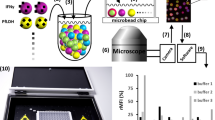
Abstract
There is considerable interest in the ability to modulate biological processes with magnetic fields. Here we demonstrate a strategy for selecting aptamers that exhibit enhanced binding to paramagnetic metal ions under a strong magnetic field. Using a high-magnetic-field (HM)-SELEX method targeting Co2+, we identified two classes of aptamers with magnetically-modulated binding behavior. One displayed a gradual 2–3-fold increase in affinity as magnetic field strength increased, while the other went from minimal target binding at ambient field strength to an affinity of ~200 μM at ≥ 6 T. Molecular simulations revealed that the magnetic field induces a global conformational rearrangement by enhancing aptamer-metal electrostatic interactions, optimizing the coordination geometry of the nucleotides. Chemical footprinting and mutational analysis confirmed the role of certain conformational changes in magnetically-induced ion binding. These results suggest opportunities to generate aptamer switches that can be used to manipulate biorecognition processes via an externally applied magnetic field in diverse applications.
Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials. The high-throughput sequencing data generated in this study have been deposited in the NCBI Gene Expression Omnibus database under accession code PRJNA1395820. The source data generated in this study are provided in the Source Data file. Source data are provided with this paper.
References
Long, X., Ye, J., Zhao, D. & Zhang, S.-J. Magnetogenetics: Remote non-invasive magnetic activation of neuronal activity with a magnetoreceptor. Sci. Bull. 60, 2107–2119 (2015).
Qin, S. et al. A magnetic protein biocompass. Nat. Mater. 15, 217–226 (2016).
Meister, M. Physical limits to magnetogenetics. eLife 5, e17210 (2016).
Winklhofer, M. & Mouritsen, H. A room-temperature ferrimagnet made of metallo-proteins? 094607 Preprint at https://doi.org/10.1101/094607 (2020).
Sigel, H. Interactions of metal ions with nucleotides and nucleic acids and their constituents. Chem. Soc. Rev. 22, 255 (1993).
Sigel, R. K. O. & Sigel, H. A stability concept for metal ion coordination to single-stranded nucleic acids and affinities of individual sites. Acc. Chem. Res. 43, 974–984 (2010).
Izatt, R. M., Christensen, J. J. & Rytting, J. H. Sites and thermodynamic quantities associated with proton and metal ion interaction with ribonucleic acid, deoxyribonucleic acid, and their constituent bases, nucleosides, and and nucleotides. Chem. Rev. 71, 439–481 (1971).
Furukawa, K. et al. Bacterial riboswitches cooperatively bind Ni2+ or Co2+ ions and control expression of heavy metal transporters. Mol. Cell 57, 1088–1098 (2015).
Dambach, M. et al. The ubiquitous yybP-ykoY riboswitch is a manganese-responsive regulatory element. Mol. Cell 57, 1099–1109 (2015).
Price, I. R., Gaballa, A., Ding, F., Helmann, J. D. & Ke, A. Mn2+-sensing mechanisms of yybP-ykoY orphan riboswitches. Mol. Cell 57, 1110–1123 (2015).
Xu, T. et al. Small DNAs that bind Nickel(II) specifically and tightly. Anal. Chem. 93, 14912–14917 (2021).
Wu, Y. et al. Simultaneous Fe2+/Fe3+ imaging shows Fe3+ over Fe2+ enrichment in Alzheimer’s disease mouse brain. Sci. Adv. 9, eade7622 (2023).
Wrzesinski, J. & Ciesiolka, J. Characterization of structure and metal ions specificity of Co2+-binding RNA aptamers. Biochemistry 44, 6257–6268 (2005).
Nishiyabu, R. et al. Nanoparticles of adaptive supramolecular networks self-assembled from nucleotides and lanthanide ions. J. Am. Chem. Soc. 131, 2151–2158 (2009).
Tsitovich, P. B., Spernyak, J. A. & Morrow, J. R. A redox-activated MRI contrast agent that switches between paramagnetic and diamagnetic states. Angew. Chem. Int Ed. 52, 13997–14000 (2013).
Gale, E. M., Jones, C. M., Ramsay, I., Farrar, C. T. & Caravan, P. A janus chelator enables biochemically responsive MRI contrast with exceptional dynamic range. J. Am. Chem. Soc. 138, 15861–15864 (2016).
