Abstract
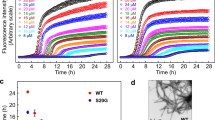
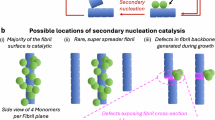
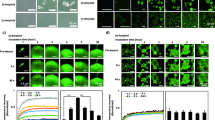
Amyloid fibrils formed by the islet amyloid polypeptide cause pancreatic beta-cell damage, resulting in reduced insulin secretion and type 2 diabetes. Changes in the amino acid sequence of this peptide can influence its aggregation rate, and animals expressing variants that do not form amyloids do not develop type 2 diabetes. Conversely, specific single amino acid changes can accelerate the aggregation rate of this peptide. Here, we employ deep mutational scanning to measure the ability of 1916 islet amyloid polypeptide variants, including substitutions, insertions, truncations and deletions, to nucleate amyloids. Our results identify a continuous stretch of residues from 15 to 32 that is particularly sensitive to mutation. This region, which is likely structured in amyloids, matches the core of the early aggregated species formed by this peptide in vitro. Within this region, mutations in residues 21 to 27 have a substantial effect, suggesting tighter structural constraints. Finally, we compare the mutational atlas of the islet amyloid polypeptide to that of amyloid beta - the peptide that aggregates in Alzheimer’s disease - and find that mutations that slow down nucleation correlate between the two amyloids, but mutations that accelerate nucleation in one amyloid cannot be used to predict mutational effects in the other.
Similar content being viewed by others
Data availability
Raw sequencing data are deposited in the European Nucleotide Archive (ENA) under project accession number: PRJEB104038. The processed data (nucleation score estimates and associated error terms) are provided in Supplementary Data 2 and have also been deposited in MaveDB84, a dedicated repository for standardized deep mutational scanning data. The processed dataset can be found by searching IAPP from the MaveDB landing page [https://www.mavedb.org/]. The Universal Resource Name (URN) for the dataset is urn:mavedb:00001253-a [https://www.mavedb.org/experiments/urn:mavedb:00001253-a]. The amyloid beta 42 nucleation dataset was obtained from Zenodo [https://doi.org/10.5281/zenodo.7255570]. The coordinates for the PDB structures used in the study were obtained with accession numbers 7M6121, 7M6221, 7M6421, 7M6521, 6Y1A14, 6ZRF23, 8R4I19, 6VW220, 8AWT22, 8AZ022, 8AZ122, 8AZ222, 8AZ322, 8AZ422, 8AZ522, 8AZ622, 8AZ722, 6ZRR23, 6ZRQ23, 7Q4M24. Variant frequency data were obtained from gnomAD v4.1.069 [https://gnomad.broadinstitute.org/gene/ENSG00000121351?dataset=gnomad_r4] and UK Biobank [https://www.ukbiobank.ac.uk/enable-your-research/register]. Source data are provided with this paper.
Code availability
All analysis scripts are available in the project’s GitHub repository [https://github.com/BEBlab/MAVE-IAPP] and in Zenodo [https://doi.org/10.5281/zenodo.18509746].
References
Opie, E. L. The relation OE diabetes mellitus to lesions of the pancreas. Hyaline degeneration of the islands OE Langerhans. J. Exp. Med. 5, 527–540 (1901).
Janson, J., Ashley, R. H., Harrison, D., McIntyre, S. & Butler, P. C. The mechanism of islet amyloid polypeptide toxicity is membrane disruption by intermediate-sized toxic amyloid particles. Diabetes 48, 491–498 (1999).
Organization, W. H. Global Report on Diabetes 83 (World Health Organization, Genève, Switzerland, 2016).
Fortin, J. S. & Benoit-Biancamano, M.-O. Wildlife sequences of islet amyloid polypeptide (IAPP) identify critical species variants for fibrillization. Amyloid 22, 194–202 (2015).
Törner, R. et al. Structural basis for the inhibition of IAPP fibril formation by the co-chaperonin prefoldin. Nat. Commun. 13, 2363 (2022).
Butler, A. E. et al. Diabetes due to a progressive defect in beta-cell mass in rats transgenic for human islet amyloid polypeptide (HIP Rat): a new model for type 2 diabetes. Diabetes 53, 1509–1516 (2004).
