Abstract
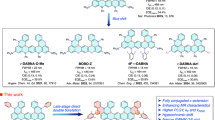
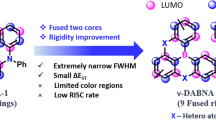
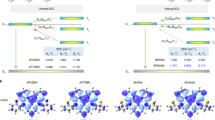
Boron/nitrogen (B/N)-doped multi-resonance thermally activated delayed fluorescence (TADF) molecules have emerged as benchmark narrowband emitters for organic light-emitting diodes (OLEDs). However, these emitters face persistent challenges in synthesis and optoelectronic performance, notably aggregation-induced spectral-broadening and inefficient reverse intersystem crossing (RISC). Here, we introduce a molecular design that incorporates a B–N–B covalent-bond into a multiple resonance (MR) framework, synergistically combining narrowband emission of para-positioned B/N with a helically distorted B–N–B configuration that enhances spin-orbit coupling and suppresses molecular aggregations. A lithium-free, stepwise nitrogen-directed borylation enables high-synthesis-yield ( > 80%) targeted emitters, affording deep-blue (452 nm) and greenish (495 nm) TADF emissions with full-width-at-half-maximum of merely 12–14 nm, near-unity photoluminescence quantum yields and accelerated RISC rates ( > 105 s−1). Corresponding OLEDs simultaneously achieve high maximum external quantum efficiencies of 37.9–38.3%, narrow electroluminescence bandwidths of 15–17 nm and decent operational stabilities. This work establishes B–N–B integrated MR-TADF systems as a versatile platform toward high-performance organic optoelectronics.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the paper and the Supplementary Information. Source data are provided with this paper.
References
Liu, Z. & Marder, T. B. B–N versus C–C: how similar are they? Angew. Chem. Int. Ed. 47, 242–244 (2008).
Bosdet, M. J. D. & Piers, W. E. B–N as a C–C substitute in aromatic systems. Can. J. Chem. 87, 8–29 (2009).
Helten, H. B=N units as part of extended π-conjugated oligomers and polymers. Chem. – Eur. J. 22, 12972–12982 (2016).
Wang, X.-Y., Wang, J.-Y. & Pei, J. BN heterosuperbenzenes: synthesis and properties. Chem. – Eur. J. 21, 3528–3539 (2015).
Kaehler, T., Bolte, M., Lerner, H.-W. & Wagner, M. Introducing perylene as a new member to the azaborine family. Angew. Chem. Int. Ed. 58, 11379–11384 (2019).
Krieg, M. et al. Construction of an internally B3N3-doped nanographene molecule. Angew. Chem. Int. Ed. 54, 8284–8286 (2015).
Dosso, J. et al. Synthesis and optoelectronic properties of hexa-peri-hexabenzoborazinocoronene. Angew. Chem. Int. Ed. 56, 4483–4487 (2017).
Giustra, Z. X. & Liu, S.-Y. The state of the art in azaborine chemistry: new synthetic methods and applications. J. Am. Chem. Soc. 140, 1184–1194 (2018).
Meng, G. et al. Amine-directed formation of B−N bonds for BN-fused polycyclic aromatic multiple resonance emitters with narrowband emission. Angew. Chem. Int. Ed. 61, e202207293 (2022).
Meng, G. et al. B–N covalent bond embedded double hetero-[n]helicenes for pure red narrowband circularly polarized electroluminescence with high efficiency and stability. Adv. Mater. 36, 2307420 (2024).
Campbell, P. G., Marwitz, A. J. V. & Liu, S.-Y. Recent advances in azaborine chemistry. Angew. Chem. Int. Ed. 51, 6074–6092 (2012).
Hatakeyama, T., Hashimoto, S., Seki, S. & Nakamura, M. Synthesis of BN-fused polycyclic aromatics via tandem intramolecular electrophilic arene borylation. J. Am. Chem. Soc. 133, 18614–18617 (2011).
