisely and sensitively map their distributions in cellular RNAs. Here, we present ELAP-seq (Enzymatic Labeling and Pull-down for Sequencing) for Ψ detection, which leverages a naturally occurring N1-methyl pseudouridine methyltransferase from Methanocaldococcus jannaschii (Mj1640). This enzyme promiscuously converts Ψ to N1-methyl-Ψ (m1Ψ) or installs a propargyl group at the same location in vitro under a mild condition, exhibiting high sensitivity and specificity, and is also functional inside cells. ELAP-seq enriches Ψ-containing RNA fragments and enables single-nucleotide-resolution Ψ detection with markedly enhanced signal-to-noise ratio and reduced sequencing and computational demands. Using ELAP-seq, we identify thousands of candidate Ψ sites in human HeLa and HEK 293 T transcriptomes, validating many previously identified sites as well as reporting additional ones. This versatile enzymatic platform expands the toolkit for sensitive labeling and detection of Ψ, advancing the study of RNA modification biology.
Similar content being viewed by others
Data availability
The NGS data generated in this paper has been deposited in the GEO database under accession code GSE236530. Source data are provided with this paper.
Code availability
The code for data analysis can be found on GitHub: https://github.com/yuruwang26/ELAP-seq-v441.
References
Roundtree, I. A., Evans, M. E., Pan, T. & He, C. Dynamic RNA modifications in gene expression regulation. Cell 169, 1187–1200 (2017).
Charette, M. & Gray, M. W. Pseudouridine in RNA: what, where, how, and why. IUBMB Life 49, 341–351 (2000).
Cerneckis, J., Cui, Q., He, C., Yi, C. & Shi, Y. Decoding pseudouridine: an emerging target for therapeutic development. Trends Pharm. Sci. 43, 522–535 (2022).
Rintala-Dempsey, A. C. & Kothe, U. Eukaryotic stand-alone pseudouridine synthases - RNA modifying enzymes and emerging regulators of gene expression? RNA Biol. 14, 1185–1196 (2017).
Repnik, K. & Potočnik, U. eQTL analysis links inflammatory bowel disease associated 1q21 locus to ECM1 gene. J. Appl. Genet. 57, 363–372 (2016).
Froukh, T. et al. Genetic basis of neurodevelopmental disorders in 103 Jordanian families. Clin. Genet. 97, 621–627 (2020).
de Brouwer, A. P. M. et al. Variants in PUS7 cause intellectual disability with speech delay, microcephaly, short stature, and aggressive behavior. Am. J. Hum. Genet. 103, 1045–1052 (2018).
Eyler, D. E. et al. Pseudouridinylation of mRNA coding sequences alters translation. Proc. Natl. Acad. Sci. USA 116, 23068–23074 (2019).
Martinez, N. M. et al. Pseudouridine synthases modify human pre-mRNA co-transcriptionally and affect pre-mRNA processing. Mol. Cell 82, 645–659.e9 (2022).
Karijolich, J. & Yu, Y.-T. Converting nonsense codons into sense codons by targeted pseudouridylation. Nature 474, 395–398 (2011).
Kierzek, E. et al. The contribution of pseudouridine to stabilities and structure of RNAs. Nucleic Acids Res. 42, 3492–3501 (2013).
Svitkin, Y. V. et al. N1-methyl-pseudouridine in mRNA enhances translation through eIF2alpha-dependent and independent mechanisms by increasing ribosome density. Nucleic Acids Res. 45, 6023–6036 (2017).
Guzzi, N. et al. Pseudouridylation of tRNA-derived fragments steers translational control in stem cells. Cell 173, 1204–1216.e26 (2018).
Guzzi, N. et al. Pseudouridine-modified tRNA fragments repress aberrant protein synthesis and predict leukaemic progression in myelodysplastic syndrome. Nat. Cell Biol. 24, 299–306 (2022).
McIntyre, W. et al. Positive-sense RNA viruses reveal the complexity and dynamics of the cellular and viral epitranscriptomes during infection. Nucleic Acids Res. 46, 5776–5791 (2018).
Nance, K. D. & Meier, J. L. Modifications in an emergency: the role of N1-methylpseudouridine in COVID-19 vaccines. ACS Cent. Sci. 7, 748–756 (2021).
Dai, Q. et al. Quantitative sequencing using BID-seq uncovers abundant pseudouridines in mammalian mRNA at base resolution. Nat. Biotechnol. 41, 344–354 (2023).
Schwartz, S. et al. Transcriptome-wide mapping reveals widespread dynamic-regulated pseudouridylation of ncRNA and mRNA. Cell 159, 148–162 (2014).
Carlile, T. M. et al. Pseudouridine profiling reveals regulated mRNA pseudouridylation in yeast and human cells. Nature 515, 143–146 (2014).
Li, X. et al. Chemical pulldown reveals dynamic pseudouridylation of the mammalian transcriptome. Nat. Chem. Biol. 11, 592–597 (2015).
Zhang, M. et al. Quantitative profiling of pseudouridylation landscape in the human transcriptome. Nat. Chem. Biol. 19, 1185–1195 (2023).
Khoddami, V. et al. Transcriptome-wide profiling of multiple RNA modifications simultaneously at single-base resolution. Proc. Natl. Acad. Sci. USA 116, 6784–6789 (2019).
Fleming, A. M. et al. Structural elucidation of bisulfite adducts to pseudouridine that result in deletion signatures during reverse transcription of RNA. J. Am. Chem. Soc. 141, 16450–16460 (2019).
