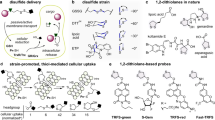
Abstract
The cyclic five-membered disulfide 1,2-dithiolane, known for its moderate reactivity and ease of preparation, holds significant promise as a recognition unit in probe design and drug development. However, potential limitations in activation specificity—most notably the diminished selectivity toward thioredoxin reductase (TrxR) caused by nonspecific reactions with abundant low-molecular-weight (LMW) thiols such as glutathione (GSH)—raise concerns about its reliability in biological settings. Here we systematically evaluate the activation behavior of 1,2-dithiolane by synthesizing a panel of prodrugs and fluorescent probes incorporating either amine- or hydroxyl-linked cargoes. Our results reveal that TrxR-mediated selective activation of the 1,2-dithiolane unit is achievable when the cargo is incorporated within an amine-based framework. In contrast, hydroxyl-linked conjugates undergo rapid cleavage by physiological GSH levels, resulting in a pronounced loss of TrxR selectivity. Generally, the recognition site 1,2-dithiolane, the linker unit and the leaving group in a cargo coordinate to determine the selectivity activated by TrxR. Overall, this study resolves ambiguities in previous reports, reconciles conflicting observations, and provides new conceptual guidance for the use of the 1,2-dithiolane scaffold in the design of biofunctional molecules.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are available in the main text and Supplementary Information, and from the corresponding author(s) upon request. Source data are provided with this paper.
References
Sies, H., Mailloux, R. J. & Jakob, U. Fundamentals of redox regulation in biology. Nat. Rev. Mol. Cell Biol. 25, 701–719 (2024).
Lennicke, C. & Cochemé, H. M. J. M. C. Redox metabolism: ROS as specific molecular regulators of cell signaling and function. Mol. Cell 81, 3691–3707 (2021).
D’Autréaux, B. & Toledano, M. B. ROS as signalling molecules: mechanisms that generate specificity in ROS homeostasis. Nat. Rev. Mol. Cell Biol. 8, 813–824 (2007).
Shadel, G. S. & Horvath, T. L. Mitochondrial ROS signaling in organismal homeostasis. Cell 163, 560–569 (2015).
Arnér, E. S. & Holmgren, A. J. E. J. O. B. Physiological functions of thioredoxin and thioredoxin reductase. Eur. J. Biochem. 267, 6102–6109 (2000).
Zhang, J., Li, X., Han, X., Liu, R. & Fang, J. Targeting the thioredoxin system for cancer therapy. Trends Pharm. Sci. 38, 794–808 (2017).
Turanov, A. A. et al. Mammalian thioredoxin reductase 1: roles in redox homoeostasis and characterization of cellular targets. Biochem. J. 430, 285–293 (2010).
Ardini, M. et al. The “Doorstop Pocket” in thioredoxin reductases─an unexpected druggable regulator of the catalytic machinery. J. Med. Chem. 67, 15947–15967 (2024).
Gencheva, R. & Arnér, E. S. J. Thioredoxin reductase inhibition for cancer therapy. Annu. Rev. Pharm. 62, 177–196 (2022).
Wientjens, C. et al. Tolerance to ferroptosis facilitates lipid metabolism and pathogenic type 2 immunity in allergic airway inflammation. Immunity 59, 1–17 (2025).
Zhang, L. et al. Highly selective off–on fluorescent probe for imaging thioredoxin reductase in living cells. J. Am. Chem. Soc. 136, 226–233 (2014).
Liu, Y. et al. A small molecule probe reveals declined mitochondrial thioredoxin reductase activity in a Parkinson’s disease model. Chem. Commun. 52, 2296–2299 (2016).
Zhao, J. et al. Loss of thioredoxin reductase function in a mouse stroke model disclosed by a two-photon fluorescent probe. Chem. Commun. 56, 14075–14078 (2020).
Ma, H., Zhang, J., Zhang, Z., Liu, Y. & Fang, J. A fast response and red emission probe for mammalian thioredoxin reductase. Chem. Commun. 52, 12060–12063 (2016).
Li, X. et al. A fast and specific fluorescent probe for thioredoxin reductase that works via disulphide bond cleavage. Nat. Commun. 10, 2745 (2019).