Hatakeyama, T., Hashimoto, S., Oba, T. & Nakamura, M. Azaboradibenzo[6]helicene: carrier inversion induced by helical homochirality. J. Am. Chem. Soc. 134, 19600–19603 (2012).
Wang, S. et al. In situ solid-state generation of (BN)2-pyrenes and electroluminescent devices. Angew. Chem. Int. Ed. 54, 15074–15078 (2015).
Zhao, K. et al. A modular approach toward BN-embedded terrylene diimides. Angew. Chem. Int. Ed. 64, e202503571 (2025).
Zhao, K. et al. “Spine surgery” of perylene diimides with covalent B–N bonds toward electron-deficient BN-embedded polycyclic aromatic hydrocarbons. J. Am. Chem. Soc. 144, 3091–3098 (2022).
Min, Y., Dou, C., Liu, D., Dong, H. & Liu, J. Quadruply B←N-fused dibenzo-azaacene with high electron affinity and high electron mobility. J. Am. Chem. Soc. 141, 17015–17021 (2019).
Yang, G., Liu, J., Yang, Y., Bin, Z. & You, J. Unveiling the centrosymmetric effect in the design of narrowband fluorescent emitters: from single to double difluoroboron cores. J. Am. Chem. Soc. 147, 1251–1261 (2025).
Guo, Y. et al. Simple boron–nitrogen covalent bond constructs multi-resonance TADF emitters: ultra-narrowband deep-blue electroluminescence. Angew. Chem. Int. Ed. 64, e202503320 (2025).
Chen, X., Tan, D. & Yang, D.-T. Multiple-boron–nitrogen (multi-BN) doped π-conjugated systems for optoelectronics. J. Mater. Chem. C. 10, 13499–13532 (2022).
Tan, D. et al. Multiple helicenes defected by heteroatoms and heptagons with narrow emissions and superior photoluminescence quantum yields. Angew. Chem. Int. Ed. 62, e202304711 (2023).
Franceschini, M. et al. Peri-acenoacene ribbons with zigzag BN-doped peripheries. J. Am. Chem. Soc. 144, 21470–21484 (2022).
Yang, D.-T. et al. Doping polycyclic arenes with nitrogen–boron–nitrogen (NBN) units. Org. Lett. 20, 6741–6745 (2018).
Wang, X. et al. Synthesis of NBN-type zigzag-edged polycyclic aromatic hydrocarbons: 1,9-diaza-9a-boraphenalene as a structural motif. J. Am. Chem. Soc. 138, 11606–11615 (2016).
Wei, H. et al. B–N–B bond embedded phenalenyl and its anions. J. Am. Chem. Soc. 139, 15760–15767 (2017).
Scholz, A. S. et al. BNB-doped phenalenyls: modular synthesis, optoelectronic properties, and one-electron reduction. J. Am. Chem. Soc. 142, 11072–11083 (2020).
Sun, Z. et al. Divergent synthesis of double heterohelicenes as strong chiral double hydrogen-bonding donors. Org. Lett. 24, 6670–6675 (2022).
Yu, Y. et al. Benzo-extended heli(aminoborane)s: inner rim BN-doped helical molecular carbons with remarkable chiroptical properties. J. Am. Chem. Soc. 146, 22600–22611 (2024).
Yu, Y. et al. -Extended heli(aminoborane)s with highly bright circularly polarized luminescence and narrowband emission. Angew. Chem. Int. Ed. 64, e202501645 (2025).
Chen, C., Du, C.-Z. & Wang, X.-Y. The rise of 1,4-BN-heteroarenes: synthesis, properties, and applications. Adv. Sci. 9, 2200707 (2022).
Hatakeyama, T. et al. Ultrapure blue thermally activated delayed fluorescence molecules: efficient HOMO–LUMO separation by the multiple resonance effect. Adv. Mater. 28, 2777–2781 (2016).
Kondo, Y. et al. Narrowband deep-blue organic light-emitting diode featuring an organoboron-based emitter. Nat. Photon. 13, 678–682 (2019).
