Abstract
Multiple drivers of global change are causing rapid biodiversity loss worldwide. However, predicting species’ trajectories remains challenging due to the dynamic and state-dependent nature of ecological responses in real-world ecosystems. Here, we leverage nonlinear time series analysis of a multi-decadal, high-resolution dataset encompassing climate, freshwater, and sediment variables, alongside estuarine macroinvertebrate abundance. Our analysis shows that key biological traits, including body size, mobility, and lifespan predict the mean and variability of the time-varying sensitivity of species to specific environmental drivers. Species with smaller body sizes or lower mobility exhibit consistently negative responses to warming. The temporal variability of species sensitivity, an aspect often overlooked in previous studies of species’ environmental responses, is strongly associated with lifespan, with shorter-lived species showing greater fluctuations over time. These findings did not always align with results from controlled laboratory or short-term field experiments, highlighting the complex, state-dependent responses of species shaped by multiple drivers of global change. We introduce a framework that links biological traits to long-term environmental responses, providing a predictive basis for trait–sensitivity relationships.
Similar content being viewed by others
Introduction
Global environmental change is multifaceted, and the concurrent action of multiple drivers of global change (e.g., global warming, water pollution, and nutrient/sediment loads) leads to uncertainty in predicting the environmental response of species1,2,3. Empirical evidence based on laboratory and field experiments has provided predictions of how species and ecosystems may respond to global changes1,2,4,5. However, little is known about how multiple global change drivers acting simultaneously affect species abundance and diversity in natural biological communities and their degree of causality over time. This gap in our understanding is due to the challenges associated with capturing the idiosyncratic, time-varying responses of species to multiple global change drivers6,7,8. Furthermore, the biology underlying the divergent responses of individual species to environmental change has rarely been assessed9,10,11. A direct test to identify which biological traits underlie such responses would provide greater generalizability and predictability of the environmental response of species5,12,13. Thus, we still lack a methodological framework that integrates causal inference, species sensitivity, and trait-based prediction to understand biodiversity responses to global changes.
Coastal and estuarine ecosystems are geographically vast and taxonomically complex, connecting continental lands and oceanic islands to their surrounding seas14,15,16,17. Estuarine macroinvertebrates, including bivalves, snails, worms, and crustaceans, are fundamental to the ecological and biogeochemical processes that influence marine ecosystem functioning15,16,18. Although macroinvertebrates are currently threatened by global climate change as well as anthropogenic disturbances, such as sedimentation and nutrient loading14,19,20,21,22,23, our ability to predict their environmental responses is limited. This knowledge gap is particularly surprising given that marine organisms are more sensitive to environmental change, especially warming, than terrestrial organisms24,25. Theoretical and empirical evidence has suggested that such climate sensitivity is due to smaller diurnal and seasonal temperature variations in the ocean than on land, and the resulting limited range of thermal tolerance of marine organisms25,26,27. Understanding how and why macroinvertebrates respond to drivers of global change over time would help to develop management strategies that prioritize the most effective actions to address specific threats to ecosystems. Such strategies will ensure the conservation and sustainable use of oceans14,22, in line with the UN Sustainable Development Goals28.
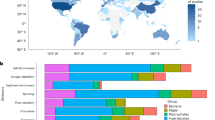
In this study, we analyze a long-term dataset of multiple global change drivers (southern oscillation index and sea surface temperature for climate variables; total suspended solids as an indicator of freshwater influence; chlorophyll-a, organic matter, and mud content of estuarine sediment as local habitat descriptors) and estuarine macroinvertebrate abundance (10–32 years of seasonal monitoring data across multiple estuarine sites, totaling ~1102 records; Supplementary Table 1) in New Zealand. Using an empirical dynamic modeling (EDM) approach6,29,30,31,32, we first quantify the causal strength of multiple global change drivers on macroinvertebrates at the community (community abundance and species richness) and species (species abundance) levels (Fig. 1a). In this study, we use the term “causal strength” to refer to the empirically detectable influence of one time-varying variable on another, as inferred from their shared dynamics in the sense of EDM7,29,30 rather than from mechanistic or intervention-based causation33.
a Convergent cross-mapping, an equation-free, nonlinear time series analysis6,29,30,31,32 was used to assess the causal strength of multiple global change drivers (climate, freshwater, and sediment variables) on macroinvertebrates. b Time-varying sensitivity of macroinvertebrates to a given driver was quantified by a locally weighted state-space regression method30,34,35,36. c Linear mixed-effects models were used to examine how biological traits determined the temporal mean and variability of species sensitivity to a given driver. The models were weighted by the causal strength of each driver on species abundance. In this figure, body size is assumed to be smallest in the small bivalve, intermediate in the worm, and largest in the large bivalve, whereas lifespan is assumed to be shorter in the worm and longer in both bivalves. We predicted that larger-bodied, longer-lived species would exhibit either more positive sensitivity or a reduced degree of negative sensitivity to intensified drivers, because they tend to be better competitors and more able to tolerate or avoid environmental stress. Accordingly, the large bivalve is expected to show positive temporal mean sensitivity, whereas the small bivalve may show negative sensitivity. In contrast, the worm, intermediate in size but short-lived, may exhibit both positive and negative sensitivity at different times, yielding a temporal mean close to zero. We further predicted that species with shorter lifespans would show greater temporal variability in sensitivity due to faster generation turnover. Thus, the worm is expected to show the largest variability (standard deviation) in sensitivity across time. Illustrations created in the Mind the Graph platform.
We then apply a locally weighted state-space regression method30,34,35,36 to accurately quantify the time-varying sensitivity of macroinvertebrates to each driver across sites (Fig. 1b). Macroinvertebrate sensitivity to global change drivers is expected to be state-dependent7,32,37; for example, warming may increase a species’ abundance in some years but decrease it in others, depending on the state of a third variable such as freshwater or sediment inputs. To capture this dynamic behavior, we quantify not only the temporal mean of species sensitivity but also its fluctuations over time, which reflect underlying shifts in ecological states7,30,38. Temporal variation in sensitivity has received limited explicit attention in previous studies of biological responses to environmental change37,38. Time-varying sensitivity is quantified as the predicted change in macroinvertebrate community abundance, species richness, and species abundance at each historical time point in response to small increases or decreases in each driver, with larger positive or negative values indicating greater sensitivity7,30. We expect that the directionality, magnitude, and range of macroinvertebrate species responses to global change drivers would differ depending on focal drivers and species.
Finally, we link differences in the sensitivity of macroinvertebrates to global change drivers with biological traits describing species morphology, physiology, and behavior that potentially determine their environmental responses (Fig. 1c)39,40,41,42. Here, the use of time-varying sensitivity alone is insufficient because species may show apparent responses to environmental drivers that are not necessarily causal. We therefore use the causal strength derived from EDM both to identify which drivers have a causal influence on macroinvertebrates and to weight the relationships between species sensitivity and biological traits. This ensures that trait-based predictions are grounded in empirically validated causal links and that the three analytical steps of causal inference, sensitivity estimation, and trait-based modeling are logically connected.
We test the overarching prediction that species with key characteristics, including larger body sizes, longer lifespans, and/or higher mobility, would show either more positive sensitivity or a reduced degree of negative sensitivity to the intensification of a given driver (Fig. 1c). These species are often superior competitors and are better able to tolerate and avoid environmental stress43,44,45. However, the expectation of an advantage for species with larger body sizes under warmer conditions runs counter to a physiologically based pattern46,47,48, observed mainly in controlled laboratory experiments49, that higher temperatures lead to smaller adult body sizes. However, in the complexity of environmental change in natural ecosystems, macroinvertebrates respond not only to global warming but also to other local environmental drivers, including freshwater and sediment inputs. For example, small inputs of organic matter or nutrients, coupled with increased metabolism and food consumption due to warming50,51 may be more advantageous for larger species. Indeed, in real ecosystems, warming-induced increases in body size within salmon populations52 and increases in the abundance of larger species at the expense of smaller species in stream-invertebrate communities53 have been observed. Moreover, we hypothesize that the temporal variability in sensitivity might be more pronounced for species with shorter lifespans because such species tend to have shorter generations, which can lead to more rapid ecological responses and thus be highly susceptible to environmental change13,54,55. These analyses allow us to predict the sensitivity of estuarine macroinvertebrates to global change drivers based on biological traits.
