Abstract
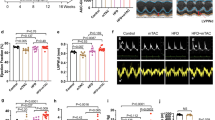
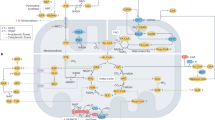
Increased protein acetylation is frequently observed in the failing heart, including in hearts with heart failure with preserved ejection fraction (HFpEF). However, its role in the pathogenesis of HFpEF remains insufficiently investigated. Here, we found that HFpEF hearts displayed significantly protein hyperacetylation, which were predominantly localized to mitochondria and particularly enriched in fatty acid oxidation (FAO) pathway. Notably, Dlat, a pyruvate metabolism enzyme, was identified as the key transacetylase for mitochondrial protein hyperacetylation. Dlat overexpression enhanced FAO-related protein acetylation and exacerbated cardiac lipid metabolism disturbances, whereas Dlat knockdown effectively mitigated FAO inhibition and HFpEF phenotypes. Moreover, we demonstrated that Dlat directly triggers the acetylation of alpha subunit of mitochondrial trifunctional protein (HADHA) at the K728 site, thereby inactivating HADHA enzymatic activity. Our study provides a mechanistic basis linking protein hyperacetylation, FAO inhibition, and HFpEF development. Manipulating mitochondrial protein acetylation may offer potential strategies for therapeutic intervention of HFpEF.
Similar content being viewed by others
Data availability
There are no restrictions on data availability. All data supporting the findings of this study are available within the main text, supplementary information, and Source data. Further information and requests for resources and reagents should be directed to and will be fulfilled by the lead contact. Lipidomics data have been deposited in the Metabolights under the accession number MTBLS12919. Proteomics data and acetylation proteomics data have been deposited in the ProteomeXchange under the accession number PXD068270. Source data are provided with this paper.
References
Dunlay, S. M., Roger, V. L. & Redfield, M. M. Epidemiology of heart failure with preserved ejection fraction. Nat. Rev. Cardiol. 14, 591–602 (2017).
Borlaug, B. A. Evaluation and management of heart failure with preserved ejection fraction. Nat. Rev. Cardiol. 17, 559–573 (2020).
Borlaug, B. A. et al. Obesity and heart failure with preserved ejection fraction: new insights and pathophysiological targets. Cardiovasc. Res. 118, 3434–3450 (2023).
Cohen, J. B. et al. Clinical phenogroups in heart failure with preserved ejection fraction: detailed phenotypes, prognosis, and response to spironolactone. JACC Heart Fail 8, 172–184 (2020).
Phan, T. T. et al. Heart failure with preserved ejection fraction is characterized by dynamic impairment of active relaxation and contraction of the left ventricle on exercise and associated with myocardial energy deficiency. J. Am. Coll. Cardiol. 54, 402–409 (2009).
Nassif, M. E. et al. The SGLT2 inhibitor dapagliflozin in heart failure with preserved ejection fraction: a multicenter randomized trial. Nat. Med. 27, 1954–1960 (2021).
Tatyana, A. S., Anna, A. A. & Wilbert, S. A. Insufficient evidence regarding benefits from sodium-glucose cotransporter-2 inhibitors in heart failure with preserved ejection fraction. Vessel 4, 35 (2020).
Kato, T. et al. Analysis of metabolic remodeling in compensated left ventricular hypertrophy and heart failure. Circ. Heart Fail 3, 420–430 (2010).
Conway, M. A. et al. Detection of low phosphocreatine to ATP ratio in failing hypertrophied human myocardium by 31P magnetic resonance spectroscopy. Lancet 338, 973–976 (1991).
Bertero, E. & Maack, C. Metabolic remodelling in heart failure. Nat. Rev. Cardiol. 15, 457–470 (2018).
Mori, J. et al. Agonist-induced hypertrophy and diastolic dysfunction are associated with selective reduction in glucose oxidation: a metabolic contribution to heart failure with normal ejection fraction. Circ. Heart Fail 5, 493–503 (2012).
Kolwicz, S. C. Jr., Purohit, S. & Tian, R. Cardiac metabolism and its interactions with contraction, growth, and survival of cardiomyocytes. Circ. Res. 113, 603–616 (2013).