Mafireyi, T. J. et al. A diselenide turn-on fluorescent probe for the detection of thioredoxin reductase. Angew. Chem. Int. Ed. 59, 15147–15151 (2020).
Zeisel, L. et al. Selective cellular probes for mammalian thioredoxin reductase TrxR1: Rational design of RX1, a modular 1,2-thiaselenane redox probe. Chem 8, 1493–1517 (2022).
Yang, Y.-P. et al. Fast imaging of mitochondrial thioredoxin reductase using a styrylpyridinium-based two-photon ratiometric fluorescent probe. Anal. Chem. 94, 4970–4978 (2022).
Zhang, Q. et al. CBNP: an anti-photobleaching probe for detecting thioredoxin reductase activity in cancer cells. Microchem. J. 219, 115906 (2025).
Mauker, P. et al. A modular scaffold for cellularly-retained fluorogenic probes for sensitive cell-resolved bioactivity imaging. Chem. Sci. 16, 22630–22637 (2025).
Sidhu, J. S., Singh, A., Garg, N. & Singh, N. Carbon dot based, naphthalimide coupled FRET pair for highly selective ratiometric detection of thioredoxin reductase and cancer screening. ACS Appl. Mater. Interfaces 9, 25847–25856 (2017).
Yan, F. et al. High-throughput fluorescent screening of thioredoxin reductase inhibitors to inhibit Mycobacterium tuberculosis. Chin. Chem. Lett. 35, 108504 (2024).
Li, X. et al. Selective activation of a prodrug by thioredoxin reductase providing a strategy to target cancer cells. Angew. Chem. Int. Ed. 57, 6141–6145 (2018).
Chang, B. et al. Puromycin prodrug activation by thioredoxin reductase overcomes its promiscuous cytotoxicity. J. Med. Chem. 66, 3250–3261 (2023).
Huang, L. et al. A furanyl acryl conjugated coumarin as an efficient inhibitor and a highly selective off-on fluorescent probe for covalent labelling of thioredoxin reductase. Chem. Commun. 50, 6987–6990 (2014).
Liang, B. et al. Mitochondria-targeted approach: remarkably enhanced cellular bioactivities of TPP2a as selective inhibitor and probe toward TrxR. Acs Chem. Biol. 11, 425–434 (2016).
Zhang, J. et al. Small molecule inhibitors of mammalian thioredoxin reductase as potential anticancer agents: an update. Med. Res. Rev. 39, 5–39 (2019).
Felber, J. G. et al. Cyclic 5-membered disulfides are not selective substrates of thioredoxin reductase, but are opened nonspecifically. Nat. Commun. 13, 1754 (2022).
Lee, M. H., Sessler, J. L. & Kim, J. S. Disulfide-based multifunctional conjugates for targeted theranostic drug delivery. Acc. Chem. Res. 48, 2935–2946 (2015).
Lee, M. H. et al. Disulfide-cleavage-triggered chemosensors and their biological applications. Chem. Rev. 113, 5071–5109 (2013).
Wang, P., Gong, Q., Hu, J., Li, X. & Zhang, X. Reactive oxygen species (ROS)-responsive prodrugs, probes, and theranostic prodrugs: applications in the ROS-related diseases. J. Med Chem. 64, 298–325 (2021).
Wang, H., Monroe, M., Leslie, F., Flexner, C. & Cui, H. Supramolecular nanomedicines through rational design of self-assembling prodrugs. Trends Pharm. Sci. 43, 510–521 (2022).
Shin, W. S. et al. Cancer targeted enzymatic theranostic prodrug: precise diagnosis and chemotherapy. Bioconjug. Chem. 27, 1419–1426 (2016).
Liu, W. et al. Hypoxia-activated anticancer prodrug for bioimaging, tracking drug release, and anticancer application. Bioconjug. Chem. 29, 3332–3343 (2018).
Kumar, R. et al. Hypoxia-directed and activated theranostic agent: imaging and treatment of solid tumor. Biomaterials 104, 119–128 (2016).
Zhao, J. et al. Integration of a diselenide unit generates fluorogenic camptothecin prodrugs with improved cytotoxicity to cancer cells. J. Med. Chem. 64, 17979–17991 (2021).