Xu, H. et al. Absolute quantitative and base-resolution sequencing reveals comprehensive landscape of pseudouridine across the human transcriptome. Nat. Methods 21, 2024–2033 (2024)
Tavakoli, S. et al. Semi-quantitative detection of pseudouridine modifications and type I/II hypermodifications in human mRNAs using direct long-read sequencing. Nat. Commun. 14, 334 (2023).
Huang, S. et al. Interferon inducible pseudouridine modification in human mRNA by quantitative nanopore profiling. Genome Biol. 22, 330 (2021).
Wurm, J. P. et al. Identification of the enzyme responsible for N1-methylation of pseudouridine 54 in archaeal tRNAs. RNA 18, 412–420 (2012).
Felix, A. S., Quillin, A. L., Mousavi, S. & Heemstra, J. M. Harnessing nature’s molecular recognition capabilities to map and study RNA modifications. Acc. Chem. Res. 55, 2271–2279 (2022).
Chatterjee, K. et al. The archaeal COG1901/DUF358 SPOUT-methyltransferase members, together with pseudouridine synthase Pus10, catalyze the formation of 1-methylpseudouridine at position 54 of tRNA. RNA 18, 421–433 (2012).
Hori, H. Transfer RNA methyltransferases with a SpoU-TrmD (SPOUT) fold and their modified nucleosides in tRNA. Biomolecules 7, 23 (2017).
Purta, E., Kaminska, K. H., Kasprzak, J. M., Bujnicki, J. M. & Douthwaite, S. YbeA is the m3Psi methyltransferase RlmH that targets nucleotide 1915 in 23S rRNA. RNA 14, 2234–2244 (2008).
Wurm, J. P. et al. The ribosome assembly factor Nep1 responsible for Bowen-Conradi syndrome is a pseudouridine-N1-specific methyltransferase. Nucleic Acids Res. 38, 2387–2398 (2010).
Taoka, M. et al. Landscape of the complete RNA chemical modifications in the human 80S ribosome. Nucleic Acids Res. 46, 9289–9298 (2018).
Cui, J., Liu, Q., Sendinc, E., Shi, Y. & Gregory, R. I. Nucleotide resolution profiling of m3C RNA modification by HAC-seq. Nucleic Acids Res. 49, e27–e27 (2020).
Zhang, W., Eckwahl, M. J., Zhou, K. I. & Pan, T. Sensitive and quantitative probing of pseudouridine modification in mRNA and long noncoding RNA. RNA 25, 1218–1225 (2019).
Purchal, M. K. et al. Pseudouridine synthase 7 is an opportunistic enzyme that binds and modifies substrates with diverse sequences and structures. Proc. Natl. Acad. Sci. USA 119, e2109708119 (2022).
Levi, O. & Arava, Y. S. Pseudouridine-mediated translation control of mRNA by methionine aminoacyl tRNA synthetase. Nucleic Acids Res. 49, 432–443 (2021).
Karikó, K. et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol. Ther. 16, 1833–1840 (2008).
Breslow, D. K. et al. A comprehensive strategy enabling high-resolution functional analysis of the yeast genome. Nat. Methods 5, 711–718 (2008).
Gutgsell, N. et al. Deletion of the Escherichia coli pseudouridine synthase gene truB blocks formation of pseudouridine 55 in tRNA in vivo, does not affect exponential growth, but confers a strong selective disadvantage in competition with wild-type cells. RNA 6, 1870–1881 (2000).
Wang, Y. et al. Enzyme-mediated alkynylation enables transcriptome-wide identification of pseudouridine modifications. https://github.com/yuruwang26/ELAP-seq-v4 (2026).
Olarerin-George, A. O. & Jaffrey, S. R. MetaPlotR: a Perl/R pipeline for plotting metagenes of nucleotide modifications and other transcriptomic sites. Bioinformatics 33, 1563–1564 (2017).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. Omics 16, 284–287 (2012).
Xiao, Y.-L. et al. Transcriptome-wide profiling and quantification of N6-methyladenosine by enzyme-assisted adenosine deamination. Nat. Biotechnol. 41, 993–1003 (2023).
Acknowledgements
This research was supported by NIH RM1 HG008935 (C.H.), R35GM131858 (M.L.), P30-CA008748 (M.L.) and startup funding from the University of Utah to Y.W. C.H. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
Y.W. and C.H. conceived the idea. Y.W. designed and performed most experiments and data analysis. K.P. helped build libraries for most samples. Y.Z. performed western blots, RT-qPCR, and cell culture. T.S. performed the dot blot assays with assistance from W.Z. (Wenxin Zhao) L.Z. assisted with building LC-MS/MS channels. C.Y. helped with the establishment of the data analysis pipeline and performed part of the sequencing experiments. W.Z. (Wen Zhang) and M.A. performed CLAP and CMC treatment experiments. Q.D. synthesized the standard RNA oligonucleotides. C.S. edited the manuscript. K.W. and M.L. synthesized the propargyl-SeAm molecule. Y.W. wrote the initial manuscript. C.H. and Y.W. edited the manuscript with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
C.H. is a scientific founder and a member of the scientific advisory board of Accent Therapeutics, Inc. C.H. is also a shareholder of Epican Genetech. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Gene Yeo, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Y., Pajdzik, K., Zhao, Y. et al. Enzyme-mediated alkynylation enables transcriptome-wide identification of pseudouridine modifications. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70597-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70597-8