Abstract
Diabetic wounds have limited effective therapies to restore tissue repair and resolve excessive inflammation. This study aimed to identify mechanisms of diabetic wound healing defects and test a therapeutic intervention using diabetic mouse and pig models. Here, we show that Smad7 transgene expression in mouse epidermis promotes wound healing in diabetic mice. To restrict the therapeutic effects of Smad7 on wounds, we develop a Smad7-based biologic (Tat-PYC-Smad7) that penetrates cells of the wound. Topical Tat-PYC-Smad7 treatment to diabetic pig and mouse wounds accelerates healing. Tat-PYC-Smad7-treated wounds exhibit reduced TGFβ/NFκB signaling, faster re-epithelialization, and better extracellular matrix remodeling compared to vehicle controls. Tat-PYC-Smad7 also attenuates neutrophil extracellular trap (NET) formation, potentially acting through reductions in MPO enzymatic activity and MPO nuclear entry, consequently reducing chromatin decondensation and the release of NET components. Our study reveals that keratinocytes and neutrophils are the two major cell types targeted by Tat-PYC-Smad7 to promote diabetic wound healing, providing insight into mechanisms of diabetic wound healing defects targetable by Smad7-based therapy.
Similar content being viewed by others
Data availability
RNA-seq data generated in this study are available in the Gene Expression Omnibus (under accession code GSE274513) [https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE274513]. Mass spectrometry data generated in this study are available in MassIVE under accession code (MSV000095545)[https://massive.ucsd.edu/ProteoSAFe/dataset.jsp?task=d2212a53e36d40ad9b872f22d98814b4]. Source data are provided with this paper.
Code availability
A customized Python script for semi-automatic wound area segmentation has been deposited and is publicly accessible at https://zenodo.org/records/18341887. For RNAseq analysis, Quality control was conducted by FastQC (https://www.bioinformatics.babraham.ac.uk/projects/fastqc/) and FastQ Screen (https://www.bioinformatics.babraham.ac.uk/projects/fastq_screen/). The RNA-seq reads were processed with BBDuk (BBMap – Bushnell B. – sourceforge.net/projects/bbmap/) and aligned to mouse genome GRCm38.p6 (release 96) with STAR [https://www.ensembl.org/Mus_musculus/Info/Index]57. The differential expression analysis was performed on TMM (trimmed-mean M values) normalized count data using a negative binomial generalized linear model and likelihood ratio test58,59 implemented in the edgeR package (version 4.0.11, https://doi.org/10.5281/zenodo.3748085)60. The Benjamini–Hochberg multiplicity correction was applied on p-values to calculate false discovery rates. GSEA was performed using pre-ranked GSEA61 and Enrichr62 implemented by the GSEApy package (version 1.1.2, https://doi.org/10.5281/zenodo.3748085), incorporating pathway collections from the Molecular Signatures Database (MSigDB) [https://www.gsea-msigdb.org/gsea/msigdb], including Hallmark, KEGG, and Gene Ontology biological process gene sets.
References
Falanga, V. Wound healing and its impairment in the diabetic foot. Lancet 366, 1736–1743 (2005).
Armstrong, D. G., Boulton, A. J. M. & Bus, S. A. Diabetic Foot Ulcers and Their Recurrence. N. Engl. J. Med. 376, 2367–2375 (2017).
Cho, H., Blatchley, M. R., Duh, E. J. & Gerecht, S. Acellular and cellular approaches to improve diabetic wound healing. Adv. Drug Deliv. Rev. 146, 267–288 (2019).
Nagai, M. K. & Embil, J. M. Becaplermin: recombinant platelet derived growth factor, a new treatment for healing diabetic foot ulcers. Expert Opin. Biol. Ther. 2, 211–218 (2002).
Cavanagh, P. R., Lipsky, B. A., Bradbury, A. W. & Botek, G. Treatment for diabetic foot ulcers. Lancet 366, 1725–1735 (2005).
Barrientos, S., Brem, H., Stojadinovic, O. & Tomic-Canic, M. Clinical application of growth factors and cytokines in wound healing. Wound Repair Regen. 22, 569–578 (2014).
Zhao, R., Liang, H., Clarke, E., Jackson, C. & Xue, M. Inflammation in Chronic Wounds. Int. J. Mol. Sci. 17 (2016).
Rodrigues, M., Kosaric, N., Bonham, C. A. & Gurtner, G. C. Wound Healing: A Cellular Perspective. Physiol. Rev. 99, 665–706 (2019).