Results
Causal strength of global change drivers
Using convergent cross-mapping29, we quantified the causal strength of multiple global change drivers on macroinvertebrates, defined as the cross-map skill (Pearson’s ρ), across 14 estuarine sites in New Zealand (see “Materials and methods” for details). The nonlinearity parameter (θ) of sequential locally weighted global linear maps34 was positive in most cases (Supplementary Tables 2 and 3), indicating nonlinear dynamics of macroinvertebrates and supporting the use of nonlinear time series analysis.
The analysis showed that all global change drivers, except for suspended solids (based on modeled freshwater loads), had significant causal forcing on macroinvertebrate community abundance and species richness (Fig. 2). These effects were significant across sites, as determined by harmonic mean P-values that combined site-specific results. No significant differences were detected in the causal strengths of the global change drivers for community abundance and species richness. Moreover, although the abundance of many species was causally driven by multiple global change factors (Supplementary Fig. 1), certain species showed no detectable causal relationships with some drivers, indicating varying environmental sensitivity across species. These analyses incorporated the time-lagged effects of up to one year (interval of time lags varied among sites) of drivers (Supplementary Tables 4 and 5) and accounted for the spurious detection of causality between variables due to seasonality in the data.
The cross-map skill, ρ, of the CCM at the maximum time series length represents causal strength, shown as boxplots to present values across sites. Cross-mapping significance was determined by comparing ρ with the maximum time series length as well as convergence (difference between ρ at the maximum and minimum time series lengths) between original and surrogate time series data. The P value was estimated for each site as the number of surrogates showing a higher ρ with the maximum time series length, as well as a higher convergence, divided by the total number of surrogates. The meta-significance was then calculated using harmonic mean P values. Negative values of ρ indicate no predictive ability; observed values are shown without truncation to display the full range of cross-map performance. See Supplementary Table 4 for the additional statistics in the CCM. SOI southern oscillation index, SST sea surface temperature, SS suspended solids, Chl-a chlorophyll a content, OM organic matter content, mud mud content, CCM convergent cross-mapping. Sample sizes differed among drivers (SOI, SST, SS: n = 14 sites; Chl-a and OM: n = 11 sites; mud: n = 9 sites); comparisons among drivers were therefore conducted using the subset of sites with complete data for all drivers (n = 9). There were no significant differences in the causal strengths of the drivers on community abundance and species richness (paired Wilcoxon tests, two-sided; P > 0.05). Boxplots show the median (center line), interquartile range (box), and whiskers extending to the most extreme values within 1.5 times the interquartile range; points represent observed causal strengths for individual sites.
Time-varying sensitivity of macroinvertebrates
A locally weighted state-space regression method was employed to evaluate the time-varying sensitivity of macroinvertebrate community abundance, species richness, and species abundance to each driver of global change across sites (Fig. 3). Specifically, at each time point, t, we quantified the expected changes in macroinvertebrate community abundance, species richness, and species abundance at t + 1 in response to a small increase (+ΔZ/2) or decrease (−ΔZ/2) in each environmental variable, Z(t). The Δmacroinvertebrate parameter/ΔZ ratio was used to estimate the impact of a specific driver at a given time point (i.e., time-varying sensitivity), with larger positive or negative values indicating greater sensitivity. On average, community abundance responded positively to the southern oscillation index, sea surface temperature, and sediment organic content but negatively to suspended solids. Species richness responded positively to the southern oscillation index and chlorophyll a, but negatively to sea surface temperature and sediment organic matter content.
Points and bars represent the mean values and 95% confidence intervals of time-varying sensitivity. A higher positive value indicates a more positive response of community abundance, species richness, and species abundance to an increase in the specific driver, and vice versa. Colored points and bars (blue, positive; red, negative) indicate that the mean sensitivity is significantly different from zero. Species sensitivity is presented according to taxonomic groups, including Polychaeta, Bivalvia, Malacostraca, and others (Anthozoa and Thecostraca). Within each taxonomic group, species are arranged in alphabetical order. SOI southern oscillation index, SST sea surface temperature, SS suspended solids, Chl-a chlorophyll a content, OM organic matter content, mud mud content. Sample sizes varied among drivers and species, reflecting differences in data availability across sites, species presence, and time-series length; details are provided in Supplementary Tables 1 and 5.
The directionality and magnitude of species responses to global change drivers largely varied depending on focal drivers and species (Fig. 3). For species-level analyses (Figs. 3 and 4), we selected 24 common macroinvertebrate species with a total abundance across sites and years >10,000. We chose these species because less abundant species were often absent during the observation periods (zero values in the dataset) and so were not suitable for the time series analysis. A total of 49 significant species responses to global change drivers were identified.
We used a wide range of traits describing species morphology, physiology, and behavior, including body size, lifespan, mobility, movement method, reproductive frequency, and structural robustness. Trait values were standardized to have a mean of 0 and a standard deviation of 1 within each trait. Higher trait values represent greater expression of the trait. Blue and orange solid lines and shaded areas in each panel show significant (P < 0.05) fits and 95% confidence intervals of linear mixed-effect models for mean and variability of time-varying sensitivity, respectively. Dashed lines represent non-significant regression lines. Statistical significance was evaluated using two-sided linear mixed-effects models or linear models. Basically, data of time-varying sensitivity for 24 common species to each driver over the observation period across 14 estuarine sites (11 sites for species sensitivity to sediment chlorophyll a and organic matter content, and 9 sites for species sensitivity to sediment mud content) were included in the analyses. Data points are not shown for visual clarity. See Supplementary Table 6 for details of the model fit summary. SOI southern oscillation index, SST sea surface temperature, SS suspended solids, Chl-a chlorophyll a content, OM organic matter content, mud mud content.
Trait-based prediction of time-varying species sensitivity
Linear mixed-effects models were applied to examine how biological traits can predict species responses to global change drivers. These models related differences in species sensitivity to each driver with biological traits including body size, lifespan, mobility, movement method, reproductive frequency, and structural robustness (trait values were scaled to have mean 0 and standard deviation 1; greater values indicate stronger expression of a given trait). All models were weighted by the causal strength of each driver for species abundance.
Five of the six biological traits, specifically body size, lifespan, mobility, reproductive frequency, and structural robustness, predicted at least one aspect of the time-varying sensitivity of macroinvertebrate species to each driver (Fig. 4; Supplementary Table 6). For the mean time-varying sensitivity, species with smaller body sizes responded negatively, whereas those with larger body sizes responded more positively to increasing sea surface temperature. As their lifespan increased, the sensitivity of species to the increasing southern oscillation index (indicating more frequent La Niña events) shifted from positive to negative. Species with lower mobility were more negatively affected by both the southern oscillation index and sea surface temperature, whereas species with higher mobility were more robust in their response to these drivers. For the southern oscillation index, structural robustness also played a role: species with higher robustness showed a more positive response than those with lower robustness.
Temporal variability in the sensitivity of species to suspended solids, sediment chlorophyll a, organic matter, and mud content decreased with increasing lifespan. Variability in sensitivity to the Southern Oscillation Index decreased with increasing mobility, whereas that to sea surface temperature decreased with increasing reproductive frequency. Species with larger body sizes showed higher variability in their sensitivity to suspended solids and sediment organic matter content, and higher structural robustness was also associated with higher variability in responses to chlorophyll a and mud content.