Höppener, J. W. et al. Extensive islet amyloid formation is induced by development of type II diabetes mellitus and contributes to its progression: pathogenesis of diabetes in a mouse model. Diabetologia 42, 427–434 (1999).
Bower, R. L. & Hay, D. L. Amylin structure-function relationships and receptor pharmacology: implications for amylin mimetic drug development. Br. J. Pharmacol. 173, 1883–1898 (2016).
Birol, M., Kumar, S., Rhoades, E. & Miranker, A. D. Conformational switching within dynamic oligomers underpins toxic gain-of-function by diabetes-associated amyloid. Nat. Commun. 9, 1312 (2018).
Rodriguez Camargo, D. C. et al. Surface-catalyzed secondary nucleation dominates the generation of toxic IAPP aggregates. Front. Mol. Biosci. 8, 757425 (2021).
Abedini, A. Time-resolved studies define the nature of toxic IAPP intermediates, providing insight for anti-amyloidosis therapeutics. eLife 5, e12977 (2016).
Cao, P., Abedini, A. & Raleigh, D. P. Aggregation of islet amyloid polypeptide: from physical chemistry to cell biology. Curr. Opin. Struct. Biol. 23, 82–89 (2013).
Xu, Y. et al. Tuning the rate of aggregation of hIAPP into amyloid using small-molecule modulators of assembly. Nat. Commun. 13, 1040 (2022).
Röder, C. et al. Cryo-EM structure of islet amyloid polypeptide fibrils reveals similarities with amyloid-β fibrils. Nat. Struct. Mol. Biol. 27, 660–667 (2020).
Abedini, A. & Raleigh, D. P. Destabilization of human IAPP amyloid fibrils by proline mutations outside of the putative amyloidogenic domain: is there a critical amyloidogenic domain in human IAPP? J. Mol. Biol. 355, 274–281 (2006).
Soriaga, A. B., Sangwan, S., Macdonald, R., Sawaya, M. R. & Eisenberg, D. Crystal structures of IAPP amyloidogenic segments reveal a novel packing motif of out-of-register beta sheets. J. Phys. Chem. B 120, 5810–5816 (2016).
Gilead, S. & Gazit, E. The role of the 14–20 domain of the islet amyloid polypeptide in amyloid formation. Exp. Diabetes Res. 2008, 256954 (2008).
Krotee, P. et al. Atomic structures of fibrillar segments of hIAPP suggest tightly mated β-sheets are important for cytotoxicity. eLife 6, e19273 (2017).
Valli, D. et al. Improving cryo-EM grids for amyloid fibrils using interface-active solutions and spectator proteins. Biophys. J. 123, 718–729 (2024).
Cao, Q., Boyer, D. R., Sawaya, M. R., Ge, P. & Eisenberg, D. S. Cryo-EM structure and inhibitor design of human IAPP (amylin) fibrils. Nat. Struct. Mol. Biol. 27, 653–659 (2020).
Cao, Q. et al. Cryo-EM structures of hIAPP fibrils seeded by patient-extracted fibrils reveal new polymorphs and conserved fibril cores. Nat. Struct. Mol. Biol. 28, 724–730 (2021).
Wilkinson, M. et al. Structural evolution of fibril polymorphs during amyloid assembly. Cell 186, 5798–5811.e26 (2023).
Gallardo, R. et al. Fibril structures of diabetes-related amylin variants reveal a basis for surface-templated assembly. Nat. Struct. Mol. Biol. 27, 1048–1056 (2020).
Yang, Y. et al. Cryo-EM structures of amyloid-β 42 filaments from human brains. Science 375, 167–172 (2022).
Colvin, M. T. et al. Atomic resolution structure of monomorphic Aβ42 amyloid fibrils. J. Am. Chem. Soc. 138, 9663–9674 (2016).
Gremer, L. et al. Fibril structure of amyloid-ß(1-42) by cryo-electron microscopy. Science 358, 116–119 (2017).
Lührs, T. et al. 3D structure of Alzheimer’s amyloid-beta(1-42) fibrils. Proc. Natl. Acad. Sci. USA 102, 17342–17347 (2005).
Wälti, M. A. et al. Atomic-resolution structure of a disease-relevant Aβ(1-42) amyloid fibril. Proc. Natl. Acad. Sci. USA 113, E4976–E4984 (2016).