Hua, T. et al. Deep-blue organic light-emitting diodes for ultrahigh-definition displays. Nat. Photon. 18, 1161–1169 (2024).
Yuan, W., Jin, Q., Du, M., Duan, L. & Zhang, Y. Tailoring ultra-narrowband tetraborylated multiple resonance emitter for high-performance blue OLED. Adv. Mater. 36, 2410096 (2024).
Oda, S. et al. One-shot synthesis of expanded heterohelicene exhibiting narrowband thermally activated delayed fluorescence. J. Am. Chem. Soc. 144, 106–112 (2022).
Cheng, Y. C. et al. Efficient, narrow-band, and stable electroluminescence from organoboron-nitrogen-carbonyl emitter. Nat. Commun. 15, 731 (2024).
Fan, X. C. et al. Stable narrowband blue OLEDs by modulating frontier molecular orbital levels. Nat. Commun. 16, 4936 (2025).
Liu, J. et al. Toward a BT.2020 green emitter through a combined multiple resonance effect and multi-lock strategy. Nat. Commun. 13, 4876 (2022).
Hao, J. et al. Late-stage direct double borylation of B/N-based multi-resonance framework enables high-performance ultra-narrowband deep-blue organic light-emitting diodes. Nat. Commun. 16, 8867 (2025).
Fan, T. et al. High-efficiency narrowband multi-resonance emitter fusing indolocarbazole donors for BT. 2020 red electroluminescence and ultralong operation lifetime. Adv. Mater. 35, 2301018 (2023).
Wang, X. et al. Improving the stability and color purity of a BT.2020 blue multiresonance emitter by alleviating hydrogen repulsion. Sci. Adv. 9, eadh1434 (2023).
Lv, X. et al. Extending the π-skeleton of multi-resonance TADF materials towards high-efficiency narrowband deep-blue emission. Angew. Chem. Int. Ed. 61, e202201588 (2022).
Hayakawa, M. et al. “Core–shell” wave function modulation in organic narrowband emitters. J. Am. Chem. Soc. 146, 18331–18340 (2024).
Oda, S. et al. Carbazole-based DABNA analogues as highly efficient thermally activated delayed fluorescence materials for narrowband organic light-emitting diodes. Angew. Chem. Int. Ed. 60, 2882–2886 (2021).
Jaska, C. A. et al. Triphenylene analogues with B2N2C2 cores: synthesis, structure, redox behavior, and photophysical properties. J. Am. Chem. Soc. 128, 10885–10896 (2006).
Bosdet, M. J. D. et al. Blue fluorescent 4a-aza-4b-boraphenanthrenes. Org. Lett. 9, 1395–1398 (2007).
Li, G. et al. 1,5,9-Triaza-2,6,10-triphenylboracoronene: BN-embedded analogue of coronene. Org. Lett. 17, 560–563 (2015).
Jin, J.-M. et al. Synergetic modulation of steric hindrance and excited state for anti-quenching and fast spin-flip multi-resonance thermally activated delayed fluorophore. Angew. Chem. Int. Ed. 63, e202401120 (2024).
Guo, L. et al. Synergetic multiple charge-transfer excited states for anti-quenching and rapid spin-flip multi-resonance thermally activated delayed fluorescence emitter. Adv. Mater. 37, 2500269 (2025).
Jiang, P. et al. Quenching-resistant multiresonance TADF emitter realizes 40% external quantum efficiency in narrowband electroluminescence at high doping level. Adv. Mater. 34, 2106954 (2022).
Wu, Y., Liu, J., Yang, G., Bin, Z. & You, J. Aromaticity localization effects in polycyclic aromatic hydrocarbons for discovering narrowband fluorescence materials. J. Am. Chem. Soc. 147, 19305–19314 (2025).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Humphrey, W., Dalke, A. & Schulten, K. V. M. D. Visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Liu, Z., Lu, T. & Chen, Q. An sp-hybridized all-carboatomic ring, cyclo[18]carbon: electronic structure, electronic spectrum, and optical nonlinearity. Carbon 165, 461–467 (2020).