Discussion
Our study provides an important advance in revealing the causal relationships linking multiple global change drivers to estuarine macroinvertebrate dynamics in real-world ecosystems. Previous studies have often relied on laboratory and field experiments or snapshot/short-term field observations to assess the dynamics of biological communities under global change1,2,3,4. Conversely, our research was based on extensive, high-quality, long-term field monitoring data that more accurately reflect the complexity of environmental change and its ecological consequences. As macroinvertebrate community abundance, species richness, and species abundance displayed nonlinear dynamics across sites (Supplementary Tables 2 and 3), a classical equation-based approach would have been inappropriate as it could not have detected causality in such complex ecological systems7,30,31. Thus, we applied an equation-free, nonlinear time series analysis6,29,30,31, through which we were able to demonstrate the importance of climate and sediment variables in driving macroinvertebrates at the community and species levels (Fig. 2; Supplementary Fig. 1). This underscores the need to prioritize management actions to address specific threats to coastal and estuarine ecosystems for both global and local environmental drivers14,22.
The key to managing threats lies in quantifying the time-varying sensitivity of macroinvertebrates to specific environmental drivers (Fig. 3). The overall positive responses of community abundance to the increasing southern oscillation index (which generally indicates warmer conditions in the western part of the South Pacific during La Niña phases) and sea surface temperature suggest an appropriate balance between increased metabolism and food consumption and increasing food resources due to warming and freshwater/sediment inputs50,56. However, species richness showed differential sensitivity to the increasing Southern Oscillation Index and sea surface temperature. This suggests differences in the causal forcing of these two climatic drivers on macroinvertebrates because the southern oscillation index reflects broader climate conditions beyond sea surface temperature, particularly precipitation patterns, through ocean–atmosphere interactions57. Furthermore, the sensitivity of individual species to the southern oscillation index differed from that to sea surface temperature (Fig. 3). Although limited to 24 common species (of many hundreds present across sites and times in the dataset), our analysis suggests that more species are negatively sensitive to sea surface temperature than to the southern oscillation index, which may explain the negative effect of increasing sea surface temperature on species richness.
Macroinvertebrates also showed varied responses to the different sediment variables (Fig. 3). Organic matter content reflects the amount of living and detrital organic material present in the sediment and is an indicator of macrobenthic food supply58,59, which is likely the reason for the positive sensitivity of community abundance to this driver. Chlorophyll a content indicates a more labile subset of the food resource pool, for example, fresh algae and microphytobenthos60. Species responded differently to these different facets of food supply, and thus, the sensitivity of species richness to organic matter content differed from that to chlorophyll a content. Although community abundance and species richness were relatively robust to increasing sediment muddiness, we found divergent species responses to this driver. As sediments become muddier, substrate permeability and oxygen penetration decrease and ammonium and sulfide increase, which can negatively affect mud-sensitive species14,61. Although the causal strength of suspended solids on community abundance and species richness was not as strong as that of the other drivers (Fig. 2), community abundance and some species responded negatively to increasing suspended solids. Suspended solids can clog the gills and feeding structures of certain suspension-feeding species14,62, which is consistent with our findings that Euchone sp. and Austrominius modestus responded negatively to increased suspended solids.
The patterns of time-varying sensitivity of the 24 individual species to specific drivers may, to some extent, appear to be inconsistent with those reported from experimental studies63,64 and spatial gradient-based observations61,65,66. In the present study, species sensitivity was quantified within the context of real-world environmental variability and long-term time series dynamics, which can diverge from the outcomes observed under more controlled experimental conditions. Species responses are highly contingent on the broader environmental context, including both the focal driver and other environmental variables, which often results in more complex and nonlinear behaviors7,30,32.
On average, species with smaller body sizes and/or lower mobility will be negatively affected by future warming of the sea surface temperature (Fig. 4). We suggest that the usual advantage of species with smaller body sizes under warmer conditions, which has been well-documented in controlled experiments46,47,48, may not be universal under complex environmental changes in real ecosystems. The effects of increasing sea surface temperature on species abundance can fluctuate over time depending on the state of freshwater and sediment inputs. Increased metabolism and food consumption due to warming48,50,51 coupled with food availability52,67 would lead to an overall positive sensitivity of larger species to warming. Mobile species are better able to avoid environmental stress because they can move to find refuges45,68 and thus are more robust to increasing sea surface temperature and the Southern Oscillation Index. Species with higher structural robustness also showed more positive responses to the Southern Oscillation Index, suggesting that greater morphological robustness may reduce vulnerability to physical or physiological stress associated with climatic fluctuations69. Oceanic and riverine nutrient supplies during La Niña phases70,71 may drive the unexpected positive sensitivity of species with shorter lifespans, as they exhibit rapid and potentially more synchronized responses to these nutrient changes.
The temporal variability in species sensitivity to global change drivers was largely predicted by the lifespan of the species. This indicates that temporal variability of species sensitivity reflects a biological signal rather than stochastic noise, underscoring its importance for understanding species’ environmental responses37,38. Short-lived species exhibit rapid ecological responses and are highly susceptible to environmental fluctuations13,54,55. This may be attributed to the increased likelihood that short-lived species are exposed to environmental stressors during critical life-history stages, especially when such disturbances occur at ecologically inappropriate times, such as during seasonal peaks in recruitment or early development72,73. As increased variability in the responses of species to environmental changes often leads to ecological breakdown in species abundance74,75, lifespan should be a robust predictor of species vulnerability to global change. In addition, our results indicate that lower mobility and reproductive frequency serve as complementary predictors of increasing variability in species sensitivity to climate drivers. Differences in body size are associated with variability in species sensitivity to sediment organic matter content, with large-bodied species appearing dependent on fluctuating macrobenthic food supply in sediments59,76. Larger-bodied invertebrates have a greater lipid storage capacity and may be more capable of coping with fluctuating food supplies77. Similarly, species with higher structural robustness showed greater variability in their sensitivity to chlorophyll-a and mud content, likely because morphologically robust species tolerate a wider range of sediment and food conditions and may therefore express a broader range of responses to short-term environmental fluctuations39,78.
While five of the six traits were associated with at least one aspect of time-varying species sensitivity, their predictive ability was generally limited, with most traits explaining particular components of sensitivity rather than providing broad or consistent predictions. Overall, the explanatory power of traits for the temporal mean and variability of species sensitivity was modest (marginal R2 ranging from 0.04 to 0.34, typically between 0.21 and 0.30), indicating that while traits can capture part of the variation in species–environment relationships, much unexplained variability remains (Fig. 4; Supplementary Table 6). This limitation likely stems from the categorical nature of the trait data used in this study39, which constrains the precision of statistical associations between traits and species sensitivities. The use of more quantitative and continuous trait measurements would likely improve the generality and predictive accuracy of trait-based models. Previous studies in other systems showed that a broader set of quantitative plant leaf traits could better predict species-abundance sensitivities to precipitation, although the analytical framework differed from ours13. Expanding trait datasets, particularly for marine benthic invertebrates, will be essential to refine predictive models and advance our understanding of the biological mechanisms underlying species responses to global change39,40,41,42.
In this study, we demonstrated that key biological traits, including body size, mobility, and lifespan, can predict the divergent responses of macroinvertebrate species to multiple global change drivers, providing a basis for generalizing species’ responses to environmental change. One challenge in increasing the predictability of trait-based ecology is the need for long-term data that capture the full range of environmental changes to which focal biota are exposed10,11,79 while accounting for the causal relationships between ecological and environmental dynamics. Here, we presented a framework to address this issue, explicitly considering the causality behind long-term environmental responses of species in detecting trait-species sensitivity relationships (Fig. 1). Given the growing accumulation of long-term data streams80,81, this framework will be applicable to a wide range of regions and ecosystems, especially those in which biological diversity is threatened by multiple global change drivers. The framework requires long-term, high-resolution time series of both environmental drivers and species abundances, ideally covering at least 30 time points29,31, to enable robust quantification of causal links and species sensitivities in the EDM. In addition, a comprehensive dataset of species traits is essential to predict species sensitivities and to link biological characteristics with causal environmental responses. Future applications of this framework will allow testing its global generalizability and extending trait-based predictions across diverse ecosystems. Our analyses largely support the overarching predictions (Fig. 1c), thus offering comprehensive evidence for a set of key traits to prioritize in the management of estuarine macroinvertebrates in response to specific drivers of global change. Finally, our work highlights the importance of integrating global change biology and trait-based ecology to externally validate how species will respond to future global changes.
