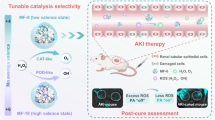
Abstract
Liquid metals (LMs) have been used for design of advanced materials by leveraging their unique liquid and electronic properties. Here, we design a series of liquid metal-buffered amorphous molybdenum sulfide nanozymes with different gallium indium ratios, in which Ga75.5In24.5 MoSX nanozymes exhibit the enzyme-like activity, superior to the crystalline MoS2. This has been verified and assigned to the roles of liquid metal: (i) serving as the building template, (ii) phase engineering with catalytic active sites, (iii) electron-rich microenvironment for improving catalytic activity. Besides, the amorphous Ga75.5In24.5 MoSX nanozyme possesses oxidase (OXD)-like and nicotinamide adenine dinucleotide oxidase (NOX)-like activity to achieve multiple-enzyme-like cascade catalytic reactions, disrupting intratumoral redox and metabolism homeostasis. As the example of leveraging liquid metal as a well-designed platform to screen high-performance nanozyme with excellent therapeutic effects, this work highlights the roles of tailored electronic structure and phase engineering for regulation of catalytic activity of nanozymes and may also broaden the application of liquid metal in catalysis and biomedical field.
Similar content being viewed by others
Data availability
The data that support the findings of this study are presented in the article and Supplementary Information. Source data are provided with this paper. All data underlying this study are available from the corresponding author upon request. Source data are provided in this paper.
References
Daeneke, T. et al. Liquid metals: fundamentals and applications in chemistry. Chem. Soc. Rev. 47, 4073–4111 (2018).
Lin, Y. et al. Attributes, fabrication, and applications of gallium-based liquid metal particles. Adv. Sci. 7, 2000192 (2020).
Kalantar-Zadeh, K. et al. Emergence of liquid metals in nanotechnology. ACS Nano 13, 7388–7395 (2019).
Kalantar-Zadeh, K. et al. Low melting temperature liquid metals and their impacts on physical cemistry. Acc. Mater. Res. 2, 577–580 (2021).
Okatenko, V. et al. The native oxide skin of liquid metal Ga nanoparticles prevents their rapid coalescence during electrocatalysis. J. Am. Chem. Soc. 144, 10053–10063 (2022).
Tang, J. et al. Liquid-metal-enabled mechanical-energy-induced CO(2) conversion. Adv. Mater. 34, e2105789 (2022).
Flores, N. et al. Polyphenol-mediated liquid metal composite architecture for solar thermoelectric generation. Adv. Mater. 36, e2308346 (2024).
Xiang, W. T. et al. Liquid-metal-based magnetic fluids. Nat. Rev. Mater. 9, 433–449 (2024).
Yu, Y. et al. Activation of Ga liquid catalyst with continuously exposed active sites for electrocatalytic C-N coupling. Angew. Chem., Int. Ed. 63, e202402236 (2024).
Upham, D. C. et al. Catalytic molten metals for the direct conversion of methane to hydrogen and separable carbon. Science 358, 917–921 (2017).
Zavabeti, A. et al. A liquid metal reaction environment for the room-temperature synthesis of atomically thin metal oxides. Science 358, 332–335 (2017).
Chen, S. et al. Liquid metal composites. Matter 2, 1446–1480 (2020).
Liu, Y. et al. Synthesis and application of core-shell liquid metal particles: a perspective of surface engineering. Mater. Horiz. 8, 56–77 (2021).
Zhao, S. et al. Liquid metals: a novel possibility of fabricating 2D metal oxides. Adv. Mater. 33, e2005544 (2021).
Idrus-Saidi, S. A. et al. Liquid metal synthesis solvents for metallic crystals. Science 378, 1118–1124 (2022).
Cao, G. et al. Liquid metal for high-entropy alloy nanoparticles synthesis. Nature 619, 73–77 (2023).
Li, W. et al. Ultrastrong MXene film induced by sequential bridging with liquid metal. Science 385, 62–68 (2024).
Liang, J. et al. High-entropy alloy array via liquid metal nanoreactor. Adv. Mater. 36, e2403865 (2024).
Zhang, Y. et al. Liquid metal nanocores initiated construction of smart DNA-polymer microgels with programmable and regulable functions and near-infrared light-driven locomotion. Angew. Chem., Int. Ed. 63, e202311678 (2024).
Wang, D. et al. Liquid metal combinatorics toward materials discovery. Adv. Mater. 35, e2303533 (2023).
Guo, S. et al. Tailoring heterostructure growth on liquid metal nanodroplets through interface engineering. J. Am. Chem. Soc. 146, 19800–19808 (2024).
Li, L. et al. Atomic engineering of two-dimensional materials via liquid metals. Chem. Soc. Rev. 53, 7158–7201 (2024).