Shuai, Z. & Peng, Q. Excited states structure and processes: understanding organic light-emitting diodes at the molecular level. Phys. Rep. 537, 123–156 (2014).
Shuai, Z. & Peng, Q. Organic light-emitting diodes: theoretical understanding of highly efficient materials and development of computational methodology. Natl. Sci. Rev. 4, 224–239 (2017).
Ha, J. M. et al. Rational molecular design of azaacene-based narrowband green-emitting fluorophores: modulation of spectral bandwidth and vibronic transitions. ACS Appl. Mater. Inter. 13, 26227–26236 (2021).
Qiu, X. et al. Narrowband emission from organic fluorescent emitters with dominant low-frequency vibronic coupling. Adv. Opt. Mater. 9, 2001845 (2021).
Neese, F. Software update: the ORCA program system—version 5.0. WIREs Comput. Mol. Sci. 12, e1606 (2022).
Chen, X.-K., Kim, D. & Brédas, J.-L. Thermally activated delayed fluorescence (TADF) path toward efficient electroluminescence in purely organic materials: molecular level insight. Acc. Chem. Res. 51, 2215–2224 (2018).
Fan, X.-C. et al. Ultrapure green organic light-emitting diodes based on highly distorted fused π-conjugated molecular design. Nat. Photon. 17, 280–285 (2023).
Xu, Y. et al. Highly efficient electroluminescent materials with high color purity based on strong acceptor attachment onto B–N-containing multiple resonance frameworks. CCS Chem 4, 2065–2079 (2022).
Sun, J. et al. Exceptionally stable blue phosphorescent organic light-emitting diodes. Nat. Photon. 16, 212–218 (2022).
Zhang, D., Cai, M., Zhang, Y., Zhang, D. & Duan, L. Sterically shielded blue thermally activated delayed fluorescence emitters with improved efficiency and stability. Mater. Horiz. 3, 145–151 (2016).
Zhang, D., Duan, L., Li, C., Li, Y. & Zhang, D. High-efficiency fluorescent organic light-emitting devices using sensitizing hosts with a small singlet–triplet exchange energy. Adv. Mater. 26, 5050–5055 (2014).
Nakanotani, H. et al. High-efficiency organic light-emitting diodes with fluorescent emitters. Nat. Commun. 5, 4016 (2014).
Chen, Y. et al. Approaching nearly 40% external quantum efficiency in organic light emitting diodes utilizing a green thermally activated delayed fluorescence emitter with an extended linear donor–acceptor–donor structure. Adv. Mater. 33, 2103293 (2021).
Song, J., Lee, H., Jeong, E. G., Choi, K. C. & Yoo, S. Organic light-emitting diodes: pushing toward the limits and beyond. Adv. Mater. 32, 1907539 (2020).
Lee, Y. T. et al. Bright, efficient, and stable pure-green hyperfluorescent organic light-emitting diodes by judicious molecular design. Nat. Commun. 15, 3174 (2024).
Acknowledgments
This work was supported by the National Natural Science Foundation of China (grant nos. 52573200, 52222308 and U25A20569), the National Key Research and Development Program (grant nos. 2023YFE0203300 and 2024YFB3612100).
Author information
Authors and Affiliations
Contributions
D.D.Z. and L.D. conceived and supervised this work. D.D.Z. proposed the molecular design concept and designed the experiments. J.Z. synthesized materials, carried out the quantum chemical calculations, fabricated the devices and wrote the paper. C.L., Q.W., and D.W.Z. helped synthesize materials. H.Z. helped measure the device's performance. G.M. helped revise the paper. D.D.Z. discussed the results and wrote and revised the paper with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Pachaiyappan Rajamalli, Juozas Grazulevicius, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhou, J., Meng, G., Zhang, H. et al. B–N–B Embedded multiple-resonance polyaromatic enabling efficient narrowband electroluminescence. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70915-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70915-0