Yang, Y. et al. Thiol–chromene “Click” reaction triggered self-Immolative for NIR visualization of thiol flux in physiology and pathology of living cells and mice. J. Am. Chem. Soc. 142, 1614–1620 (2020).
Hu, G., Xu, H.-D. & Fang, J. Sulfur-based fluorescent probes for biological analysis: a review. Talanta 279, 126515 (2024).
Ma, T. et al. Selenenylsulfide bond as a general scaffold for constructing thiol probes with enhanced response rate. Anal. Chem. 97, 6629–6637 (2025).
Ciccolini, J., Mercier, C., Dahan, L. & André, N. Integrating pharmacogenetics into gemcitabine dosing—time for a change? Nat. Rev. Clin. Oncol. 8, 439–444 (2011).
Weiss, J. T. et al. Development and bioorthogonal activation of palladium-labile prodrugs of gemcitabine. J. Med. Chem. 57, 5395–5404 (2014).
Yarmolinsky, M. B. & Haba, G. L. D. L. Inhibition by puromycin of amino acid incorporation into protein. Proc. Natl. Acad. Sci. USA 45, 1721–1729 (1959).
Xi, J. et al. Redox dyshomeostasis-driven prodrug strategy for enhancing camptothecin-based chemotherapy: Selenization of SN38 as a case study. Bioorg. Chem. 160, 108468 (2025).
Staben, L. R. et al. Targeted drug delivery through the traceless release of tertiary and heteroaryl amines from antibody-drug conjugates. Nat. Chem. 8, 1112–1119 (2016).
Alouane, A., Labruere, R., Le Saux, T., Schmidt, F. & Jullien, L. Self-immolative spacers: kinetic aspects, structure-property relationships, and applications. Angew. Chem. Int. Ed. 54, 7492–7509 (2015).
Wang, X., Zhuang, Y., Wang, Y., Jiang, M. & Yao, L. The recent developments of camptothecin and its derivatives as potential anti-tumor agents. Eur. J. Med. Chem. 260, 115710 (2023).
Bhuniya, S. et al. An activatable theranostic for targeted cancer therapy and imaging. Angew. Chem. Int. Ed. 53, 4469–4474 (2014).
Kim, E. J. et al. An activatable prodrug for the treatment of metastatic tumors. J. Am. Chem. Soc. 136, 13888–13894 (2014).
Zhao, L. et al. Baylis-Hillman adducts as a versatile module for constructing fuorogenic release system. J. Med. Chem. 65, 6056–6069 (2022).
Qin, G. et al. A selective mitochondria-targeted fluorescent probe for imaging cysteine in drug-induced liver injury. Talanta 282, 127056 (2024).
Qin, G. et al. A nonconsumptive fluorescent probe for precise detection of hydrogen peroxide in nonalcoholic fatty liver disease and inflammation. Anal. Chem. 97, 7195–7202 (2025).
Zhou, Z. et al. Construction of a stable TrxR1 knockout HCT-116 cell line using CRISPR/Cas9 gene editing system. Chin. J. Biotechnol. 38, 1074–1085 (2022).
Du, Y., Zhang, H., Lu, J. & Holmgren, A. Glutathione and glutaredoxin act as a backup of human thioredoxin reductase 1 to reduce thioredoxin 1 preventing cell death by aurothioglucose*. J. Biol. Chem. 287, 38210–38219 (2012).
Mandal, P. K. et al. Loss of thioredoxin reductase 1 renders tumors highly susceptible to pharmacologic glutathione deprivation. Cancer Res. 70, 9505–9514 (2010).
Ma, T. et al. Diselenides as novel effective fluorescence quenchers to construct a two-photon fluorescent probe for thiols in a mouse stroke model. Chem. Commun. 61, 1910–1913 (2025).
Stafford, W. C. et al. Irreversible inhibition of cytosolic thioredoxin reductase 1 as a mechanistic basis for anticancer therapy. Sci. Transl. Med. 10, eaaf7444 (2018).
Xue, Y. et al. Stimulus-cleavable chemistry in the field of controlled drug delivery. Chem. Soc. Rev. 50, 4872–4931 (2021).
Deng, Z., Hu, J. & Liu, S. Disulfide-based self-Immolative linkers and functional bioconjugates for biological applications. Macromol. Rapid Comm. 41, e1900531 (2020).
