Abstract
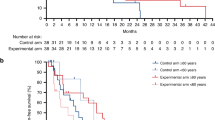
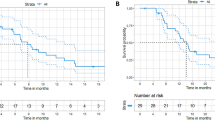
Immunotherapy achieved remarkable results in patients with deficient mismatch repair (dMMR)/microsatellite instable (MSI) metastatic colorectal carcinoma (mCRC). However, its efficacy in proficient MMR (pMMR)/microsatellite stable (MSS) mCRC remains limited. In the phase II NIVACOR trial, we evaluated the activity and safety of FOLFOXIRI/bevacizumab plus nivolumab as first-line therapy in patients with RAS/BRAF-mutated mCRC (NCT04072198). The primary endpoint of the trial was the Objective Response Rate (ORR) whereas secondary endpoints were safety profile, overall survival (OS), progression free survival (PFS), duration of response (DoR) and quality of life. The primary endpoint was met. Among the 73 enrolled patients, 76.7% achieved an objective response (95% CI, 65.4 to 85.8%), while the disease control rate was 97.3% (95% CI, 90.5 to 99.7%). The median progression-free survival (mPFS) was 10.1 months (95% CI, 9.0 to 14.3 months), and the median overall survival (mOS) was not reached. Treatment-related adverse events of grade 3 or higher occurred in 48 patients out 73 enrolled patients (65.8%). Comprehensive genomic profiling and RNA sequencing analysis revealed genomic and transcriptomic profiles associated with treatment response in pMMR/MSS patients. Alterations in pathways such as PI3K/AKT, chemokine signaling and DNA repair showed correlation with treatment activity. These findings highlight the potential synergy between immune checkpoint inhibitors and cytotoxic chemotherapy in selected patients with pMMR/MSS mCRC.
Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within the article. To protect the privacy of the patients in the study, clinical information of individual RAS and BRAF mutated mCRC patients have been anonymized and reported in Fig. 1, Supplementary Fig. 2 and in the Supplementary Data file. Source data and further clinical data can be requested to the corresponding author within the limitations of the patient informed consent. De-identified sequencing data have been deposited in the Genome Sequencing Archive (accession code HRA016779). The access is restricted and requires approval by the NGDC Data Access Committee upon request to the corresponding author. The accession request will be reviewed within a timeframe of 2-4 weeks and data will be available in accordance with participant consent and applicable regulations. Sequencing data will be also available in the Zenodo repository (https://doi.org/10.5281/zenodo.17358523).
References
Siegel, R. L., Miller, K. D., Fuchs, H. E. & Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 72, 7–33 (2022).
Biller, L. H. & Schrag, D. Diagnosis and treatment of metastatic colorectal cancer: a review. JAMA 325, 669–685 (2021).
Cervantes, A. et al. Metastatic colorectal cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann. Oncol. 34, 10–32 (2023).
Cremolini, C. et al. FOLFOXIRI plus bevacizumab versus FOLFIRI plus bevacizumab as first-line treatment of patients with metastatic colorectal cancer: updated overall survival and molecular subgroup analyses of the open-label, phase 3 TRIBE study. Lancet Oncol. 16, 1306–1315 (2015).
Cancer Genome Atlas, N. Comprehensive molecular characterization of human colon and rectal cancer. Nature 487, 330–337 (2012).
Ma, J., Setton, J., Lee, N. Y., Riaz, N. & Powell, S. N. The therapeutic significance of mutational signatures from DNA repair deficiency in cancer. Nat. Commun. 9, 3292 (2018).
Boland, C. R. & Goel, A. Microsatellite instability in colorectal cancer. Gastroenterology 138, 2073–2087.e2073 (2010).
Andre T. et al. Safety and efficacy of anti-PD-1 antibody dostarlimab in patients (pts) with mismatch repair deficient (dMMR) GI cancers. J. Clin. Oncol. 38, 3 (2021).
Diaz, L. A. Jr. et al. Pembrolizumab versus chemotherapy for microsatellite instability-high or mismatch repair-deficient metastatic colorectal cancer (KEYNOTE-177): final analysis of a randomised, open-label, phase 3 study. Lancet Oncol. 23, 659–670 (2022).
Lenz, H. J. et al. First-line nivolumab plus low-dose ipilimumab for microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: the phase II Checkmate 142 study. J. Clin. Oncol. 40, 161–170 (2022).
