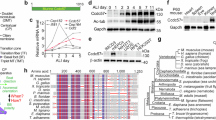
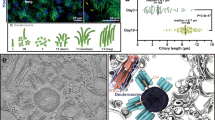
Abstract
Multiciliated cells generate numerous centrioles to support motile cilia essential for fluid flow. This process was thought to rely on the deuterosome, a DEUP1-dependent structure. Here, we show that DEUP1 plays a critical role in the basal foot (BF)—an asymmetric appendage of the basal body required for coordinated ciliary beating. Using high-resolution imaging, we localize DEUP1 to the middle tier of the BF, adjacent to CNTRL. In DEUP1 knockout mice, BF architecture is disrupted, leading to loss of rotational planar polarity, reduced cerebrospinal fluid flow, and ciliary degeneration with age. These defects are recapitulated in Xenopus epidermis, demonstrating evolutionary conservation. Notably, DEUP1 loss alone does not affect basal body or cilia number during early or mature adult stages, with overt degeneration emerging only in aged animals. Together, these findings establish DEUP1 as a key structural regulator of BF integrity that is required for the long-term maintenance of coordinated ciliary motility.
Similar content being viewed by others
Data availability
Source data are available for graphs plotted in Figs. 1–7 and supplementary Figs. 1–6. The data that support the findings of this study are openly available in figshare with [DOI: 10.6084/m9.figshare.31006945]. Source data are provided with this paper.
References
Spassky, N. & Meunier, A. The development and functions of multiciliated epithelia. Nat. Rev. Mol. Cell Biol. 18, 423–436 (2017).
Spassky, N. et al. Coupling between hydrodynamic forces and planar cell polarity orients mammalian motile cilia. Nat. Cell Biol. 12, 341–350 (2010).
Mirzadeh, Z., Han, Y., Soriano-Navarro, M., Garcia-Verdugo, J. M. & Alvarez-Buylla, A. Cilia organize ependymal planar polarity. J. Neurosci. 30, 2600–2610 (2010).
Breunig, J. J., Arellano, J. I. & Rakic, P. Cilia in the brain: going with the flow. Nat. Neurosci. 13, 654–655 (2010).
Lyu, Q., Li, Q., Zhou, J. & Zhao, H. Formation and function of multiciliated cells. J. cell Biol. 223, 1 (2024).
Kumar, D. & Reiter, J. How the centriole builds its cilium: of mothers, daughters, and the acquisition of appendages. Curr. Opin. Struct. Biol. 66, 41–48 (2021).
Zhao, H. et al. The Cep63 paralogue Deup1 enables massive de novo centriole biogenesis for vertebrate multiciliogenesis. Nat. cell Biol. 15, 1434–1444 (2013).
Al Jord, A. et al. Centriole amplification by mother and daughter centrioles differs in multiciliated cells. Nature. 516, 104–107 (2014).
Zhao, H. et al. Parental centrioles are dispensable for deuterosome formation and function during basal body amplification. EMBO Rep. 20, e46735 (2019).
Mercey, O. et al. Massive centriole production can occur in the absence of deuterosomes in multiciliated cells. Nat. Cell Biol. 21, 1544–1552 (2019).
Mazo, G., Soplop, N., Wang, W., Uryu, K. & Tsou, M. B. Spatial control of primary ciliogenesis by subdistal appendages alters sensation-associated properties of cilia. Dev. Cell. 39, 424–437 (2016).
Chong, W. M. et al. Super-resolution microscopy reveals coupling between mammalian centriole subdistal appendages and distal appendages. eLife. 9, e53580 (2020).
Yang, T. T. et al. Super-resolution architecture of mammalian centriole distal appendages reveals distinct blade and matrix functional components. Nat. Commun. 9, 2023–11 (2018).
Tanos, B. E. et al. Centriole distal appendages promote membrane docking, leading to cilia initiation. Genes Dev. 27, 163–168 (2013).
Kanie, T. et al. The CEP19-RABL2 GTPase complex binds IFT-B to initiate intraflagellar transport at the ciliary base. Dev. Cell. 42, 22–36.e12 (2017).
Li, S., Fernandez, J., Marshall, W. F. & Agard, D. A. Three-dimensional structure of basal body triplet revealed by electron cryo-tomography. EMBO J. 31, 552–562 (2012).
Kunimoto, K. et al. Coordinated ciliary beating requires Odf2-mediated polarization of basal bodies via basal feet. Cell. 148, 189–200 (2012).
Ryu, H. et al. The molecular dynamics of subdistal appendages in multi-ciliated cells. Nat. Commun. 12, 1–16 (2021).
Soares, H., Carmona, B., Nolasco, S., Melo, L. V. & Gonçalves, J. Cilia distal domain: diversity in evolutionarily conserved structures. Cells. 8, 160 (2019).
Herawati, E. et al. Multiciliated cell basal bodies align in stereotypical patterns coordinated by the apical cytoskeleton. J. Cell Biol. 214, 571–586 (2016).
Mirzadeh, Z., Doetsch, F., Sawamoto, K., Wichterle, H. & Alvarez-Buylla, A. The subventricular zone en-face: wholemount staining and ependymal flow. Journal of Visualized Experiments https://doi.org/10.3791/193 (2010).