Xiao, Y. et al. Diabetic wound regeneration using peptide-modified hydrogels to target re-epithelialization. Proc. Natl. Acad. Sci. USA 113, E5792–e5801 (2016).
Stojadinovic, O. et al. Deregulation of keratinocyte differentiation and activation: a hallmark of venous ulcers. J. Cell Mol. Med 12, 2675–2690 (2008).
Lan, C. C., Wu, C. S., Kuo, H. Y., Huang, S. M. & Chen, G. S. Hyperglycaemic conditions hamper keratinocyte locomotion via sequential inhibition of distinct pathways: new insights on poor wound closure in patients with diabetes. Br. J. Dermatol. 160, 1206–1214 (2009).
Wilgus, T. A., Roy, S. & McDaniel, J. C. Neutrophils and Wound Repair: Positive Actions and Negative Reactions. Adv. Wound Care 2, 379–388 (2013).
Raziyeva, K. et al. Immunology of Acute and Chronic Wound Healing. Biomolecules 11, 700 (2021).
Eming, S. A., Krieg, T. & Davidson, J. M. Inflammation in Wound Repair: Molecular and Cellular Mechanisms. J. Investig. Dermatol. 127, 514–525 (2007).
Wong, S. L. et al. Diabetes primes neutrophils to undergo NETosis, which impairs wound healing. Nat. Med. 21, 815–819 (2015).
Lekstrom-Himes, J. A. & Gallin, J. I. Immunodeficiency diseases caused by defects in phagocytes. N. Engl. J. Med. 343, 1703–1714 (2000).
Hawkes, J. E., Adalsteinsson, J. A., Gudjonsson, J. E. & Ward, N. L. Research Techniques Made Simple: Murine Models of Human Psoriasis. J. Invest. Dermatol. 138, e1–e8 (2018).
Michaels, J. et al. db/db mice exhibit severe wound-healing impairments compared with other murine diabetic strains in a silicone-splinted excisional wound model. Wound Repair Regen. 15, 665–670 (2007).
Sullivan, T. P., Eaglstein, W. H., Davis, S. C. & Mertz, P. The pig as a model for human wound healing. Wound Repair Regen. 9, 66–76 (2001).
Wang, X. J., Han, G., Owens, P., Siddiqui, Y. & Li, A. G. Role of TGF beta-mediated inflammation in cutaneous wound healing. J. Investig. Dermatol Symp. Proc. 11, 112–117 (2006).
Li, F. et al. Smad7 Ameliorates TGF-beta-mediated skin inflammation and associated wound healing defects but not susceptibility to experimental skin Carcinogenesis. J. Invest Dermatol 139, 940–950 (2019).
Nakao, A. et al. Identification of Smad7, a TGFbeta-inducible antagonist of TGF-beta signalling. Nature 389, 631–635 (1997).
Massagué, J. TGF-beta signal transduction. Annu. Rev. Biochem. 67, 753–791 (1998).
He, W. et al. Overexpression of Smad7 results in severe pathological alterations in multiple epithelial tissues. EMBO J. 21, 2580–2590 (2002).
Coleman, D. L. Obese and diabetes: two mutant genes causing diabetes-obesity syndromes in mice. Diabetologia 14, 141–148 (1978).
Han, G. et al. Smad7-Induced β-Catenin Degradation Alters Epidermal Appendage Development. Dev. Cell 11, 301–312 (2006).
Thomas, A. C., Eijgelaar, W. J., Daemen, M. J. & Newby, A. C. Foam cell formation in vivo converts macrophages to a pro-fibrotic phenotype. PLoS One 10, e0128163 (2015).
McDermott, K., Fang, M., Boulton, A. J. M., Selvin, E. & Hicks, C. W. Etiology, Epidemiology, and Disparities in the Burden of Diabetic Foot Ulcers. Diab. Care 46, 209–221 (2022).
Han, G., Li, F., Ten Dijke, P. & Wang, X. J. Temporal Smad7 transgene induction in mouse epidermis accelerates skin wound healing. Am. J. Pathol. 179, 1768–1779 (2011).
Lei, Q. et al. A2AR-mediated CXCL5 upregulation on macrophages promotes NSCLC progression via NETosis. Cancer Immunol. Immunother. 73, 108 (2024).
Kinoshita, M. et al. Neutrophils initiate and exacerbate Stevens-Johnson syndrome and toxic epidermal necrolysis. Sci. Transl. Med. 13, eaax2398 (2021).
Yin, C. et al. IL-33/ST2 induces neutrophil-dependent reactive oxygen species production and mediates gout pain. Theranostics 10, 12189–12203 (2020).