Methods
Long-term macroinvertebrate data
State-of-the-environment monitoring (by the Earth Sciences New Zealand, for two regional councils in New Zealand) of macroinvertebrate species abundance is currently undertaken at 14 estuarine sites in five estuaries (Kaipara, Mahurangi, Manukau, Okura, and Raglan) on the North Island of New Zealand (Supplementary Table 1). These estuaries are morphologically diverse and large (especially the Kaipara and Manukau estuaries). They support a diverse array of human activities and are an integral part of New Zealand’s cultural identity82. Monitoring was conducted over periods ranging from 10 to 32 years, with variation in sampling intervals depending on the estuary (biannually to six times a year). Biannual sampling (in spring and autumn) represents the minimum design of this monitoring program, which was developed to detect long-term ecological changes in macroinvertebrate communities driven by chronic increases in turbidity and fine sediments during these seasons83. We used data at their original sampling intervals for each site, without adjusting them to the lowest common frequency (i.e., twice per year), as the subsequent analyses were performed on time series standardized within each site. Approximately 1120 independent sampling events were recorded (Supplementary Table 1).
As previously shown84, both macroinvertebrate community abundance (total abundance of all species) and species richness exhibited a significant positive long-term trend across sites (Supplementary Fig. 2), indicating regional-scale increases in community abundance and diversity over the monitoring period. Nevertheless, pronounced year-to-year fluctuations within sites suggest that the underlying dynamics are complex and potentially nonlinear. Our nonlinear time series analyses required continuous and equidistant data6,29,31; therefore, we imputed rare missing values for species abundance at some sites using a Kalman smoother algorithm85. Across the datasets of all the 14 sites, the values of species abundance, ranging from 2.8 to 14.6%, were imputed over the time series (Supplementary Table 1). Ongoing global warming has led to an increase in sea surface temperature, and the recent intensification of agriculture and urban development in estuary catchments has contributed to increased sediment loads and decreased water quality (Supplementary Fig. 3). Estuarine macroinvertebrates are threatened by these environmental changes, and local authorities in New Zealand are required to gather information, monitor, and commission research as necessary for effective environmental management86.
The collection of estuarine macroinvertebrates, sample processing, sorting, and identification followed standard protocols, along with strict quality assurance and quality control procedures87,88,89,90. At each estuarine site of ~1 ha, 12 replicate sediment samples (six at three sites in the Okura estuary) were collected using a hand-held PVC corer (13 cm diameter, 15 cm depth) to assess the macrobenthic fauna. Macrobenthic samples were sieved through a 0.5 mm mesh and preserved in 70% isopropanol. Benthic macrofauna were then sorted, identified to the lowest possible taxonomic level (typically genus or species), and counted. The total abundance of each macroinvertebrate species from 12 or 6 sediment samples per sampling period was used for the analysis. Each ~1 ha site was divided into an equal number of grid cells of equal size, with all grid cells sampled on each occasion, but with the exact positions of sediment core replicates randomized each time to avoid re-sampling of previously disturbed areas. The standard protocols and processing of samples by a single organization provided highly reliable and temporally consistent data suitable for investigating long-term macroinvertebrate dynamics under global change. Although 453 macroinvertebrate species (identified to different taxonomic levels) were recorded across years and sites, we used the 24 most common macroinvertebrate species (total abundance across sites and years >10,000) for species-level analyses, as datasets with many zeros (absences) are unsuitable for the type of time series analysis that we used6,30.
Long-term data of multiple global change drivers
We compiled climate (Southern Oscillation Index and sea surface temperature), freshwater (suspended solids), and sediment (chlorophy a, organic matter, and mud content in sediments) variables that potentially drive macroinvertebrate dynamics at community and species levels. The Southern Oscillation Index is a general indicator of climate variability22,23,70. Because the Southern Oscillation Index reflects broader climate conditions beyond sea surface temperature, especially changes in precipitation driven by ocean-atmosphere interactions57, it may be an important factor shaping the dynamics of coastal benthic macrofauna23. Sea surface temperature (°C) represents another climatic driver potentially influencing the abundance and behavior of marine biota22,23. The variable suspended solids (g/L) indicates exposure to reduced water clarity and seston quality at each site19,84. Sediment chlorophyll-a (mg/g), organic matter (%), and mud (%dry weight of particles <63 μm) content can vary in response to land surface water runoff and freshwater inputs, potentially influencing macroinvertebrate communities14,20,84. Although suspended solids measure the total suspended particulate matter, it is generally dominated by inorganic suspended sediment particles at intertidal estuarine sites in New Zealand84.
We examined correlations among all environmental variables within each site and found that most pairs of variables showed non-significant correlations, with limited cases exhibiting moderate but not strong associations (Supplementary Table 7). Given that significant collinearity was rare, and that causal strength was evaluated independently for each driver–macroinvertebrate pair, any potential correlation among drivers does not affect our causal inference (for details, see the section on the causality analysis). Accordingly, all variables were treated as potential drivers of macroinvertebrate dynamics rather than nuisance factors.
We derived the Southern Oscillation Index values from the National Oceanographic and Atmospheric Administration (https://www.cpc.ncep.noaa.gov). Monthly values corresponding to the macroinvertebrate time series were used. The Southern Oscillation index measures pressure variability between Tahiti and Darwin, Australia, and is linked to global changes in weather and ocean current patterns57,70. The monthly sea surface temperature values for each estuary site were derived from satellite imagery (https://oceancolor.gsfc.nasa.gov) by extracting point data from the grid cell (4 km) encompassing or nearest to each site.
To quantify suspended solids, we generated high-frequency (daily) data at all freshwater entry points into the estuaries using the New Zealand River Maps “steady state” contaminant model91,92. We modeled flows from Earth Sciences New Zealand’s TopNet component of the New Zealand Water Model (NZWaM) to calculate terminal reach loads93. We developed a distance-based metric for contaminant exposure at each site by summing the contaminant load divided by the distance from the site to each entry point. The monthly average of this summed, distance-weighted load from all entry points was then used as the freshwater exposure to suspended solids for each site84. As values for suspended solids were strongly correlated with total nitrogen and phosphorus values in the time series at all sites84, we used suspended solids alone to represent freshwater contaminants.
At each macroinvertebrate sampling event, three sediment samples for measuring chlorophyll-a, organic matter, and mud content in the sediment were collected using a 26 mm diameter, 20 mm deep PVC corer84. Sediments from each core were homogenized and subsampled for analysis87,94. Chlorophyll a was extracted from the freeze-dried sediment by boiling in 90% ethanol, and the extract was measured spectrophotometrically. An acidification step was performed to separate the degradation products (phaeophytin) from chlorophyll a95. The organic matter content was determined by drying the sediment at 60 °C for 48 h, followed by combustion at 400 °C for 5.5 h. The organic matter content is presented as the percentage of dry weight lost during ignition87,96. To determine the sediment mud content, sub-samples were first digested in ~9% hydrogen peroxide until the frothing stopped. They were then wet-sieved through 2000, 500, 250, and 63 μm meshes, with the <63 μm fraction further separated using a pipette into >3.9 μm and ≤3.9 μm sizes. All fractions were dried at 60 °C to a constant weight (~48 h) and then weighed. The percentage of dry weight of silt (3.9–63 μm) and clay (≤3.9 μm) was used as the mud content. As was the case for macroinvertebrate abundance, we imputed only minor missing values for climate, freshwater, and sediment variables at some sites (Supplementary Table 1).
Macroinvertebrate trait data
Using an extensive trait database of marine benthic invertebrates in New Zealand39, we compiled biological traits describing species morphology, physiology, and behavior that potentially determine species’ environmental responses, that is, response traits39,40,41,42. Such traits have been widely used as a proxy for the environmental responses of species, rather than as a direct test of which traits determine responses to a particular environmental driver5,12,13. All trait information in the database is categorical39, which can complicate interpretations of associations between traits and the sensitivity of macroinvertebrates to global change drivers. Therefore, we focused on traits with explicit directionality, including body size, degree of attachment (ability of an organism to attach to a substrate), lifespan, mobility, movement method, reproductive frequency, rigidity (external structural hardness of an adult organism), sediment position (relative position of an organism in the sediment), and structural robustness.