Zhang, P., Wu, J., Xiao, F., Zhao, D. & Luan, Y. Disulfide bond based polymeric drug carriers for cancer chemotherapy and relevant redox environments in mammals. Med. Res. Rev. 38, 1485–1510 (2018).
Zhao, J. et al. Glutathione-triggered prodrugs: design strategies, potential applications, and perspectives. Med. Res. Rev. 44, 1013–1054 (2023).
Zhang, C. et al. Design and synthesis of a ratiometric photoacoustic imaging probe activated by selenol for visual monitoring of pathological progression of autoimmune hepatitis. Chem. Sci. 12, 4883–4888 (2021).
Moser, P. et al. Fast release of carboxylic acid inside cells. ChemMedChem, 20, e202500056 (2025).
Wang, Q., Guan, J., Wan, J. & Li, Z. Disulfide based prodrugs for cancer therapy. RSC Adv. 10, 24397–24409 (2020).
Turell, L., Botti, H., Carballal, S., Radi, R. & Alvarez, B. Sulfenic acid—A key intermediate in albumin thiol oxidation. J. Chromatogr. B 877, 3384–3392 (2009).
Jia, H. et al. Fluorophore-dependent cleavage of disulfide bond leading to a highly selective fluorescent probe of thioredoxin. Anal. Chem. 91, 8524–8531 (2019).
Lee, M. H. et al. Toward a chemical marker for inflammatory disease: a fluorescent probe for membrane-localized thioredoxin. J. Am. Chem. Soc. 136, 8430–8437 (2014).
Wang, Y., Zhang, L. & Chen, L. Glutathione peroxidase-activatable two-photon ratiometric fluorescent probe for redox mechanism research in aging and mercury exposure mice models. Anal. Chem. 92, 1997–2004 (2020).
Zuo, X. et al. A fluorescent probe for specifically measuring the overall thioredoxin and glutaredoxin reducing activity in bacterial cells. Analyst 147, 834–840 (2022).
Zhao, Y. et al. A fluorescent probe to detect quick disulfide reductase activity in bacteria. Antioxidants 11, 377 (2022).
Lü, X. et al. Construction and identification of a HEK293 cell line with stable TrxR1 overexpression. J. South Med. Univ. 42, 554–560 (2022).
Petukhova, V. Z. et al. Non-covalent inhibitors of thioredoxin glutathione reductase with schistosomicidal activity in vivo. Nat. Commun. 14, 3737 (2023).
Zhong, Z. et al. NFATc1-mediated expression of SLC7A11 drives sensitivity to TXNRD1 inhibitors in osteoclast precursors. Redox Biol. 63, 102711 (2023).
Engelman, R., Ziv, T., Arnér, E. S. J. & Benhar, M. Inhibitory nitrosylation of mammalian thioredoxin reductase 1: molecular characterization and evidence for its functional role in cellular nitroso-redox imbalance. Free Radic. Biol. Med. 97, 375–385 (2016).
Zhou, J. et al. Discovery of novel hydroxyamidine based indoleamine 2,3-dioxygenase 1 (IDO1) and thioredoxin reductase 1 (TrxR1) dual inhibitors. Eur. J. Med. Chem. 245, 114860 (2023).
Flowers, B. et al. Pleiotropic anti-cancer activities of novel non-covalent thioredoxin reductase inhibitors against triple negative breast cancer. Free Radic. Biol. Med. 227, 201–209 (2025).
Fan, Q.-Z. et al. Design, synthesis, and biological evaluation of a novel indoleamine 2,3-dioxigenase 1 (IDO1) and thioredoxin reductase (TrxR) dual inhibitor. Bioorg. Chem. 105, 104401 (2020).
Zhong, M. et al. Synthesis and evaluation of Piperine analogs as thioredoxin reductase inhibitors to cause oxidative stress-induced cancer cell apoptosis. Bioorg. Chem. 138, 106589 (2023).
Zheng, Y.-L. et al. Redox-based strategy for selectively inducing energy crisis inside cancer cells: an example of modifying dietary curcumin to target mitochondria. J. Agr. Food Chem. 70, 2898–2910 (2022).
Wang, J. et al. Targeting thioredoxin reductase 1 by Andrographolide contributes to inducing ROS-mediated apoptosis in human NSCLC cells. Chem.-Biol. Interact. 421, 111778 (2025).