Ohata, S. & Álvarez-Buylla, A. Planar organization of multiciliated ependymal (E1) cells in the brain ventricular epithelium. Trends Neurosci. 39, 543–551 (2016).
Kim, S. K. et al. CLAMP/Spef1 regulates planar cell polarity signaling and asymmetric microtubule accumulation in the Xenopus ciliated epithelia. J. Cell Biol. 217, 1633–1641 (2018).
Wallingford, J. B. & Mitchell, B. Strange as it may seem: the many links between Wnt signaling, planar cell polarity, and cilia. Genes Dev. 25, 201–213 (2011).
Park, T. J., Mitchell, B. J., Abitua, P. B., Kintner, C. & Wallingford, J. B. Dishevelled controls apical docking and planar polarization of basal bodies in ciliated epithelial cells. Nat. Genet 40, 871–879 (2008).
Mitchell, B., Jacobs, R., Li, J., Chien, S. & Kintner, C. A positive feedback mechanism governs the polarity and motion of motile cilia. Nature. 447, 97–101 (2007).
Ishikawa, H., Kubo, A., Tsukita, S. & Tsukita, S. Odf2-deficient mother centrioles lack distal/subdistal appendages and the ability to generate primary cilia. Nat. Cell Biol. 7, 517–524 (2005).
Nguyen, Q. P. H. et al. Comparative super-resolution mapping of basal feet reveals a modular but distinct architecture in primary and motile cilia. Dev. cell. 55, 209–223.e7 (2020).
Kokovay, E. et al. VCAM1 is essential to maintain the structure of the SVZ niche and acts as an environmental sensor to regulate SVZ lineage progression. Cell Stem Cell. 11, 220–230 (2012).
Siller, S. S. et al. Conditional knockout mice for the distal appendage protein CEP164 reveal its essential roles in airway multiciliated cell differentiation. PLOS Genet. 13, e1–e1007128 (2017).
Hatch, E. M., Kulukian, A., Holland, A. J., Cleveland, D. W. & Stearns, T. Cep152 interacts with Plk4 and is required for centriole duplication. J. cell Biol. 191, 721–729 (2010).
Kishimoto, N. & Sawamoto, K. Planar polarity of ependymal cilia. Differentiation. 83, 86 (2012).
Brooks, E. & Wallingford, J. Multiciliated cells. Curr. Biol. 24, R973–R982 (2014).
Ran, F. A. et al. In vivo genome editing using Staphylococcus aureus Cas9. Nature. 520, 186–191 (2015).
Mahuzier, A. et al. Ependymal cilia beating induces an actin network to protect centrioles against shear stress. Nat. Commun. 9, 2279–227 (2018).
Mercey, O. et al. Dynamics of centriole amplification in centrosome-depleted brain multiciliated progenitors. Sci. Rep. 9, 13060–11 (2019).
Werner, M. E. et al. Actin and microtubules drive differential aspects of planar cell polarity in multiciliated cells. J. Cell Biol. 195, 19–26 (2011).
Labedan, P. et al. Dissection and staining of the mouse brain ventricular wall for the analysis of ependymal cell cilia organization. Bio-protocol https://doi.org/10.21769/BioProtoc.1757 (2016).
Delgehyr, N. et al. in Methods in Cilia and Flagella. 19–35 (Elsevier Science & Technology, United States, 2015).
Pan, X. et al. Directional ciliary beats across epithelia require Ccdc57-mediated coupling between axonemal orientation and basal body polarity. Nat. Commun. 15, 10249–16 (2024).
Fisher, N. I. in Statistical Analysis of Circular Data (Cambridge University Press, Cambridge, 1993).
Takagishi, M., Yue, Y., Gray, R. S., Verhey, K. J. & Wallingford, J. B. Motor protein Kif6 regulates cilia motility and polarity in brain ependymal cells. Dis. Model. Mech. 17, dmm050137 (2024).
Le, T. T., Im, G. H., Lee, C. H., Choi, S. H. & Kim, S. Mapping cerebral perfusion in mice under various anesthesia levels using highly sensitive BOLD MRI with transient hypoxia. Sci. Adv. 10, eadm7605 (2024).
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) under grant RS-2024-00341130 (S.P.), and by grant RS-2023-KH134516 (S.P.) from the Korea Health Industry Development Institute (KHIDI) and the Korea Dementia Research Center (KDRC). H.L. and J.L. received additional support from the NRF through grants RS-2021-NR061936 and RS-2024-00449811, respectively. We thank the Institute for Basic Science (IBS) Center for Neuroscience Imaging Research (IBS-R015-D1) for providing MRI time and professional technical support. We also thank Kaushik Neha for assistance with microinjection of EGFP-tagged Xenopus deup1 mRNA into embryos.
Author information
Authors and Affiliations
Contributions
H.L., J.L., and M.S. contributed to the experimental design, conducted the experiments, and analyzed the data. R.S.G. and J.K. were responsible for microinjection of morpholinos or mRNAs and provided Xenopus embryos. S.P. conceived and supervised the overall study, designed all experiments, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
;Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, H., Lee, J., Shin, M. et al. DEUP1 functions as a scaffold for basal foot integrity and planar polarity in multiciliated cells. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70661-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70661-3