In the original database, some of the trait categories were fuzzy coded, meaning that a species could partially belong to multiple modalities within a trait category. For example, in the body size category, a species spanning all three size modalities, large (>20 mm), medium (5–20 mm), and small (0.5–5 mm), was assigned fuzzy membership values of 0.33, 0.33, and 0.33, whereas a species occurring exclusively in the large modality was assigned 1, 0, and 0, respectively39. To convert categorical traits into continuous variables, we assigned directional dummy scores to each modality within a trait category, with higher scores representing greater expression of the trait (Supplementary Table 8). For body size, we assigned 3, 2, and 1 for large, medium, and small modalities, respectively. We then multiplied the fuzzy-coded membership values by the corresponding dummy scores and summed the products to obtain a single composite trait value for each species. Because the range of assigned dummy scores differed among traits depending on the number of modalities, the resulting composite trait values were standardized to have a mean of 0 and a standard deviation of 1 within each trait. In some cases, where species were unevenly distributed among trait modalities, the standardized trait values exhibited a positive or negative skew.
We assessed the orthogonality of trait spectra using Pearson’s correlation coefficients (Supplementary Fig. 4). Consequently, we removed the degree of attachment, rigidity, and sediment position from the analysis of trait–species sensitivity relationships due to their high correlations (|r| > 0.6) with other traits.
Causality analysis between multiple global change drivers and macroinvertebrates
To determine the potential causality between multiple drivers of global change and macroinvertebrate community abundance (total abundance across species), species richness, and individual species abundance, we used empirical dynamic modeling (EDM) to detect causality, that is, convergent cross-mapping (CCM6,29,30,31). Species richness was used as the measure of species diversity because it is a conventional and widely comparable metric in studies of biodiversity responses to global changes1,2, and it equally accounts for both rare and abundant species. The core principle of CCM is to test causality by assessing the predictive relationship between two variables. Specifically, if variable X (e.g., sea surface temperature) exerts a causal influence on variable Y (e.g., community abundance), then information from X should be present in the dynamics of Y. In this context, the attractor reconstructed for Y should also have predictive power for the state of X. The prediction skill of cross mapping (cross-map skill), was quantified by calculating the Pearson correlation coefficient (ρ) between the predicted and observed values of X. To assess robustness, this process was repeated for various subsets of the X time series, each with different lengths. A consistent increase in cross-map skill as the time series lengthens (i.e., convergence) indicates that variable X likely exerts a causal influence on Y29. CCM was conducted separately for each site, because applying spatial CCM97, which pools data across sites for joint prediction, would be unlikely to satisfy the assumption of shared underlying dynamics, given differences in sampling intervals and species composition among sites. Additional theoretical details of CCM have been provided in previous papers29,31.
We standardized all-time series data to have a zero mean and unit variance prior to CCM. In this analysis, prediction skill was assessed using Pearson’s correlation coefficient (ρ) through leave-one-out cross-validation. Takens’ theorem states that the true dynamics of a system can be reconstructed from the time-lagged coordinates of a single time series98. The embedding dimension refers to the number of time-lagged coordinates used in the reconstruction29,31. As the embedding dimension is typically unknown in real-world cases, it must be estimated based on factors such as system complexity, time series length, and noise29.
We therefore employed the S-map (sequential locally weighted global linear maps) method34 to identify the optimal embedding dimension (E) and to assess the nonlinearity of the time series for community abundance, species richness, and individual species abundance, with the nonlinearity being indicated by the nonlinear localization parameter (θ). S-map, a model-free method, uses local linear regression at a given embedding dimension to predict the near future and evaluate whether a system exhibits linear or nonlinear dynamics31,34. It forecasts the system trajectory by applying locally weighted linear regression to all state-space data points31,34.
We assessed the predictability of S-map across all combinations of E (2–8) and θ (0–10) in increments of 1 and 0.1, respectively. The optimal values of E and θ were selected based on the highest prediction skill (ρ). The nonlinear localization parameter θ controls the weighting of points in the local linear map. When θ = 0, all points are weighted equally, making S-map equivalent to a global linear map. For θ > 0, nearby points receive a greater weight, allowing the map to account for nonlinear, state-dependent behavior6,30,31. The weighting function of S-map follows an exponential decay kernel, w(d) = exp( − θd/dm), where d is the Euclidean distance between the predicted and actual time points, and dm is the mean distance across all pairs. We confirmed that community abundance, species richness, and species abundance generally exhibited nonlinear dynamics (Supplementary Tables 2 and 3).
We generated 1000 surrogate time series for each driver that preserved the same seasonal cycle as the original time series but with seasonal anomalies (difference between the observed value for a given month and the monthly average across all years) randomly shuffled30,99. Synchronization of time series due to shared seasonality often leads to spurious detection of causality between focal variables6,30,99. This problem necessitates a null test using a surrogate time series. To judge the significance of CCM, we thus compared the cross-map skill (ρ) at the maximum time series length and the convergence (difference between ρ at the maximum and minimum time series lengths) between the original and surrogate time series data6,30,31.
CCM with the optimal value of E was then applied to both the original and surrogate time series of each global change driver to assess whether they could be predicted from the time series of community abundance, species richness, or species abundance. The cross-map skill, ρ, was calculated at the minimum (optimal embedding dimension E + 1) and maximum time series lengths. The cross-map skill at the maximum time series length indicates the causal strength of each driver on the macroinvertebrate parameters7,29,30,100. The P-value was estimated by counting how many surrogate time series data points had higher ρ values with the maximum time series length and greater convergence than those for the original time series, with a correction for finite sampling7. In addition, to account for potential time-delayed effects of global change drivers on macroinvertebrates23,84,101, lagged CCM102 was performed for time lags spanning a year (interval of time lags varied among sites; for example, 0, −1, −2, −3, and −4 seasons at the sites in the Kaipara estuary). The best result (highest ρ at the maximum time series length) was obtained from the lagged CCM analyses (Supplementary Tables 4 and 5).
To determine the meta-significance of CCM tests across estuarine sites, we applied a recently proposed method to combine P-values103. The meta-significance detected by this method may be relatively conservative compared to other methods to combine P-values because this method controls the family-wise error rate and is robust to interdependent samples of P-values.
We compared the causal strength of global change drivers on macroinvertebrate community abundance and species richness using paired Wilcoxon tests across 9 of the 14 estuarine sites, where causal strength was fully assessed for all drivers. Sediment data records started relatively recently at three sites in the Okura Estuary (Supplementary Table 1), with fewer than 30 time points, which is shorter than the time series length required for a nonlinear time series analysis suggested previously6,29,31. In addition, at two sites in the Raglan estuary, the sediment mud content data were very sparse. Consequently, we did not evaluate the potential causal relationships between all sediment variables and macroinvertebrates at the Okura site, nor those between sediment mud content and macroinvertebrates at the Raglan site.
Quantifying time-varying sensitivity of macroinvertebrates to global change drivers
To quantify the time-varying sensitivity of macroinvertebrates to each global change driver at each site, we used a locally weighted state-space regression method30,34,35,36. For each historical time point, t, we applied S-map34 to predict macroinvertebrate community abundance, species richness, and species abundance at t + 1 using a small increase (+ΔZ/2) and decrease (−ΔZ/2) in each environmental variable, Z(t), thereby estimating the local response (i.e., state-dependent sensitivity) of the response variable to each driver. This perturbation analysis was conducted using a bivariate embedding, where the state space was reconstructed from the lagged coordinates of the target variable and a single environmental driver. Thus, the sensitivity to each driver was evaluated independently, without incorporating interactions among multiple drivers. The forecasts were performed using the optimal values of E and θ for each macroinvertebrate parameter (see the previous section). We set ΔZ to 5% of the standard deviation of each driver, Z(t). The Δmacroinvertebrate parameter/ΔZ ratio was used to estimate the impact of a particular driver at a given time point (i.e., time-varying sensitivity), with larger positive or negative values indicating greater sensitivity in magnitude.