Liu, H. et al. Solid–liquid phase transition induced electrocatalytic switching from hydrogen evolution to highly selective CO2 reduction. Nat. Catal. 4, 202–211 (2021).
Fatima, S. S. et al. Current state and future prospects of liquid metal catalysis. Nat. Catal. 6, 1131–1139 (2023).
Lambie, S. et al. Dynamic activation of Ga sites by Pt dopant in low-temperature liquid-metal catalysts. Angew. Chem., Int. Ed. 62, e202219009 (2023).
Moritz, M. et al. Supported catalytically active liquid metal solutions: liquid metal catalysis with ternary alloys, enhancing activity in propane dehydrogenation. ACS Catal 14, 6440–6450 (2024).
Rahim, M. A. et al. Low-temperature liquid platinum catalyst. Nat. Chem. 14, 935–941 (2022).
Taccardi, N. et al. Gallium-rich Pd-Ga phases as supported liquid metal catalysts. Nat. Chem. 9, 862–867 (2017).
Tang, J. et al. Dynamic configurations of metallic atoms in the liquid state for selective propylene synthesis. Nat. Nanotechnol. 19, 306–310 (2024).
Yu, H. et al. Laser-generated supranano liquid metal as efficient electron mediator in hybrid perovskite solar cells. Adv. Mater. 32, e2001571 (2020).
Yan, J. et al. Advances in liquid metals for biomedical applications. Chem. Soc. Rev. 47, 2518–2533 (2018).
Wang, L. et al. Emerging liquid metal biomaterials: from design to application. Adv. Mater. 34, e2201956 (2022).
Chechetka, S. A. et al. Light-driven liquid metal nanotransformers for biomedical theranostics. Nat. Commun. 8, 15432 (2017).
Zhang, L. et al. Liquid metal as bioinspired and unusual modulator in bioorthogonal catalysis for tumor inhibition therapy. Angew. Chem., Int. Ed. 62, e202218159 (2023).
Hu, J. J. et al. Photo-controlled liquid metal nanoparticle-enzyme for starvation/photothermal therapy of tumor by win-win cooperation. Biomaterials 217, 119303 (2019).
Zhu, P. et al. Inorganic nanoshell-stabilized liquid metal for targeted photonanomedicine in NIR-II biowindow. Nano Lett. 19, 2128–2137 (2019).
Li, Z. et al. Liquid metal swimming nanorobots. Acc. Mater. Res. 3, 122–132 (2021).
Lin, Z. et al. Bubble-propelled Janus gallium/zinc micromotors for the active treatment of bacterial infections. Angew. Chem. Int. Ed. 60, 8750–8754 (2021).
Xie, W. et al. Gallium-based liquid metal particles for therapeutics. Trends Biotechnol. 39, 624–640 (2021).
Zhang, Y. et al. Synthesis of liquid gallium@reduced graphene 0xide core-shell nanoparticles with enhanced photoacoustic and photothermal performance. J. Am. Chem. Soc. 144, 6779–6790 (2022).
Yan, J. et al. Activating tumor-selective liquid metal nanomedicine through galvanic replacement. Adv. Mater. 36, e2307817 (2024).
Yang, N. et al. A general in-situ reduction method to prepare core-shell liquid-metal/metal nanoparticles for photothermally enhanced catalytic cancer therapy. Biomaterials 277, 121125 (2021).
Lu, Y. et al. Transformable liquid-metal nanomedicine. Nat. Commun. 6, 10066 (2015).
Liang, M. & Yan, X. Nanozymes: from new concepts, mechanisms, and standards to applications. Acc. Chem. Res. 52, 2190–2200 (2019).
Zhang, Y. et al. Nanozymes for nanohealthcare. Nat. Rev. Methods Primers 4, 36 (2024).
Huang, Y. et al. Nanozymes: classification, catalytic mechanisms, activity regulation, and Applications. Chem. Rev. 119, 4357–4412 (2019).
Zhang, L. et al. Biosystem-inspired engineering of nanozymes for biomedical applications. Adv. Mater. 36, e2211147 (2024).
Ji, S. F. et al. Matching the kinetics of natural enzymes with a single-atom iron nanozyme. Nat. Catal. 4, 407–417 (2021).
Liu, C. et al. Intrinsic strain-mediated ultrathin ceria nanoantioxidant. J. Am. Chem. Soc. 145, 19086–19097 (2023).
Fan, H. et al. Surface ligand engineering ruthenium nanozyme superior to horseradish peroxidase for enhanced immunoassay. Adv. Mater. 36, e2300387 (2024).
Xu, B. et al. A bioinspired five-coordinated single-atom iron nanozyme for tumor catalytic therapy. Adv. Mater. 34, e2107088 (2022).