El Hajj, J., Reddy, S., Verma, N., Huang, E. H. & Kazmi, S. M. Immune checkpoint inhibitors in pMMR/MSS colorectal cancer. J. Gastrointest. Cancer 54, 1017–1030 (2023).
Aldea, M. et al. Overcoming resistance to tumor-targeted and immune-targeted therapies. Cancer Discov. 11, 874–899 (2021).
Antoniotti, C. et al. Upfront FOLFOXIRI plus bevacizumab with or without atezolizumab in the treatment of patients with metastatic colorectal cancer (AtezoTRIBE): a multicentre, open-label, randomised, controlled, phase 2 trial. Lancet Oncol. 23, 876–887 (2022).
Antoniotti, C. et al. Upfront fluorouracil, leucovorin, oxaliplatin, and irinotecan plus bevacizumab with or without atezolizumab for patients with metastatic colorectal cancer: updated and overall survival results of the ATEZOTRIBE study. J. Clin. Oncol. 42, 2637–2644 (2024).
Esposito Abate, R. et al. Harmonization of tumor mutation burden testing with comprehensive genomic profiling assays: an IQN Path initiative. J. Immunother. Cancer 12, e007800 (2024).
Guinney, J. et al. The consensus molecular subtypes of colorectal cancer. Nat. Med 21, 1350–1356 (2015).
Dienstmann, R. et al. Consensus molecular subtypes and the evolution of precision medicine in colorectal cancer. Nat. Rev. Cancer 17, 268 (2017).
Joanito, I. et al. Single-cell and bulk transcriptome sequencing identifies two epithelial tumor cell states and refines the consensus molecular classification of colorectal cancer. Nat. Genet 54, 963–975 (2022).
Normanno, N. et al. Resistance to immune checkpoint inhibitors in colorectal cancer with deficient mismatch repair/microsatellite instability: misdiagnosis, pseudoprogression and/or tumor heterogeneity? Explor Target Antitumor Ther. 5, 495–507 (2024).
Li, Y. et al. Efficacy and safety of anti-PD-1/PD-L1 therapy in the treatment of advanced colorectal cancer: a meta-analysis. BMC Gastroenterol. 22, 431 (2022).
Rachiglio, A. M. et al. Colorectal cancer genomic biomarkers in the clinical management of patients with metastatic colorectal carcinoma. Explor Target Antitumor Ther. 1, 53–70 (2020).
Lenz, H. J. et al. Modified FOLFOX6 plus bevacizumab with and without nivolumab for first-line treatment of metastatic colorectal cancer: phase 2 results from the CheckMate 9X8 randomized clinical trial. J. Immunother. Cancer 12, e008409 (2024).
Herting, C. J. et al. A multi-center, single-arm, phase Ib study of pembrolizumab (MK-3475) in combination with chemotherapy for patients with advanced colorectal cancer: HCRN GI14-186. Cancer Immunol. Immunother. 70, 3337–3348 (2021).
Wang, H., Tang, R., Jiang, L. & Jia, Y. The role of PIK3CA gene mutations in colorectal cancer and the selection of treatment strategies. Front Pharm. 15, 1494802 (2024).
Voutsadakis, I. A. The landscape of PIK3CA mutations in colorectal cancer. Clin. Colorectal Cancer 20, 201–215 (2021).
Voutsadakis, I. A. High tumor mutation burden (TMB) in microsatellite stable (MSS) colorectal cancers: Diverse molecular associations point to variable pathophysiology. Cancer Treat. Res Commun. 36, 100746 (2023).
Ugai, T. et al. Association of PIK3CA mutation and PTEN loss with expression of CD274 (PD-L1) in colorectal carcinoma. Oncoimmunology 10, 1956173 (2021).
Matteucci, L., Bittoni, A., Gallo, G., Ridolfi, L. & Passardi, A. Immunocheckpoint inhibitors in microsatellite-stable or proficient mismatch repair metastatic colorectal cancer: are we entering a new era? Cancers (Basel) 15, 5189 (2023).
Herrera, F. G., Bourhis, J. & Coukos, G. Radiotherapy combination opportunities leveraging immunity for the next oncology practice. CA Cancer J. Clin. 67, 65–85 (2017).
Zhao, Y. & Chen, S. Targeting DNA double-strand break (DSB) repair to counteract tumor radio-resistance. Curr. Drug Targets 20, 891–902 (2019).