Analyzing relationships between biological traits and macroinvertebrate species sensitivity
Using the time-varying sensitivity values of each species to each global change driver estimated from the S-map analysis (see previous section), we examined how biological traits, including body size, lifespan, mobility, movement method, reproductive frequency, and structural robustness, determine species’ responses to specific environmental drivers. First, we calculated the mean and variability (standard deviation) of the time-varying sensitivity of each species to each driver across time points (Fig. 1b) to represent dynamic features, that is, the average strength and temporal fluctuation of species’ responses to specific environmental drivers7,30,38. We related the mean or variability of time-varying species sensitivity to each driver with biological traits (trait values were scaled) using linear mixed-effects models (Fig. 1c). All models included the within-year sampling frequency at each site (ranging from two to six observations; Supplementary Table 1), standardized to have a mean of 0 and a standard deviation of 1, as a covariate to account for potential confounding effects arising from heterogeneous temporal resolution of sampling on the estimated trait–sensitivity relationships. Random intercepts for taxonomic groups (i.e., Polychaeta, Bivalvia, Malacostraca, and others [Anthozoa and Thecostraca]) and site identity were also considered. Because the estimated random-effect variance for taxonomic groups was effectively zero in all cases, this term was first excluded from the final model structure. For site identity, the random-effect variance was effectively zero in two models, which were then fitted as linear models without the random effect (see Supplementary Table 6). To account for the causality behind long-term species’ environmental responses in detecting the trait–species sensitivity relationships, the models were weighted by the causal strength (cross-map skill at the maximum time series length) of each driver on species abundance revealed by CCM. As a cross-map skill below 0 indicates no predictive ability (predictions should not be anticorrelated with observations)29, we set negative values of causal strength to a small positive constant (1 × 10−6) in the model weights. Using alternative constants in the model weights (1 × 10−4 or 1 × 10−8) produced identical results, confirming that the choice of constant did not affect the model outcomes.
The main computations for CCM and S-map were performed using the “rEDM” package104, and all statistical analyses were performed in R version 4.4.1105.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The full-time series raw data analyzed in this study were provided with permission from Auckland Council, Waikato Regional Council, and Earth Sciences New Zealand and are subject to data use agreements; therefore, they are not publicly available. The supplementary data required for the essential steps in the analyses and for reproducing the results have been deposited in figshare106. Requests for access to the original data should be directed to the corresponding authors, who will coordinate with the data providers in accordance with the data use agreements.
Code availability
The code required to reproduce the results of this study, together with the supplementary dataset, has been deposited in figshare106.
References
Komatsu, K. J. et al. Global change effects on plant communities are magnified by time and the number of global change factors imposed. Proc. Natl. Acad. Sci. USA 116, 17867–17873 (2019).
Rillig, M. C. et al. The role of multiple global change factors in driving soil functions and microbial biodiversity. Science 366, 886–890 (2019).
Rillig, M. C. et al. Increasing the number of stressors reduces soil ecosystem services worldwide. Nat. Clim. Chang. 13, 478–483 (2023).
Reich, P. B. et al. Synergistic effects of four climate change drivers on terrestrial carbon cycling. Nat. Geosci. 13, 787–793 (2020).
Henn, J. J. et al. Long-term alpine plant responses to global change drivers depend on functional traits. Ecol. Lett. 27, e14518 (2024).
Ushio, M. et al. Fluctuating interaction network and time-varying stability of a natural fish community. Nature 554, 360–363 (2018).
Sasaki, T. et al. Dryland sensitivity to climate change and variability using nonlinear dynamics. Proc. Natl. Acad. Sci. USA 120, e2305050120 (2023).
Deyle, E. R. et al. Predicting climate effects on Pacific sardine. Proc. Natl. Acad. Sci. USA 110, 6430–6435 (2013).
Gallagher, R. V. et al. A guide to using species trait data in conservation. One Earth 4, 927–936 (2021).
Schleuning, M. et al. Trait-based assessments of climate-change impacts on interacting species. Trends Ecol. Evol. 35, 319–328 (2020).
Green, S. J., Brookson, C. B., Hardy, N. A. & Crowder, L. B. Trait-based approaches to global change ecology: moving from description to prediction. Proc. Biol. Sci. 289, 20220071 (2022).
Pacifici, M. et al. Species’ traits influenced their response to recent climate change. Nat. Clim. Chang. 7, 205–208 (2017).
Wilcox, K. R. et al. Plant traits related to precipitation sensitivity of species and communities in semiarid shortgrass prairie. N. Phytol. 229, 2007–2019 (2021).
Thrush, S. F., Hewitt, J. E. & Cummings, V. J. Muddy waters: elevating sediment input to coastal and estuarine habitats. Front. Ecol. Environ. 2, 299–306 (2004).
Lohrer, A., Thrush, S. & Gibbs, M. Bioturbators enhance ecosystem function through complex biogeochemical interactions. Nature 431, 1092–1095 (2004).
Griffiths, J. R. et al. The importance of benthic-pelagic coupling for marine ecosystem functioning in a changing world. Glob. Chang. Biol. 23, 2179–2196 (2017).
Costanza, R., Michael Kemp, W. & Boynton, W. R. Predictability, scale, and biodiversity in coastal and estuarine ecosystems: implications for management. Ambio 88, 96 (1993).
Bulmer, R. H. et al. Blue carbon habitats in Aotearoa New Zealand—opportunities for conservation, restoration, and carbon sequestration. Restor. Ecol. 32, e14225 (2024).
Robins, P. E. et al. Impact of climate change on UK estuaries: a review of past trends and potential projections. Estuar. Coast. Shelf Sci. 169, 119–135 (2016).
Clark, D. E. et al. Influence of land-derived stressors and environmental variability on compositional turnover and diversity of estuarine benthic communities. Mar. Ecol. Prog. Ser. 666, 1–18 (2021).
Pelletier, M. et al. Benthic macroinvertebrate community response to environmental changes over seven decades in an urbanized estuary in the northeastern United States. Mar. Environ. Res. 169, 105323 (2021).
Smith, K. E. et al. Biological impacts of marine heatwaves. Ann. Rev. Mar. Sci. 15, 119–145 (2023).
Hewitt, J. E., Ellis, J. I. & Thrush, S. F. Multiple stressors, nonlinear effects and the implications of climate change impacts on marine coastal ecosystems. Glob. Chang. Biol. 22, 2665–2675 (2016).
Lenoir, J. et al. Species better track climate warming in the oceans than on land. Nat. Ecol. Evol. 4, 1044–1059 (2020).
Pinsky, M., Eikeset, A. M., McCauley, D., Payne, J. & Sunday, J. Greater vulnerability to warming of marine versus terrestrial ectotherms. Nature 569, 108–111 (2019).
Sunday, J., Bates, A. & Dulvy, N. Thermal tolerance and the global redistribution of animals. Nat. Clim. Change 2, 686–690 (2012).
Pinsky, M. L., Selden, R. L. & Kitchel, Z. J. Climate-driven shifts in marine species ranges: Scaling from organisms to communities. Ann. Rev. Mar. Sci. 12, 153–179 (2020).
Winther, J.-G. et al. Integrated ocean management for a sustainable ocean economy. Nat. Ecol. Evol. 4, 1451–1458 (2020).
Sugihara, G. et al. Detecting causality in complex ecosystems. Science 338, 496–500 (2012).
Deyle, E. R., Maher, M. C., Hernandez, R. D., Basu, S. & Sugihara, G. Global environmental drivers of influenza. Proc. Natl. Acad. Sci. USA 113, 13081–13086 (2016).
Chang, C. W., Ushio, M. & Hsieh, C. H. Empirical dynamic modeling for beginners. Ecol. Res. 32, 785–796 (2017).