Jana, D. et al. A defect-engineered nanozyme for targeted NIR-II photothermal immunotherapy of cancer. Adv. Mater. 36, e2206401 (2024).
Dong, H. et al. Depletable peroxidase-like activity of Fe(3)O(4) nanozymes accompanied with separate migration of electrons and iron ions. Nat. Commun. 13, 5365 (2022).
Kim, K. et al. Geometric tuning of single-atom FeN(4) sites via edge-generation enhances multi-enzymatic properties. Adv. Mater. 35, e2207666 (2023).
Wang, L. et al. A molybdenum disulfide nanozyme with charge-enhanced activity for ultrasound-mediated cascade-catalytic tumor ferroptosis. Angew. Chem. Int. Ed. 62, e202217448 (2023).
Zeng, R. et al. Precise tuning of the D-band center of dual-atomic enzymes for catalytic therapy. J. Am. Chem. Soc. 146, 10023–10031 (2024).
Zhao, Q. et al. Rearranging spin electrons by axial-ligand-induced orbital splitting to regulate enzymatic activity of single-atom nanozyme with destructive d-π conjugation. J. Am. Chem. Soc. 146, 14875–14888 (2024).
Yun, Q. et al. Recent progress on phase engineering of nanomaterials. Chem. Rev. 123, 13489–13692 (2023).
Gu, J. et al. Phase engineering of nanostructural metallic materials: classification, structures, and applications. Chem. Rev. 124, 1247–1287 (2024).
Wu, G. et al. A general synthesis approach for amorphous noble metal nanosheets. Nat. Commun. 10, 4855 (2019).
Zhang, J. et al. Amorphous MnRuO(x) containing microcrystalline for enhanced acidic oxygen-evolution activity and stability. Angew. Chem. Int. Ed. 63, e202405641 (2024).
Wu, Q. et al. Engineering liquid metal-based nanozyme for enhancing microwave dynamic therapy in breast cancer PDX model. J. Nanobiotechnol. 21, 399 (2023).
Xu, X. et al. Galvanic replacement synthesis of VO(x)@EGaIn-PEG core-shell nanohybrids for peroxidase mimics. ACS Appl. Mater. Interfaces 16, 21975–21986 (2024).
Ghasemian, M. B. et al. Coating of gallium-based liquid metal particles with molybdenum oxide and oxysulfide for electronic band structure modulation. Nanoscale 15, 5891–5898 (2023).
Huang, H. L. et al. Liquid metal alloys enable efficient formate electrosynthesis. Adv. Funct. Mater. 34, 2408966 (2024).
Anderson, T. J. & Ansara, I. The Ga-In (Gallium-Indium) system. J. Phase Equilib. 12, 64–72 (1991).
Li, L. Y. et al. Valence-engineered catalysis-selectivity regulation of molybdenum oxide nanozyme for acute kidney injury therapy and post-cure assessment. Nat. Commun. 15, 8720 (2024).
Guo, X. et al. Charge self-regulation in 1T”‘-MoS2 structure with rich S vacancies for enhanced hydrogen evolution activity. Nat. Commun. 13, 5954 (2022).
Tran, P. D. et al. Coordination polymer structure and revisited hydrogen evolution catalytic mechanism for amorphous molybdenum sulfide. Nat. Mater. 15, 640–646 (2016).
Yu, S. & Kaviany, M. Electrical, thermal, and species transport properties of liquid eutectic Ga-In and Ga-In-Sn from first principles. J. Chem. Phys. 140, 064303 (2014).
Hu, X. et al. Biodegradation-mediated enzymatic activity-tunable molybdenum oxide nanourchins for tumor-specific cascade catalytic therapy. J. Am. Chem. Soc. 142, 1636–1644 (2020).
Xiao, W. & Loscalzo, J. Metabolic responses to reductive stress. Antioxid. Redox Signaling 32, 1330–1347 (2020).
Katsyuba, E. et al. NAD(+) homeostasis in health and disease. Nat. Metab. 2, 9–31 (2020).
Liu, Y. et al. Single-atom nanozyme with asymmetric electron distribution for tumor catalytic therapy by disrupting tumor redox and energy metabolism homeostasis. Adv. Mater. 35, 2208512 (2023).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (T2495262, X.Q., 22437006, X.Q., 22237006, J.R.).
Author information
Authors and Affiliations
Contributions
X.Q. and J.R. designed research; W.Z. and J.Z. performed research and analyzed data; W.Z. and X.Q. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, W., Zhu, J., Ren, J. et al. Screening and regulation of nanozyme activity via liquid metals coined electron rearrangement and phase engineering. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70795-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70795-4