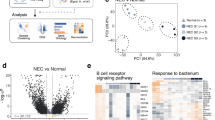
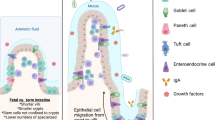
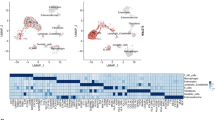
Abstract
Cell polarity is essential for maintaining intestinal epithelial organization and function. Here we show that combined loss of polarity by epithelial loss of Cdc42 with oncogenic Kras expression in mice causes small intestine failure leading to weight loss, inflammation, epithelial necroptosis, and lethality. These phenotypic defects are characterized by a loss of intestinal stem cells, disrupted epithelial architecture, altered hippo signaling, elevated inflammatory cytokines, and activation of necroptotic cell death, that closely resemble necrotizing enterocolitis (NEC). Single-cell transcriptomic analysis reveals a coordinated dysregulation of polarity machinery, inflammatory pathways, and necroptosis program. Suppression of YAP, IL-1, TNFα signaling or necroptosis rescues the intestinal pathology. Similar NEC-like phenotypes arise when Cdc42 loss and oncogenic Kras activation are initiated from intestinal stem cells. These findings provide mechanism insights involving polarity-YAP-IL1/TNFα signaling induced necroptosis for the synergistic effect of hyperactivation of Kras signaling and loss of polarity in disrupting intestinal epithelia.
Similar content being viewed by others
Data availability
All data are included in the Supplementary Information or available from the authors, as are unique reagents used in this Article. The raw numbers for charts and graphs are available in the Source Data file whenever possible. Source data are provided with this paper. The scRNA-seq data generated in this study were deposited at the Gene Expression Omnibus (RRID:SCR_005012) under accession number GSE294390 (GSM8903735, GSM8903736, GSM8903737, GSM8903738). The hyperlinks for these data are. GSE294390(study). GSM8903735(WT). GSM8903736(CDC42 KO). GSM8903737(Kras). GSM8903738(CDC42 KO/Kras Source data are provided with this paper.
References
Etienne-Manneville, S. & Hall, A. Rho GTPases in cell biology. Nature 420, 629–635 (2002).
Woods, B., Kuo, C. C., Wu, C. F., Zyla, T. R. & Lew, D. J. Polarity establishment requires localized activation of Cdc42. J. Cell Biol. 211, 19–26 (2015).
Woods, B. & Lew, D. J. Polarity establishment by Cdc42: Key roles for positive feedback and differential mobility. Small GTPases 10, 130–137 (2019).
Melendez, J. et al. Cdc42 coordinates proliferation, polarity, migration, and differentiation of small intestinal epithelial cells in mice. Gastroenterology 145, 808–819 (2013).
Zhang, Z. et al. CDC42 controlled apical-basal polarity regulates intestinal stem cell to transit amplifying cell fate transition via YAP-EGF-mTOR signaling. Cell Rep. 38, 110009 (2022).
Downward, J. Targeting RAS signalling pathways in cancer therapy. Nat. Rev. Cancer 3, 11–22 (2003).
Goel, S., Huang, J. & Klampfer, L. K-Ras, intestinal homeostasis and colon cancer. Curr. Clin. Pharm. 10, 73–81 (2015).
Haigis, K. M. et al. Differential effects of oncogenic K-Ras and N-Ras on proliferation, differentiation and tumour progression in the colon. Nat. Genet. 40, 600–608 (2008).
Prior, I. A., Hood, F. E. & Hartley, J. L. The frequency of ras mutations in cancer. Cancer Res. 80, 2969–2974 (2020).
Ferreira, A. et al. Crucial role of oncogenic KRAS mutations in apoptosis and autophagy regulation: therapeutic implications. Cells 11, 2183 (2022).
Meng, M. et al. The current understanding on the impact of KRAS on colorectal cancer. Biomed. Pharmacother. 140, 111717 (2021).
Negri, F. & Bottarelli, L. de'Angelis, G. L. & Gnetti, L. KRAS: a druggable target in colon cancer patients. Int. J. Mol. Sci. 23, 4120 (2022).
Neu, J. & Walker, W. A. Necrotizing enterocolitis. N. Engl. J. Med. 364, 255–264 (2011).
Hackam, D. J. & Sodhi, C. P. Bench to bedside - new insights into the pathogenesis of necrotizing enterocolitis. Nat. Rev. Gastroenterol. Hepatol. 19, 468–479 (2022).
Becht, E. et al. Dimensionality reduction for visualizing single-cell data using UMAP. Nat. Biotechnol. 37, 38–44 (2018).
Barry, E. R. et al. Restriction of intestinal stem cell expansion and the regenerative response by YAP. Nature 493, 106–110 (2013).
Gregorieff, A., Liu, Y., Inanlou, M. R., Khomchuk, Y. & Wrana, J. L. Yap-dependent reprogramming of Lgr5(+) stem cells drives intestinal regeneration and cancer. Nature 526, 715–718 (2015).
Broz, P. & Dixit, V. M. Inflammasomes: mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 16, 407–420 (2016).
Vijayaraj, S. L. et al. The ubiquitylation of IL-1beta limits its cleavage by caspase-1 and targets it for proteasomal degradation. Nat. Commun. 12, 2713 (2021).
Pasparakis, M. & Vandenabeele, P. Necroptosis and its role in inflammation. Nature 517, 311–320 (2015).