Xiao, Y. et al. Aβ(1-42) fibril structure illuminates self-recognition and replication of amyloid in Alzheimer’s disease. Nat. Struct. Mol. Biol. 22, 499–505 (2015).
Vandiver, L. In vitro interactions between amyloid beta and islet amyloid polypeptide. Biophys. J. 106, 56a (2014).
Bharadwaj, P. et al. Amylin and beta amyloid proteins interact to form amorphous heterocomplexes with enhanced toxicity in neuronal cells. Sci. Rep. 10, 10356 (2020).
Kachkin, D. V. et al. The Aβ42 peptide and IAPP physically interact in a yeast-based assay. Int. J. Mol. Sci. 24, 14122 (2023).
Hu, R., Zhang, M., Chen, H., Jiang, B. & Zheng, J. Cross-seeding interaction between β-amyloid and human islet amyloid polypeptide. ACS Chem. Neurosci. 6, 1759–1768 (2015).
Tabet, D., Parikh, V., Mali, P., Roth, F. P. & Claussnitzer, M. Scalable functional assays for the interpretation of human genetic variation. Annu. Rev. Genet. 56, 441–465 (2022).
Fowler, D. M. & Fields, S. Deep mutational scanning: a new style of protein science. Nat. Methods 11, 801–807 (2014).
Starita, L. M. et al. Variant interpretation: functional assays to the rescue. Am. J. Hum. Genet. 101, 315–325 (2017).
Findlay, G. M. Linking genome variants to disease: scalable approaches to test the functional impact of human mutations. Hum. Mol. Genet. 30, R187–R197 (2021).
Beltran, A., Jiang, X., Shen, Y. & Lehner, B. Site-saturation mutagenesis of 500 human protein domains. Nature 637, 885–894 (2025).
Matreyek, K. A. et al. Multiplex assessment of protein variant abundance by massively parallel sequencing. Nat. Genet. 50, 874–882 (2018).
Seuma, M., Faure, A., Badia, M., Lehner, B. & Bolognesi, B. The genetic landscape for amyloid beta fibril nucleation accurately discriminates familial Alzheimer’s disease mutations. eLife 10, e63364 (2021).
Seuma, M., Lehner, B. & Bolognesi, B. An atlas of amyloid aggregation: the impact of substitutions, insertions, deletions and truncations on amyloid beta fibril nucleation. Nat. Commun. 13, 7084 (2022).
Tatarek-Nossol, M. et al. Inhibition of hIAPP amyloid-fibril formation and apoptotic cell death by a designed hIAPP amyloid-core-containing hexapeptide. Chem. Biol. 12, 797–809 (2005).
Chandramowlishwaran, P. et al. Mammalian amyloidogenic proteins promote prion nucleation in yeast. J. Biol. Chem. 293, 3436–3450 (2018).
Lee, K.-H., Zhyvoloup, A. & Raleigh, D. P. Amyloidogenicity and cytotoxicity of des-Lys-1 human amylin provides insight into amylin self-assembly and highlights the difficulties of defining amyloidogenicity. Protein Eng. Des. Sel. 32, 87–93 (2019).
Lee, K.-H., Noh, D., Zhyvoloup, A. & Raleigh, D. P. Analysis of Prairie Vole amylin reveals the importance of the N-terminus and residue 22 in amyloidogenicity and cytotoxicity. Biochemistry 59, 408–419 (2020).
Akter, R. et al. Evolutionary adaptation and amyloid formation: does the reduced amyloidogenicity and cytotoxicity of ursine amylin contribute to the metabolic adaption of bears and polar bears? Isr. J. Chem. 57, 750–761 (2017).
Fortier, M. et al. Contribution of the 12–17 hydrophobic region of islet amyloid polypeptide in self-assembly and cytotoxicity. Front. Mol. Biosci. 9, 1017336 (2022).
Khemtemourian, L., Guillemain, G., Foufelle, F. & Killian, J. A. Residue-specific effects of human islet polypeptide amyloid on self-assembly and cell toxicity. Biochim. Biophys. Acta Biomembr. 1859, 745–753 (2017).
Tu, L.-H. & Raleigh, D. P. Role of aromatic interactions in amyloid formation by islet amyloid polypeptide. Biochemistry 52, 333–342 (2013).