Aratani, Y. Myeloperoxidase: Its role for host defense, inflammation, and neutrophil function. Arch. Biochem Biophys. 640, 47–52 (2018).
Zhang, H. et al. Molecular insight into pentraxin-3: Update advances in innate immunity, inflammation, tissue remodeling, diseases, and drug role. Biomed. Pharmacother. 156, 113783 (2022).
Lerman, I. & Hammes, S. R. Neutrophil elastase in the tumor microenvironment. Steroids 133, 96–101 (2018).
Ou, Q. et al. TcpC inhibits neutrophil extracellular trap formation by enhancing ubiquitination mediated degradation of peptidylarginine deiminase 4. Nat. Commun. 12, 3481 (2021).
Thiam, H. R. et al. NETosis proceeds by cytoskeleton and endomembrane disassembly and PAD4-mediated chromatin decondensation and nuclear envelope rupture. Proc. Natl. Acad. Sci. 117, 7326 (2020).
Thiam, H. R., Wong, S. L., Wagner, D. D. & Waterman, C. M. Cellular Mechanisms of NETosis. Annu Rev. Cell Dev. Biol. 36, 191–218 (2020).
Dufrane, D. et al. Streptozotocin-induced diabetes in large animals (pigs/primates): role of GLUT2 transporter and beta-cell plasticity. Transplantation 81, 36–45 (2006).
Metzler, K. D. et al. Myeloperoxidase is required for neutrophil extracellular trap formation: implications for innate immunity. Blood 117, 953–959 (2011).
Cibrian, D., de la Fuente, H. & Sánchez-Madrid, F. Metabolic Pathways That Control Skin Homeostasis and Inflammation. Trends Mol. Med 26, 975–986 (2020).
Li, F. et al. Smad7 Ameliorates TGF-β-mediated skin inflammation and associated wound healing defects but not susceptibility to experimental skin Carcinogenesis. J. Invest. Dermatol. 139, 940–950 (2019).
Han, G. et al. Preventive and therapeutic effects of Smad7 on radiation-induced oral mucositis. Nat. Med. 19, 421–428 (2013.
Boss, M.-K. et al. Therapeutic Intervention Using a Smad7-Based Tat Protein to Treat Radiation-Induced Oral Mucositis. Int. J. Radiat. Oncol.*Biol.*Phys. 112, 759–770 (2021).
Osuka, A., Ogura, H., Ueyama, M., Shimazu, T. & Lederer, J. A. Immune response to traumatic injury: harmony and discordance of immune system homeostasis. Acute Med Surg. 1, 63–69 (2014).
Saharinen, P., Eklund, L. & Alitalo, K. Therapeutic targeting of the angiopoietin–TIE pathway. Nat. Rev. Drug Discov. 16, 635–661 (2017).
Zhang, Y., Ma, K. L., Ruan, X. Z. & Liu, B. C. Dysregulation of the Low-Density Lipoprotein Receptor Pathway Is Involved in Lipid Disorder-Mediated Organ Injury. Int J. Biol. Sci. 12, 569–579 (2016).
Korbecki, J. et al. The Role of CXCL16 in the pathogenesis of cancer and other diseases. Int J. Mol. Sci. 22, 3490 (2021).
Biernacka, A., Dobaczewski, M. & Frangogiannis, N. G. TGF-β signaling in fibrosis. Growth Factors 29, 196–202 (2011).
Luo, J. et al. Smad7 Promotes Healing of Radiotherapy-Induced Oral Mucositis without Compromising Oral Cancer Therapy in a Xenograft Mouse Model. Clin. Cancer Res. 25, 808–818 (2019).
Metzler, K. D., Goosmann, C., Lubojemska, A., Zychlinsky, A. & Papayannopoulos, V. A myeloperoxidase-containing complex regulates neutrophil elastase release and actin dynamics during NETosis. Cell Rep. 8, 883–896 (2014).
Papayannopoulos, V., Metzler, K. D., Hakkim, A. & Zychlinsky, A. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J. Cell Biol. 191, 677–691 (2010).
de Oliveira, S., Rosowski, E. E. & Huttenlocher, A. Neutrophil migration in infection and wound repair: going forward in reverse. Nat. Rev. Immunol. 16, 378–391 (2016).
Yang, C. T. et al. Hydrogen sulfide primes diabetic wound to close through inhibition of NETosis. Mol. Cell Endocrinol. 480, 74–82 (2019).