Seo, J., Nam, Y. W., Kim, S., Oh, D. B. & Song, J. Necroptosis molecular mechanisms: Recent findings regarding novel necroptosis regulators. Exp. Mol. Med. 53, 1007–1017 (2021).
Klinke, M. et al. Development of an improved murine model of necrotizing enterocolitis shows the importance of neutrophils in NEC pathogenesis. Sci. Rep. 10, 8049 (2020).
Mikus, P. et al. Determination of novel highly effective necrostatin Nec-1s in rat plasma by high-performance liquid chromatography hyphenated with quadrupole-time-of-flight mass spectrometry. Molecules 23, 1946 (2018).
Xie, J., Zhang, Y. & Jiang, L. Role of Interleukin-1 in the pathogenesis of colorectal cancer: a brief look at anakinra therapy. Int Immunopharmacol. 105, 108577 (2022).
Dinarello, C. A. Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol. Rev. 281, 8–27 (2018).
Quintero, M. & Samuelson, L. C. Paneth cells: dispensable yet irreplaceable for the intestinal stem cell niche. Cell Mol. Gastroenterol. Hepatol. 19, 101443 (2025).
Schuijers, J., van der Flier, L. G., van Es, J. & Clevers, H. Robust cre-mediated recombination in small intestinal stem cells utilizing the olfm4 locus. Stem Cell Rep. 3, 234–241 (2014).
Liu, M. et al. RHOA GTPase controls YAP-mediated EREG signalling in small intestinal stem cell maintenance. Stem Cell Rep. 9, 1961–1975 (2017).
Barker, N. et al. Identification of stem cells in the small intestine and colon by the marker gene Lgr5. Nature 449, 1003–1007 (2007).
Wodarz, A. & Nathke, I. Cell polarity in development and cancer. Nat. Cell Biol. 9, 1016–1024 (2007).
Muthuswamy, S. K. & Xue, B. Cell polarity as a regulator of cancer cell behaviour plasticity. Annu Rev. Cell Dev. Biol. 28, 599–625 (2012).
Peglion, F. & Etienne-Manneville, S. Cell polarity changes in cancer initiation and progression. J. Cell Biol. 223, e202308069 (2024).
Zhu, G., Pei, L., Xia, H., Tang, Q. & Bi, F. Role of oncogenic KRAS in the prognosis, diagnosis and treatment of colorectal cancer. Mol. Cancer 20, 143 (2021).
Yang, S. et al. Programmed death of intestinal epithelial cells in neonatal necrotizing enterocolitis: a mini-review. Front Pediatr. 11, 1199878 (2023).
Zhang, Z., Liu, M. & Zheng, Y. Role of Rho GTPases in stem cell regulation. Biochem Soc. Trans. 49, 2941–2955 (2021).
Seo, Y., Park, S. Y., Kim, H. S. & Nam, J. S. The Hippo-YAP signaling as guardian in the pool of intestinal stem cells. Biomedicines 8, 560 (2020).
Yang, J. & Shi, Y. Paneth cell development in the neonatal gut: pathway regulation, development, and relevance to necrotizing enterocolitis. Front Cell Dev. Biol. 11, 1184159 (2023).
Frank, D. & Vince, J. E. Pyroptosis versus necroptosis: similarities, differences, and crosstalk. Cell Death Differ. 26, 99–114 (2019).
Kearney, C. J. & Martin, S. J. An inflammatory perspective on necroptosis. Mol. Cell 65, 965–973 (2017).
Newton, K. & Manning, G. Necroptosis and inflammation. Annu Rev. Biochem. 85, 743–763 (2016).
Silke, J., Rickard, J. A. & Gerlic, M. The diverse role of RIP kinases in necroptosis and inflammation. Nat. Immunol. 16, 689–697 (2015).
Zhou, Y. et al. RIPK3 signaling and its role in regulated cell death and diseases. Cell Death Discov. 10, 200 (2024).
Campbell J., Y. S., Wang Z., Corbett S., Koga Y. Celda: cellular latent dirichlet allocation. R package version 1.22.0. (2024).
McGinnis, C. S., Murrow, L. M. & Gartner, Z. J. DoubletFinder: doublet detection in single-cell RNA sequencing data using artificial nearest neighbours. Cell Syst. 8, 329–337 e324 (2019).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587 e3529 (2021).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Acknowledgments
We thank the Cincinnati Children’s Hospital Medical Center Discover Together Biobank for support of this study, as well as participants and their families, whose help and participation made this work possible. We thank the expert technical support of James F. Johnson. This work was partly supported by NIH P30 DK078392 (L.D.), NIH U54 DK126108 (Y.Z.), NIH R01 AG063967 (Y.Z.), and NIH R01 CA278756 (Y.Z.).
Author information
Authors and Affiliations
Contributions
Z.Z. conceived the project, designed experiments, performed experiments, carried out data analyses and interpretation, and wrote the paper with input from the rest of the authors. C.F. performed the flow cytometry study. R.J. assisted in performing experiments and manuscript preparation. P.S. provided human NEC samples for analysis. M.A. performed bioinformatic analysis on the single-cell RNAseq data. Y.Z. provided overall supervision of the study, conceived the project, designed experiments, and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interest.
Peer review
Peer review information
Nature Communications thanks Youqiong Ye and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Z., Fan, C., Jorgensen, R. et al. Loss of polarity by Cdc42 depletion and oncogenic Kras activation in the mouse intestinal epithelia leads to a necrotizing enterocolitis (NEC)-like disease. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70677-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70677-9