McGrail, D. J. et al. High tumor mutation burden fails to predict immune checkpoint blockade response across all cancer types. Ann. Oncol. 32, 661–672 (2021).
Acknowledgements
This was a nonprofit study, conducted in the GOIRC clinical research network. Bristol Myers Squibb partially covered the costs of the study. The study was also supported by Ricerca Corrente grant L3/8 from Ministero della Salute to REA. NN have received support from Ministero della Salute (Project T3-AN-06 “Sviluppo di una piattaforma per la implementazione clinica della oncologia di precisione nelle regioni del centro-sud Italia”). We thank all patients and their families, all caregivers, the referring oncologists and the GOIRC group for the participating centers.
Author information
Authors and Affiliations
Contributions
Conceptualization: F.I., N.N., C.P. and A.D. Data Curation: S.T., A.D. and G.M. Investigation: R.E.A., S.T., F.B., L.A., G.N., F.P., G.T., T.L., R.B., G.R., E.G., M.L., E.M., S.L. and A.R. Validation: R.E.A. and S.T. Resources: D.F., M.R.M. and A.D.L. Formal analysis: R.E.A., D.R., and G.M. Funding acquisition: C.P. and A.D. Supervision: N.N., C.P. and A.D. Writing – original draft: R.E.A., N.N., A.D. and G.M. Writing–review & editing All authors.
Corresponding author
Ethics declarations
Competing interests
Angela Damato: outside the submitted work, has received personal fees for the advisory role, speaker engagements, and travel and accommodation expenses from Ipsen, Servier, BMS, Merck Serono, Amgen, and Daiichi Sankyo. Francesca Bergamo: received personal honoraria as invited speaker from Eli-Lilly, MSD, Bristol Myers Squibb, AstraZeneca, Bayer, Pierre Fabre; participation in advisory board for AAA Novartis, Takeda, Teysuno. Filippo Pietrantonio: honoraria: Servier, Bayer, AstraZeneca, Lilly, MSD, Amgen, Pierre-Fabre, Merck-Serono, BMS, Astellas. Consulting or Advisory Role: Merck-Serono, Amgen, Servier, MSD, Organon, Bayer. Research Funding: Bristol Myers Squibb (Inst), AstraZeneca (Inst), Incyte, Agenus. Giuseppe Tonini: advisory board member of Molteni, MSD, Novartis, Roche, and Pharmamar. Elisa Giommoni: advisory board of Eli Lilly, Amgen, Viatris, BMS, Servier. Sara Leonardi: received honoraria (as invited speaker) from Roche, Eli Lilly, BMS, Servier, Merck Serono, Pierre Fabre, GSK, and Amgen; consulting fee (advisory boards) from Amgen, Astellas, Bayer, Merck Serono, Eli Lilly, AstraZeneca, Incyte, Daiichi-Sankyo, BMS, Servier, Merck Sharp & Dohme (MSD), GSK, Takeda, Rottapharm, and Beigene; as well as grants or funds (to institution) from Amgen, Merck Serono, Bayer, Roche, Eli Lilly, AstraZeneca, and BMS. Nicola Normanno: outside the submitted work personal fees for the advisory role, speaker engagements, and travel and accommodation expenses from AstraZeneca, Bayer, Biocartis, Bristol Myers Squibb, Eli Lilly, Illumina, Incyte, MERCK, Merck Sharp & Dohme, Novartis, Roche, Servier, Thermofisher; financial support to research projects from Astrazeneca, Biocartis, Illumina, MERCK, QIAGEN, Roche, Sophia Genetics, Thermofisher. Carmine Pinto: outside the submitted work personal fees for the advisory role, speaker engagements, and travel and accommodation expenses from Amgen, Astellas, AstraZeneca, Bayer, Bristol Meyer Squibb, Celgene, Clovis Oncology, Eisai, Ipsen, Janssen, Incyte, Merck-Serono, Merck Sharp and Dohme, Novartis, Roche, Sandoz, Sanofi, and Servier. The other authors have no conflicts of interest to declare.
Peer review
Peer review information
Nature Communications thanks Youqiong Ye and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Damato, A., Esposito Abate, R., Tessitore, S. et al. First-line Nivolumab plus FOLFOXIRI/Bevacizumab in advanced RAS/BRAF-mutated colorectal cancer: efficacy, safety and biomarker discovery from the phase II NIVACOR trial. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70620-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70620-y