Zakharchenko, A., Guz, N., Laradji, A. M., Katz, E. & Minko, S. Magnetic field remotely controlled selective biocatalysis. Nat. Catal. 1, 73–81 (2017).
Liu, X. et al. Magnetic-field-oriented mixed-valence-stabilized ferrocenium anion-exchange membranes for fuel cells. Nat. Energy 7, 329–339 (2022).
Rangel, A. E., Hariri, A. A., Eisenstein, M. & Soh, H. T. Engineering aptamer switches for multifunctional stimulus-responsive nanosystems. Adv. Mater. 32, 2003704 (2020).
Zhu, H. et al. Spatial control of in vivo CRISPR–Cas9 genome editing via nanomagnets. Nat. Biomed. Eng. 3, 126–136 (2018).
Park, J.-W., Tatavarty, R., Kim, D. W., Jung, H.-T. & Gu, M. B. Immobilization-free screening of aptamers assisted by graphene oxide. Chem. Commun. 48, 2071–2073 (2012).
Stangherlin, S., Lui, N., Lee, J. H. & Liu, J. Aptamer-based biosensors: from SELEX to biomedical diagnostics. TrAC Trends Anal. Chem. 191, 118349 (2025).
Shi, H. et al. Selection and application of DNA aptamers against sulfaquinoxaline assisted by graphene oxide–based SELEX. Food Anal. Methods 14, 250–259 (2021).
Wang, J. et al. A general transition metal binding aptamer following the Irving–Williams series. Chem. Sci. 16, 14286–14294 (2025).
Alkhamis, O. et al. Exploring the relationship between aptamer binding thermodynamics, affinity, and specificity. Nucleic Acids Res. 53, gkaf219 (2025).
Slavkovic, S. & Johnson, P. E. Analysis of aptamer-small molecule binding interactions using isothermal titration calorimetry. in Nucleic Acid Aptamers (eds Mayer, G. & Menger, M. M.) vol. 2570 105–118 (Springer US, New York, NY, 2023).
Cowan, J. A. Recognition of a cognate RNA aptamer by neomycin B: quantitative evaluation of hydrogen bonding and electrostatic interactions. Nucleic Acids Res. 28, 2935–2942 (2000).
Black, C. B. & Cowan, J. A. Quantitative evaluation of electrostatic and hydrogen-bonding contributions to metal cofactor binding to nucleic acids. J. Am. Chem. Soc. 116, 1174–1178 (1994).
Ding, Y., Heng, Y., Wong, K., Chen, Q. & Liu, J. Enriching higher affinity aptamers by addressing the kinetic aspect of the DNA strand-displacement reaction. Angew. Chem. Int. Ed n/a, e14445 (2025).
Aich, P. et al. M-DNA: a complex between divalent metal ions and DNA which behaves as a molecular wire. J. Mol. Biol. 294, 477–485 (1999).
Lee, J. S., Latimer, L. J. P. & Reid, R. S. A cooperative conformational change in duplex DNA induced by Zn2+ and other divalent metal ions. Biochem. Cell Biol. 71, 162–168 (1993).
Bailey, T. L. et al. MEME SUITE: Tools for motif discovery and searching. Nucleic Acids Res. 37, W202–W208 (2009).
Hou, T., Wang, J., Li, Y. & Wang, W. Assessing the performance of the MM/PBSA and MM/GBSA methods. 1. The accuracy of binding free energy calculations based on molecular dynamics simulations. J. Chem. Inf. Model. 51, 69–82 (2011).
Zhou, M. et al. Small DNAs that specifically and tightly bind transition metal ions. J. Am. Chem. Soc. 145, 8776–8780 (2023).
Smestad, J., Wilbanks, B. & Maher, L. J. An in vitro selection strategy identifying naked DNA that localizes to cell nuclei. J. Am. Chem. Soc. 141, 18375–18379 (2019).
Chen, H. et al. Exploring the formation and recognition of an Important G-Quadruplex in a HIF1α promoter and its transcriptional inhibition by a Benzo[c]phenanthridine derivative. J. Am. Chem. Soc. 136, 2583–2591 (2014).