Wang, R. et al. Pretargeted mitochondrial delivery of organoarsenicals for cancer immunotherapy. J. Am. Chem. Soc. 147, 38534–38548 (2025).
Rullo, A. et al. Unravelling the anti-cancer mechanisms elicited by non-covalent thioredoxin reductase inhibitors for triple negative breast cancer therapy. Redox Biol. 90, 103980 (2026).
Zhong, Z. et al. Beyond thiol-enzyme inhibition: sterically bulky NHC-Au(I) complexes are catalytically active anticancer agents with reprogrammed immunomodulatory function. J. Am. Chem. Soc. 148, 8511–8523 (2026).
Zhang, L., Zhao, J., Yi, M., Osama, A. & Fang, J. Structural optimization of dithiaarsanes-enhanced cytotoxicity to leukemia cells with improved thioredoxin reductase inhibition. Chem. Asian J. 21, e00977 (2026).
Seim, G. L. et al. Nitric oxide-driven modifications of lipoic arm inhibit α-ketoacid dehydrogenases. Nat. Chem. Biol. 19, 265–274 (2022).
Gu, F. et al. Tumour microenvironment-responsive lipoic acid nanoparticles for targeted delivery of docetaxel to lung cancer. Sci. Rep. 6, 36281 (2016).
Lv, S.-Y. et al. Review of lipoic acid: from a clinical therapeutic agent to various emerging biomaterials. Int. J. Pharm. 627, 122201 (2022).
Kang, D. H., Kim, J., Lee, J. & Kang, S. W. The small molecule peroxiredoxin mimetics restore growth factor signalings and reverse vascular remodeling. Free Radic. Biol. Med. 229, 300–311 (2025).
Kang, D. H. et al. Vascular injury involves the overoxidation of peroxiredoxin type II and is recovered by the peroxiredoxin activity mimetic that induces reendothelialization. Circulation 128, 834–844 (2013).
Zeisel, L. et al. Piperazine-fused cyclic disulfides unlock high-performance bioreductive probes of thioredoxins and bifunctional reagents for thiol redox biology. J. Am. Chem. Soc. 146, 5204–5214 (2024).
Felber, J. G. et al. Selective, modular probes for thioredoxins enabled by rational tuning of a unique disulfide structure motif. J. Am. Chem. Soc. 143, 8791–8803 (2021).
Eberhardt, J., Santos-Martins, D., Tillack, A. F. & Forli, S. AutoDock Vina 1.2. 0: new docking methods, expanded force field, and Python bindings. J. Chem. Inf. Model. 61, 3891–3898 (2021).
Trott, O. & Olson, A. J. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31, 455–461 (2010).
Acknowledgements
This research was supported by the China Postdoctoral Science Foundation under Grant Number 2025M783554 (J.Z.).
Author information
Authors and Affiliations
Contributions
J.Z. conceived and led the study, designed and coordinated the overall experimental strategy, synthesized all compounds, performed all spectroscopic and HPLC analyses, processed and interpreted the data, wrote the original draft of the manuscript, and contributed funding support for the research. H.L. performed the TrxR1-knockout–related cellular imaging, cytotoxicity assays, and associated experiments (Figs. 7D and 9 and Supplementary Figs. 11 and 16–18). T.L. conducted the computational docking studies (Supplementary Figs. 6 and 12). J.W. conducted the cytotoxicity assays shown in Supplementary Fig. 9. B.C. provided prodrug Se1-DOX, and T.M. provided probe AFC-SS. M.Y. performed the cellular imaging experiments in Supplementary Fig. 15. S.-H.W. contributed computational docking support. X.L. analyzed the data and contributed to discussion. B.Z. provided instrumentation support and reviewed key data. C.Z. provided the HCT116 cell lines (WT, NC, and TrxR1-KO) and TrxR, and supervised the experimental design, execution, and data analysis of all HCT116-related studies, including the TRi-1 inhibition experiments. J.F. contributed to project conception, supervised the research, provided input on data interpretation, performed manuscript writing and submission as well as revision, and offered funding and instrumentation support. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Lin Yuan and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, J., Liu, H., Liu, T. et al. Insights into the activation patterns of 1,2-dithiolane unit in biofunctional molecules. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70678-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70678-8