Peng, B. Y. et al. A novel and quick PCR-based method to genotype mice with a leptin receptor mutation (db/db mice). Acta Pharm. Sin. 39, 117–123 (2018).
Luo, J. et al. Smad7 Promotes Healing of Radiotherapy-Induced Oral Mucositis without Compromising Oral Cancer Therapy in a Xenograft Mouse Model. Clin. Cancer Res 25, 808–818 (2019).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
McCarthy, D. J., Chen, Y. & Smyth, G. K. Differential expression analysis of multifactor RNA-Seq experiments with respect to biological variation. Nucleic Acids Res 40, 4288–4297 (2012).
Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol. 11, R25 (2010).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Chen, E. Y. et al. Enrichr: interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinforma. 14, 128 (2013).
Li, A. G., Lu, S. L., Zhang, M. X., Deng, C. & Wang, X. J. Smad3 knockout mice exhibit a resistance to skin chemical carcinogenesis. Cancer Res. 64, 7836–7845 (2004).
Acknowledgements
This work was funded by National Institutes of Health grants DE024659, DE028718, and AR078669 to X.J.W. and C.D.Y, and R01GM148769 to S.R.C. Y.K. received funding through the Milstein Medical Asian American Partnership Foundation Awards in Dermatology and a Sequencing Plot Award from the Genomics and Microarray Core at the University of Colorado Denver. We thank Dr. David Orlicky for pathology support. We thank Sinclair Research Center and Sinclair BioResources for the establishment of diabetic pig models and treatment tests. We thank Ivan Lu for his assistance with HL60 cell culture, Yi Wang for her assistance with organizing the gross images of the wounds, Qian Chen with cell pellet embedding, and Dr. Ingrid Brust-Mascher from the Advanced Imaging Facility at the UC Davis School of Veterinary Medicine for her expert support with confocal imaging. We extend our appreciation to several entities for their valuable contributions: the University of Colorado Cancer Center Cell Technologies Shared Resource for their assistance with cell culture and use of the IncuCyte, the Mass Spectrometry Proteomics Shared Resource Facility at the University of Colorado for their mass spectrometry proteomic analyses, the Gates Center for Regenerative Medicine at the University of Colorado’s Anschutz Medical Campus for their histological support, and the RNA Biosciences Initiative at the University of Colorado for their support with RNA sequencing. Additionally, we acknowledge the Biostatistics and Bioinformatics Shared Resource at the University of Colorado Cancer Center for their assistance with processing RNAseq data. These shared resources are supported by the University of Colorado Cancer Center Support Grant (P30CA046934). The study used the UC Davis Comprehensive Cancer Center Molecular Pharmacology and Chemical Biology Shared Resource, supported by the National Cancer Institute of the National Institutes of Health under award number P30CA093373.
Author information
Authors and Affiliations
Contributions
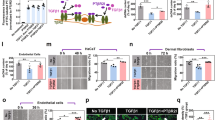
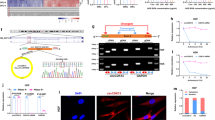
Y.K., C.D.Y., and X.J.W. conceived and designed the study. Y.K. performed the experiments, analyzed the data, and wrote the manuscript. B.Z.L. performed bioinformatics analyses and created all elements in Fig. 1h and the pig illustration in Fig. 2a. F.L. conducted a subset of the K5.Smad7/db/db mouse wound experiments and analyzed the data. R.K.R. performed ELISA and ADA assays and analyzed the data. D.W. and S.W. purified the Tat-PYC-Smad7 protein. S.R.C. provided expertise in neutrophil and NETosis functional studies and contributed to data interpretation. S.T.H., S.S., and S.R.C. assisted with manuscript editing. C.D.Y. and X.J.W. supervised the project and contributed to manuscript writing and editing.
Corresponding authors
Ethics declarations
Competing interests
Christian Young and Xiao-Jing Wang are inventors of a patent application filed to the US Patent and Trademark Office pertaining to the Tat-PYC-Smad7 drug product aspect(s) of this work (PCT/US2022/076242). Yao Ke and Xiao-Jing Wang are inventors of a patent application filed to the US Patent and Trademark Office pertaining to the Tat-PYC-Smad7 treatment to netosis-related indications (application # US63/792,252). The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Bing Tang, Irena Pastar, who co-reviewed with Raji Rao Nagalla, Shuofei Yang, Hawa Racine Thiam, and Jianliang Shen for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ke, Y., Li, BZ., Li, F. et al. Smad7-based biologic targeting epidermis and stroma promotes healing of diabetic wounds in mice and pigs. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70790-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70790-9