Hahn, V. S. et al. Myocardial metabolomics of human heart failure with preserved ejection fraction. Circulation 147, 1147–1161 (2023).
Yoshii, A. et al. Blunted cardiac mitophagy in response to metabolic stress contributes to HFpEF. Circ. Res. 135, 1004–1017 (2024).
Shvedunova, M. & Akhtar, A. Modulation of cellular processes by histone and non-histone protein acetylation. Nat. Rev. Mol. Cell Biol. 23, 329–349 (2022).
Williams, A. S. et al. Disruption of acetyl-lysine turnover in muscle mitochondria promotes insulin resistance and redox stress without overt respiratory dysfunction. Cell Metab. 31, 131–147 (2020).
Alrob, O. A. et al. Obesity-induced lysine acetylation increases cardiac fatty acid oxidation and impairs insulin signalling. Cardiovasc. Res. 103, 485–497 (2014).
Dikalov, S. I. & Dikalova, A. E. Crosstalk between mitochondrial hyperacetylation and oxidative stress in vascular dysfunction and hypertension. Antioxid. Redox Signal 31, 710–721 (2019).
Li, P., Ge, J. & Li, H. Lysine acetyltransferases and lysine deacetylases as targets for cardiovascular disease. Nat. Rev. Cardiol. 17, 96–115 (2020).
Zhang, X., Wang, Y., Li, H., Wang, D. W. & Chen, C. Insights into the post-translational modifications in heart failure. Ageing Res. Rev. 100, 102467 (2024).
Tong, D. et al. NAD(+) repletion reverses heart failure with preserved ejection fraction. Circ. Res. 128, 1629–1641 (2021).
Schiattarella, G. G. et al. Nitrosative stress drives heart failure with preserved ejection fraction. Nature 568, 351–356 (2019).
Wang, N. et al. Pyruvate metabolism enzyme DLAT promotes tumorigenesis by suppressing leucine catabolism. Cell Metab. 37, 1381–1399 (2025.
Shan, C. et al. Lysine acetylation activates 6-phosphogluconate dehydrogenase to promote tumor growth. Mol. Cell 55, 552–565 (2014).
Li, Y. et al. Recent advances in pyruvate dehydrogenase kinase inhibitors: structures, inhibitory mechanisms and biological activities. Bioorg. Chem. 144, 107160 (2024).
Chi, Z. et al. Histone deacetylase 3 couples mitochondria to Drive IL-1β-dependent inflammation by configuring fatty acid oxidation. Mol. Cell 80, 43–58 (2020).
Thapa, D. et al. The protein acetylase GCN5L1 modulates hepatic fatty acid oxidation activity via acetylation of the mitochondrial β-oxidation enzyme HADHA. J. Biol. Chem. 293, 17676–17684 (2018).
Al-Habsi, M. et al. Spermidine activates mitochondrial trifunctional protein and improves antitumor immunity in mice. Science 378, eabj3510 (2022).
Roh, J., Hill, J. A., Singh, A., Valero-Muñoz, M. & Sam, F. Heart failure with preserved ejection fraction: heterogeneous syndrome, diverse preclinical models. Circ. Res. 130, 1906–1925 (2022).
Lopaschuk, G. D., Karwi, Q. G., Tian, R., Wende, A. R. & Abel, E. D. Cardiac energy metabolism in heart failure. Circ. Res. 128, 1487–1513 (2021).
Watson, W. D. et al. Retained metabolic flexibility of the failing human heart. Circulation 148, 109–123 (2023).
Ritterhoff, J. & Tian, R. Metabolic mechanisms in physiological and pathological cardiac hypertrophy: new paradigms and challenges. Nat. Rev. Cardiol. 20, 812–829 (2023).
Hu, L. et al. Mfn2/Hsc70 complex mediates the formation of mitochondria-lipid droplets membrane contact and regulates myocardial lipid metabolism. Adv. Sci. 11, e2307749 (2024).