Ye, H. et al. Equation-free mechanistic ecosystem forecasting using empirical dynamic modeling. Proc. Natl. Acad. Sci. USA 112, E1569–E1576 (2015).
Grace, J. B. et al. Causal interpretations can be based on mechanistic knowledge. J. Ecol. 113, 3084–3098 (2025).
Sugihara, G., Grenfell, B. T., May, R. M. & Tong, H. Nonlinear forecasting for the classification of natural time series. Philos. Trans. R. Soc. Lond. Ser. A 348, 477–495 (1994).
Cenci, S., Sugihara, G. & Saavedra, S. Regularized S-map for inference and forecasting with noisy ecological time series. Methods Ecol. Evol. 10, 650–660 (2019).
Hsieh, C.-H., Glaser, S. M., Lucas, A. J. & Sugihara, G. Distinguishing random environmental fluctuations from ecological catastrophes for the North Pacific Ocean. Nature 435, 336–340 (2005).
Craine, J. M. et al. Timing of climate variability and grassland productivity. Proc. Natl. Acad. Sci. USA 109, 3401–3405 (2012).
Chang, C.-W. et al. Reconstructing large interaction networks from empirical time series data. Ecol. Lett. 24, 2763–2774 (2021).
Lam-Gordillo, O., Lohrer, A., Hewitt, J. & Dittmann, S. NZTD—The New Zealand Trait Database for shallow-water marine benthic invertebrates. Sci. Data 10, 502 (2023).
Bolam, S. G., Cooper, K. & Downie, A.-L. Mapping marine benthic biological traits to facilitate future sustainable development. Ecol. Appl. 33, e2905 (2023).
Morim, T., Henriques, S., Vasconcelos, R. & Dolbeth, M. A roadmap to define and select aquatic biological traits at different scales of analysis. Sci. Rep. 13, 22947 (2023).
McLaverty, C., Dinesen, G. E., Gislason, H., Brooks, M. E. & Eigaard, O. R. Biological traits of benthic macrofauna show sizebased differences in response to bottom trawling intensity. Mar. Ecol. Prog. Ser. 671, 1–19 (2021).
Millien, V. et al. Ecotypic variation in the context of global climate change: revisiting the rules. Ecol. Lett. 9, 853–869 (2006).
Verberk, W. C. E. P. et al. Shrinking body sizes in response to warming: explanations for the temperature-size rule with special emphasis on the role of oxygen. Biol. Rev. 96, 247–268 (2021).
Lauchlan, S. S. & Nagelkerken, I. Species range shifts along multistressor mosaics in estuarine environments under future climate. Fish. Fish. (Oxf.) 21, 32–46 (2020).
Forster, J., Hirst, A. G. & Atkinson, D. Warming-induced reductions in body size are greater in aquatic than terrestrial species. Proc. Natl. Acad. Sci. USA 109, 19310–19314 (2012).
Peck, L. S., Clark, M. S., Morley, S. A., Massey, A. & Rossetti, H. Animal temperature limits and ecological relevance: effects of size, activity and rates of change. Funct. Ecol. 23, 248–256 (2009).
Deutsch, C. et al. Impact of warming on aquatic body sizes explained by metabolic scaling from microbes to macrofauna. Proc. Natl. Acad. Sci. USA 119, e2201345119 (2022).
Bonacina, L., Fasano, F., Mezzanotte, V. & Fornaroli, R. Effects of water temperature on freshwater macroinvertebrates: a systematic review. Biol. Rev. 98, 191–221 (2023).
Scapini, F., Innocenti Degli, E. & Defeo, O. Behavioral adaptations of sandy beach macrofauna in face of climate change impacts: a conceptual framework. Estuar. Coast. Shelf Sci. 225, 106236 (2019).
Boscolo-Galazzo, F., Crichton, K. A., Barker, S. & Pearson, P. N. Temperature dependency of metabolic rates in the upper ocean: a positive feedback to global climate change? Glob. Planet. Change 170, 201–212 (2018).
Solokas, M. A. et al. Shrinking body size and climate warming: many freshwater salmonids do not follow the rule. Glob. Chang. Biol. 29, 2478–2492 (2023).
Nelson, D. et al. Experimental whole-stream warming alters community size structure. Glob. Chang. Biol. 23, 2618–2628 (2017).
Waples, R. S. & Audzijonyte, A. Fishery-induced evolution provides insights into adaptive responses of marine species to climate change. Front. Ecol. Environ. 14, 217–224 (2016).
Chessman, B. C. Identifying species at risk from climate change: traits predict the drought vulnerability of freshwater fishes. Biol. Conserv. 160, 40–49 (2013).
Rubalcaba, J. G. Metabolic responses to cold and warm extremes in the ocean. PLoS Biol. 22, e3002479 (2024).
Dayem, K. E., Noone, D. C. & Molnar, P. Tropical western Pacific warm pool and maritime continent precipitation rates and their contrasting relationships with the Walker Circulation. J. Geophys. Res. 112, D06101 (2007).
Arbi, I., Liu, S., Zhang, J., Wu, Y. & Huang, X. Detection of terrigenous and marine organic matter flow into a eutrophic semi-enclosed bay by δ13C and δ15N of intertidal macrobenthos and basal food sources. Sci. Total Environ. 613, 847–860 (2018). 614.
Szczepanek, M., Silberberger, M. J., Koziorowska-Makuch, K., Nobili, E. & Kędra, M. The response of coastal macrobenthic food-web structure to seasonal and regional variability in organic matter properties. Ecol. Indic. 132, 108326 (2021).
Huot, Y. et al. Relationship between photosynthetic parameters and different proxies of phytoplankton biomass in the subtropical ocean. Biogeosciences 4, 853–868 (2007).
Thrush, S. F. et al. Habitat change in estuaries: predicting broad-scale responses of intertidal macrofauna to sediment mud content. Mar. Ecol. Prog. Ser. 263, 101–112 (2003).
Lohrer, A. M., Hewitt, J. E. & Thrush, S. F. Assessing far-field effects of terrigenous sediment loading in the coastal marine environment. Mar. Ecol. Prog. Ser. 315, 13–18 (2006).
Lohrer, A. M. et al. Terrestrially derived sediment: response of marine macrobenthic communities to thin terrigenous deposits. Mar. Ecol. Prog. Ser. 273, 121–138 (2004).
Lohrer, A. M. et al. Deposition of terrigenous sediment on subtidal marine macrobenthos: response of two contrasting community types. Mar. Ecol. Prog. Ser. 307, 115–125 (2006).
Ellis, J. et al. Predicting macrofaunal species distributions in estuarine gradients using logistic regression and classification systems. Mar. Ecol. Prog. Ser. 316, 69–83 (2006).
Anderson, M. J. Animal-sediment relationships re-visited: Characterising species’ distributions along an environmental gradient using canonical analysis and quantile regression splines. J. Exp. Mar. Bio. Ecol. 366, 16–27 (2008).
Pennock, C. A., Budy, P., Atkinson, C. L. & Barrett, N. Effects of increased temperature on arctic slimy sculpin Cottus cognatus is mediated by food availability: Implications for climate change. Freshw. Biol. 66, 549–561 (2021).
Lamb, R. W., Smith, F. & Witman, J. D. Consumer mobility predicts impacts of herbivory across an environmental stress gradient. Ecology 101, e02910 (2020).
Beauchard, O., Veríssimo, H., Queirós, A. M. & Herman, P. M. J. The use of multiple biological traits in marine community ecology and its potential in ecological indicator development. Ecol. Indic. 76, 81–96 (2017).
Krause, J. R., Roden, A., Briceño, H. & Fourqurean, J. W. Climate oscillations drive nutrient availability and seagrass abundance at a regional scale. Limnol. Oceanogr. https://doi.org/10.1002/lno.12787 (2025)
Basterretxea, G., Font-Muñoz, J. S., Hernández-Carrasco, I. & Sañudo-Wilhelmy, S. A. Global variability of high-nutrient low-chlorophyll regions using neural networks and wavelet coherence analysis. Ocean Sci. 19, 973–990 (2023).