McManus, S. A. & Li, Y. A deoxyribozyme with a novel guanine quartet-helix pseudoknot structure. J. Mol. Biol. 375, 960–968 (2008).
Mao, Y. et al. Evolution of a highly functional circular DNA aptamer in serum. Nucleic Acids Res. 48, 10680–10690 (2020).
Zhou, W., Saran, R. & Liu, J. Metal sensing by DNA. Chem. Rev. 117, 8272–8325 (2017).
Xu, J. et al. Magnetic sensitivity of cryptochrome 4 from a migratory songbird. Nature 594, 535–540 (2021).
Li, J. H. et al. Directed manipulation of membrane proteins by fluorescent magnetic nanoparticles. Nat. Commun. 11, 4259 (2020).
Jiang, H., Fu, H., Min, T., Hu, P. & Shi, J. Magnetic-manipulated NK cell proliferation and activation enhance immunotherapy of orthotopic liver cancer. J. Am. Chem. Soc. 145, 13147–13160 (2023).
Liu, W. et al. Remote tuning of built-in magnetoelectric microenvironment to promote bone regeneration by modulating cellular exposure to arginylglycylaspartic acid peptide. Adv. Funct. Mater. 31, 2006226 (2021).
Lee, J. et al. Non-contact long-range magnetic stimulation of mechanosensitive ion channels in freely moving animals. Nat. Mater. 20, 1029–1036 (2021).
Greenberg, B. D., Ziemann, U., Harmon, A., Murphy, D. L. & Wassermann, E. M. Decreased neuronal inhibition in cerebral cortex in obsessive compulsive disorder on transcranial magnetic stimulation. Lancet 352, 881–882 (1998).
Stanley, S. A., Sauer, J., Kane, R. S., Dordick, J. S. & Friedman, J. M. Remote regulation of glucose homeostasis in mice using genetically encoded nanoparticles. Nat. Med. 21, 92–98 (2015).
Chen, Y. et al. Plasmonic/magnetic nanoarchitectures: From controllable design to biosensing and bioelectronic interfaces. Biosens. Bioelectron. 219, 114744 (2023).
Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 31, 3406–3415 (2003).
Wang, J. et al. Optimization of RNA 3D structure prediction using evolutionary restraints of nucleotide–nucleotide interactions from direct coupling analysis. Nucleic Acids Res. 45, 6299–6309 (2017).
Wang, J., Wang, J., Huang, Y. & Xiao, Y. 3dRNA v2.0: An updated web server for RNA 3D structure prediction. IJMS 20, 4116 (2019).
Zhang, Y., Jiang, Y., Peng, J. & Zhang, H. Rational design of nonbonded point charge models for divalent metal cations with Lennard-Jones 12-6 potential. J. Chem. Inf. Model. 61, 4031–4044 (2021).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (U23A20265, 32072306 to L.Z.), and the Anhui Provincial Natural Science Foundation (2408085MC072 to L.W.). We thank the staff members of the SM1 superconducting magnet (https://cstr.cn/31125.02.SHMFF.SM1.MG) at the Steady High Magnetic Field Facility, CAS (https://cstr.cn/31125.02.SHMFF), for providing technical support and assistance. The analysis work of this article was partially carried out at the Instrumental Analysis Center, Hefei University of Technology.
Author information
Authors and Affiliations
Contributions
Conceptualization: L.W., H.T.S., and L.Z. Methodology: S.G., L.W., Y.M., H.T.S., and L.Z. Investigation and Validation: S.G. and L.Y. Visualization: S.G. Data Curation: S.G. and L.Y. Formal analysis: S.G. and L.W. Resources: L.W., Y.M., and L.Z. Writing—Original Draft: S.G. and L.W. Writing—review & Editing: S.G., L.W., M.E., H.T.S,. and L.Z. Supervision, Project administration: L.W., H.T.S., and L.Z. Funding acquisition: L.W. and L.Z. All authors have read and agreed to the submitted version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
S. G., L. W., and L. Z. are inventors on a patent granted to Hefei University of Technology related to the screening of magnetic-sensitive aptamers for Co2+ (application number 202510331988.8). The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Hongzhou Gu, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gao, S., Wang, L., Yao, L. et al. Aptamers with magnetically tunable affinity for divalent cobalt ions. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70871-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70871-9