Geng, J. et al. Moderate-intensity interval exercise exacerbates cardiac lipotoxicity in high-fat, high-calories diet-fed mice. Nat. Commun. 16, 613 (2025).
Liu, X. et al. Mitochondrial protein hyperacetylation underpins heart failure with preserved ejection fraction in mice. J. Mol. Cell Cardiol. 165, 76–85 (2022).
Narita, T., Weinert, B. T. & Choudhary, C. Functions and mechanisms of non-histone protein acetylation. Nat. Rev. Mol. Cell Biol. 20, 156–174 (2019).
Kane, A. E. & Sinclair, D. A. Sirtuins and NAD(+) in the development and treatment of metabolic and cardiovascular diseases. Circ. Res. 123, 868–885 (2018).
Davidson, M. T. et al. Extreme acetylation of the cardiac mitochondrial proteome does not promote heart failure. Circ. Res. 127, 1094–1108 (2020).
Hu, Q. et al. Increased Drp1 acetylation by lipid overload induces cardiomyocyte death and heart dysfunction. Circ. Res. 126, 456–470 (2020).
Rahmatullah, M., Radke, G. A., Andrews, P. C. & Roche, T. E. Changes in the core of the mammalian-pyruvate dehydrogenase complex upon selective removal of the lipoyl domain from the transacetylase component but not from the protein X component. J. Biol. Chem. 265, 14512–14517 (1990).
Patel, M. S., Korotchkina, L. G. & Sidhu, S. Interaction of E1 and E3 components with the core proteins of the human pyruvate dehydrogenase complex. J. Mol. Catal. B Enzym 61, 2–6 (2009).
Hiromasa, Y., Fujisawa, T., Aso, Y. & Roche, T. E. Organization of the cores of the mammalian pyruvate dehydrogenase complex formed by E2 and E2 plus the E3-binding protein and their capacities to bind the E1 and E3 components. J. Biol. Chem. 279, 6921–6933 (2004).
Jaswal, J. S., Keung, W., Wang, W., Ussher, J. R. & Lopaschuk, G. D. Targeting fatty acid and carbohydrate oxidation–a novel therapeutic intervention in the ischemic and failing heart. Biochim. Biophys. Acta 1813, 1333–1350 (2011).
Hirschey, M. D. et al. SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 464, 121–125 (2010).
Koves, T. R. et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 7, 45–56 (2008).
Liu, L. et al. DGAT1 expression increases heart triglyceride content but ameliorates lipotoxicity. J. Biol. Chem. 284, 36312–36323 (2009).
Laaksonen, R. et al. Plasma ceramides predict cardiovascular death in patients with stable coronary artery disease and acute coronary syndromes beyond LDL-cholesterol. Eur. Heart J. 37, 1967–1976 (2016).
Eisenberg, T. et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 22, 1428–1438 (2016).
Hu, L. et al. Nicotinamide riboside promotes Mfn2-mediated mitochondrial fusion in diabetic hearts through the SIRT1-PGC1α-PPARα pathway. Free Radic. Biol. Med. 183, 75–88 (2022).
Acknowledgements
We appreciate all technicians in Innovation Center of Military Medicine of AFMU for their excellent technical assistance. This work was supported by grants from the National Natural Science Foundation of China: No. 82300443 (Lang Hu), No. 82400450 (Ying Wang) and No. 82470388 (Yan Li) and the Key Industrial Chain Projects of Shaanxi Province (Yan Li, 2024SF-ZDCYL-01-03).
Author information
Authors and Affiliations
Contributions
L.H., Y.L., and Y.W. conceptualized the study and designed the research. Y.W., D.G., J.Z., X.Y., C.W., J.G., Q.L., and P.C. performed the experiments. Y.W., D.G., J.Z., N.S., X.N., Y.L., Y.G., and L.H. contributed to the data analysis. Y.W., L.H., and D.G. drafted the article. L.H. and Y.L. revised and edited the article. All authors have read and approve the final article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Tao Li, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Y., Guo, D., Zhu, J. et al. Pyruvate metabolism enzyme Dlat induces mitochondria protein hyperacetylation to limit fatty acid oxidation in the HFpEF heart. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70703-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70703-w