Young, L. M., Tu, L.-H., Raleigh, D. P., Ashcroft, A. E. & Radford, S. E. Understanding co-polymerization in amyloid formation by direct observation of mixed oligomers. Chem. Sci. 8, 503–509 (2017).
Godin, E., Nguyen, P. T., Zottig, X. & Bourgault, S. Identification of a hinge residue controlling islet amyloid polypeptide self-assembly and cytotoxicity. J. Biol. Chem. 294, 8452–8463 (2019).
Ridgway, Z. et al. Analysis of proline substitutions reveals the plasticity and sequence sensitivity of human IAPP amyloidogenicity and toxicity. Biochemistry 59, 634–646 (2020).
Akter, R. et al. Amyloidogenicity, cytotoxicity, and receptor activity of bovine amylin: implications for xenobiotic transplantation and the design of nontoxic amylin variants. ACS Chem. Biol. 13, 2748–2759 (2018).
Miller, M. E. T. et al. Analysis of sheep and goat IAPP provides insight into IAPP amyloidogenicity and cytotoxicity. Biochemistry 61, 2600–2610 (2022).
Fox, A. et al. Selection for non-amyloidogenic mutants of islet amyloid polypeptide (IAPP) identifies an extended region for amyloidogenicity. Biochemistry 49, 7783–7789 (2010).
Bakou, M. et al. Key aromatic/hydrophobic amino acids controlling a cross-amyloid peptide interaction versus amyloid self-assembly. J. Biol. Chem. 292, 14587–14602 (2017).
Cao, P. et al. Sensitivity of amyloid formation by human islet amyloid polypeptide to mutations at residue 20. J. Mol. Biol. 421, 282–295 (2011).
Ridgway, Z. et al. Analysis of baboon IAPP provides insight into amyloidogenicity and cytotoxicity of human IAPP. Biophys. J. 118, 1142–1151 (2020).
Tenidis, K. et al. Identification of a penta- and hexapeptide of islet amyloid polypeptide (IAPP) with amyloidogenic and cytotoxic properties. J. Mol. Biol. 295, 1055–1071 (2000).
Wiltzius, J. J. W. et al. Atomic structure of the cross-beta spine of islet amyloid polypeptide (amylin). Protein Sci. 17, 1467–1474 (2008).
Faidon Brotzakis, Z. et al. Determination of the structure and dynamics of the fuzzy coat of an amyloid fibril of IAPP using cryo-electron microscopy. Biochemistry 62, 2407–2416 (2023).
Notredame, C., Higgins, D. G. & Heringa, J. T-coffee: a novel method for fast and accurate multiple sequence alignment. J. Mol. Biol. 302, 205–217 (2000).
Tartaglia, G. G. & Vendruscolo, M. The Zyggregator method for predicting protein aggregation propensities. Chem. Soc. Rev. 37, 1395–1401 (2008).
Fernandez-Escamilla, A.-M., Rousseau, F., Schymkowitz, J. & Serrano, L. Prediction of sequence-dependent and mutational effects on the aggregation of peptides and proteins. Nat. Biotechnol. 22, 1302–1306 (2004).
Sormanni, P., Aprile, F. A. & Vendruscolo, M. The CamSol method of rational design of protein mutants with enhanced solubility. J. Mol. Biol. 427, 478–490 (2015).
Moffat, L. & Jones, D. T. Increasing the accuracy of single sequence prediction methods using a deep semi-supervised learning framework. Bioinformatics 37, 3744–3751 (2021).
Cheng, J. et al. Accurate proteome-wide missense variant effect prediction with AlphaMissense. Science 381, eadg7492 (2023).
Orenbuch, R. et al. Proteome-wide model for human disease genetics. Nat. Genet. https://doi.org/10.1101/2023.11.27.23299062 (2025).
Chen, S. et al. A genomic mutational constraint map using variation in 76,156 human genomes. Nature 625, 92–100 (2024).
Westermark, P., Andersson, A. & Westermark, G. T. Islet amyloid polypeptide, islet amyloid, and diabetes mellitus. Physiol. Rev. 91, 795–826 (2011).
Sakagashira, S. et al. Missense mutation of amylin gene (S20G) in Japanese NIDDM patients. Diabetes 45, 1279–1281 (1996).