Pruett, J. L. et al. Life-stage-dependent effects of multiple flood-associated stressors on a coastal foundational species. Ecosphere 13, e4343 (2022).
Pineda, M. C. et al. Tough adults, frail babies: an analysis of stress sensitivity across early life-history stages of widely introduced marine invertebrates. PLoS ONE 7, e46672 (2012).
Carpenter, S. R. & Brock, W. A. Rising variance: a leading indicator of ecological transition: variance and ecological transition. Ecol. Lett. 9, 311–318 (2006).
Scheffer, M. et al. Anticipating critical transitions. Science 338, 344–348 (2012).
Cohen, J. E., Jonsson, T. & Carpenter, S. R. Ecological community description using the food web, species abundance, and body size. Proc. Natl. Acad. Sci. USA 100, 1781–1786 (2003).
Senti, T. & Gifford, M. Seasonal and taxonomic variation in arthropod macronutrient content. Food Webs 38, e00328 (2024).
Lam-Gordillo, O., Baring, R. & Dittmann, S. Taxonomic and functional patterns of benthic communities in southern temperate tidal flats. Front. Mar. Sci. 8, 723749 (2021).
Ross, S. R. P.-J. & Sasaki, T. Limited theoretical and empirical evidence that response diversity determines the resilience of ecosystems to environmental change. Ecol. Res. https://doi.org/10.1111/1440-1703.12434 (2023)
LaDeau, S. L., Han, B. A., Rosi-Marshall, E. J. & Weathers, K. C. The next decade of big data in ecosystem science. Ecosystems 20, 274–283 (2017).
Hampton, S. E. et al. Big data and the future of ecology. Front. Ecol. Environ. 11, 156–162 (2013).
Thrush, S. F. et al. The Many Uses and Values of Estuarine Ecosystems. Ecosystem Services in New Zealand–conditions and Trends. (Manaaki Whenua Press, Lincoln, New Zealand, 2013) pp 226–237.
Hewitt, J. & Gibbs, M. Assessment of the Estuarine Ecological Monitoring Programme to 2010 (2012).
Lam-Gordillo, O. et al. Climatic, oceanic, freshwater, and local environmental drivers of New Zealand estuarine macroinvertebrates. Mar. Environ. Res. 197, 106472 (2024).
Hyndman, R. obJ. & Khandakar, Y. easmin Automatic time series forecasting: the forecast Package for R. J. Stat. Softw. 27, 22 (2008).
Ministry for the Environment of New Zealand. Resource Management Act 1991. (1991).
Douglas, E. J. et al. Macrofaunal functional diversity provides resilience to nutrient enrichment in coastal sediments. Ecosystems 20, 1324–1336 (2017).
Lam-Gordillo, O. et al. Integrating rapid habitat mapping with community metrics and functional traits to assess estuarine ecological conditions: a New Zealand case study. Mar. Pollut. Bull. 206, 116717 (2024).
Greenfield, B. L. et al. Protocol for the identification of Benthic Estuarine and marine macroinvertebrates: a guide for parataxonomists. C-SIG Coastal Taxonomic Resource Tool https://www.coastalsociety.org.nz/content/view/publications/c-sig-species-heys-coastal-taxonomic-resource-tool/ (2023).
Hewitt, J. E., Hailes, S. F., Greenfield, B. L. Protocol for Processing, Identification and Quality Assurance of New Zealand Marine Benthic Invertebrate Samples. Prepared for Northland Regional Council 36 (2014).
Whitehead, A. L. & Booker, D. J. Communicating biophysical conditions across New Zealand’s rivers using an interactive webtool. N. Z. J. Mar. Freshw. Res. 53, 278–287 (2019).
Whitehead, A., Fraser, C. & Snelder, T. Spatial Modelling of River Water-Quality State: Incorporating Monitoring Data From 2016 to 2020. (NIWA Client Report 2022).
Bandaragoda, C., Tarboton, D. G. & Woods, R. Application of TOPNET in the distributed model intercomparison project. J. Hydrol. (Amst.) 298, 178–201 (2004).
Douglas, E. J. et al. Sedimentary environment influences ecosystem response to nutrient enrichment. Estuaries Coast 41, 1994–2008 (2018).
Knap, A. H., Michaels, A., Close, A. R. & Ducklow, H. Protocols for the Joint Global Ocean Flux Study (JGOFS) Core Measurements (1996).
Douglas, E. J., Lohrer, A. M. & Pilditch, C. A. Biodiversity breakpoints along stress gradients in estuaries and associated shifts in ecosystem interactions. Sci. Rep. 9, 17567 (2019).
Clark, T. et al. Spatial convergent cross mapping to detect causal relationships from short time series. Ecology 96, 1174–1181 (2015).
Takens, F. Dynamical Systems and Turbulence. (Springer-Verlag, 1981) pp 366–381.
Merz, E. et al. Disruption of ecological networks in lakes by climate change and nutrient fluctuations. Nat. Clim. Chang. 13, 389–396 (2023).
Chang, C.-W. et al. Causal networks of phytoplankton diversity and biomass are modulated by environmental context. Nat. Commun. 13, 1140 (2022).
Dittmann, S. et al. Drought and flood effects on macrobenthic communities in the estuary of Australia’s largest river system. Estuar. Coast. Shelf Sci. 165, 36–51 (2015).
Ye, H., Deyle, E. R., Gilarranz, L. J. & Sugihara, G. Distinguishing time-delayed causal interactions using convergent cross mapping. Sci. Rep. 5, 1–9 (2015).
Wilson, D. J. The harmonic mean p-value for combining dependent tests. Proc. Natl. Acad. Sci. USA 116, 1195–1200 (2019).
Ye, H., Clark, A., Deyle, E. & Sugihara, G. rEDM: an R package for Empirical Dynamic Modeling. 1–23 (2019).
R Development Core Team. R: A Language and Environment for Statistical Computing (2024).
Sasaki, T. & Lohrer, A. M. Supplementary data and codes for the article: Biological traits predict species’ time-varying responses to multiple global change drivers. figshare https://doi.org/10.6084/m9.figshare.31245691 (2026).
Acknowledgements
We thank the Auckland Council and the Waikato Regional Council for funding and their commitment to long-term macroinvertebrate monitoring in New Zealand estuaries. We gratefully acknowledge the field teams and Earth Sciences New Zealand parataxonomists for generating consistently robust time series datasets. This work was financially supported by bilateral collaboration project between New Zealand and Japan grant JPJSBP120241001 (T.S. and A.M.L.) from the Royal Society of New Zealand and the Ministry of Education, Culture, Sports, Science and Technology of Japan, Joint Research Program of Arid Land Research Center, Tottori University grant 06B2005 (T.S.), Asahi Glass Foundation (T.S.), New Zealand’s Strategic Science Investment Funding grant C01X0703 (A.M.L., O.L.G., and E.J.D.), and JST, CREST, Japan grant JPMJCR24J2 (M.K., S.S., and Y.T.).
Author information
Authors and Affiliations
Contributions
T.S., Y.I., O.L.G., K.M.C., E.J.D., R.V.G.G., B.G., S.H., K.C., N.I.I., Y.T., S.S., M.K., J.E.H., S.F.T., and A.M.L. contributed to the conceptualization of the study. Data curation was performed by O.L.G., Y.I., and T.S. Formal analyses were conducted by T.S. and Y.I. The original draft of the paper was written by T.S., Y.I., O.L.G., and A.M.L. T.S., Y.I., O.L.G., K.M.C., E.J.D., R.V.G.G., B.G., S.H., K.C., N.I.I., Y.T., S.S., M.K., J.E.H., S.F.T., and A.M.L. contributed to reviewing and editing the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Leon Barmuta, who co-reviewed with Bridget Ellen White, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sasaki, T., Iwachido, Y., Lam-Gordillo, O. et al. Biological traits predict species’ time-varying responses to multiple global change drivers. Nat Commun 17, 3950 (2026). https://doi.org/10.1038/s41467-026-70606-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-70606-w