Cho, Y. M., Kim, M., Park, K. S., Kim, S. Y. & Lee, H. K. S20G mutation of the amylin gene is associated with a lower body mass index in Korean type 2 diabetic patients. Diabetes Res. Clin. Pract. 60, 125–129 (2003).
Sakagashira, S. et al. S20G mutant amylin exhibits increased in vitro amyloidogenicity and increased intracellular cytotoxicity compared to wild-type amylin. Am. J. Pathol. 157, 2101–2109 (2000).
Sudlow, C. et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12, e1001779 (2015).
Evans, P. H., Pereira Gray, D. J., Wright, C. & Langley, P. Diagnosing type 2 diabetes and identifying high-risk individuals using the new glycated haemoglobin (HbA1c) criteria. Br. J. Gen. Pract. 63, 235 (2013).
Liu, W., Han, J., Gong, W., Zhang, F. & Cao, Q. Structure of pancreatic hIAPP fibrils derived from patients with type 2 diabetes. Cell 189, 1201–1210.e10 (2026).
Rodriguez Camargo, D. C. et al. The redox environment triggers conformational changes and aggregation of hIAPP in type II diabetes. Sci. Rep. 7, 44041 (2017).
Yan, X. et al. Intra-condensate demixing of TDP-43 inside stress granules generates pathological aggregates. Cell 188, 4123–4140 (2025).
Faure, A. J., Schmiedel, J. M., Baeza-Centurion, P. & Lehner, B. DiMSum: an error model and pipeline for analyzing deep mutational scanning data and diagnosing common experimental pathologies. Genome Biol. 21, 207 (2020).
Bolognesi, B. et al. The mutational landscape of a prion-like domain. Nat. Commun. 10, 4162 (2019).
Joosten, R. P. et al. A series of PDB related databases for everyday needs. Nucleic Acids Res. 39, D411–D419 (2011).
Tokuriki, N., Stricher, F., Schymkowitz, J., Serrano, L. & Tawfik, D. S. The stability effects of protein mutations appear to be universally distributed. J. Mol. Biol. 369, 1318–1332 (2007).
Wu, M. C. et al. Rare-variant association testing for sequencing data with the sequence kernel association test. Am. J. Hum. Genet. 89, 82–93 (2011).
Rubin, A. F. et al. MaveDB 2024: a curated community database with over seven million variant effects from multiplexed functional assays. Genome Biol. 26, 13 (2025).
Topolska, M., Beltran, A. & Lehner, B. Deep indel mutagenesis reveals the impact of amino acid insertions and deletions on protein stability and function. Nat. Commun. 16, 2617 (2025).
Acknowledgements
M.B. was supported by the fellowship “Ayudas para contratos predoctorales para la formación de doctores 2019” (PRE2019-088300) from the Spanish Ministry of Science, Innovation and Universities. The project that gave rise to these results has received funding from “la Caixa” Research Foundation under the grant agreement LCF/PR/HR21/52410004 (project ‘DeepAmyloids’). Work in the lab of B.B. is also supported by the Spanish Ministry of Science, Innovation and Universities (PID2021-127761OB-I00 and RYC2020-028861-I, funded by MCIN/AEI/10.13039/501100011033, “ERDF A way of making Europe” and “ESF Investing in your future”), and by the European Union (ERC Consolidator, Glam-MAP, 101125484). Views and opinions expressed are, however, those of the author(s) only and do not necessarily reflect those of the European Union or the European Research Council. Neither the European Union nor the granting authority can be held responsible for them. IBEC is a member of the CERCA Program/Generalitat de Catalunya. We thank the Chernoff and the Lehner lab for providing strains and plasmids, and the CRG Genomics core technology for sequencing. We thank Prof. John Perry, Dr. Yajie Zhao, Prof. Ben Lehner, Carla Folgado, and the B.B. lab members for discussing our data.
Author information
Authors and Affiliations
Contributions
B.B. conceptualized and supervised the project. C.B. and M.B. designed the libraries and performed the experiments. M.B. analyzed the data and conducted the UK Biobank analyses. B.B. and M.B. interpreted the results and wrote the manuscript. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Badia, M., Batlle, C. & Bolognesi, B. Massively parallel quantification of mutational impact on IAPP amyloid formation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70611-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70611-z