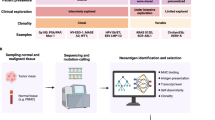
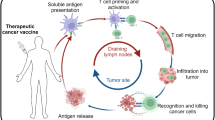
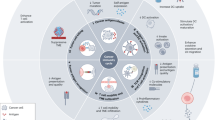
Abstract
Personalized vaccines demonstrate remarkable potential in leveraging tumor-specific adaptive immunity for cancer therapy. Nevertheless, current platforms face persistent challenges, including premature systemic clearance and imprecise antigen-presenting cell targeting, culminating in transient and inefficient antitumor immunity. Furthermore, the technical complexity, extended timelines, and prohibitive costs required for tumor-specific neoantigen identification continue to impede the clinical translation of personalized cancer vaccines. Here, we report a tumor-derived extracellular vesicle-based scaffold vaccine that elicits robust and durable antitumor immunity for personalized cancer immunotherapy. Following subcutaneous administration, the in situ-formed hydrogel vaccine serves as a sustained reservoir for tumor-derived extracellular vesicle antigens and adjuvants while recruiting antigen-presenting dendritic cells to accumulate within the scaffold. Upon exposure to this antigen-rich depot, immature dendritic cells undergo efficient activation, with subsequently matured dendritic cells migrating to draining lymph nodes, where they induce potent and persistent tumor-specific CD8+ T-cell responses that suppress tumor progression across multiple murine models. Specifically, when using tumor-derived extracellular vesicles isolated from surgically excised tumor tissues, the patient-tailored vaccines demonstrate remarkable efficacy in preventing postoperative recurrence. Our findings validate the robust and durable therapeutic efficacy of this vaccine platform, highlighting its potential as a customizable strategy for personalized cancer immunotherapy.
Similar content being viewed by others
Data availability
The RNA sequencing data have been deposited in the NCBI Sequence Read Archive (SRA) under accession number PRJNA1330898. All other data supporting the findings of this study are included in the main article, supplementary information, or source data file. Source data are provided with this paper.
References
Kang, S. et al. Engineered IL-7 synergizes with IL-12 immunotherapy to prevent T cell exhaustion and promote memory without exacerbating toxicity. Sci. Adv. 9, eadh9879 (2023).
Ben-Akiva, E., Chapman, A., Mao, T. & Irvine, D. J. Linking vaccine adjuvant mechanisms of action to function. Sci. Immunol. 10, eado5937 (2025).
Kirchhammer, N., Trefny, M. P., Auf der Maur, P., Läubli, H. & Zippelius, A. Combination cancer immunotherapies: emerging treatment strategies adapted to the tumor microenvironment. Sci. Transl. Med. 14, eabo3605 (2022).
Lin, M. J. et al. Cancer vaccines: the next immunotherapy frontier. Nat. Cancer 3, 911–926 (2022).
Dolton, G. et al. Targeting of multiple tumor-associated antigens by individual T cell receptors during successful cancer immunotherapy. Cell 186, 3333–3349.e27 (2023).
Sahin, U. & Türeci, Ö Personalized vaccines for cancer immunotherapy. Science 359, 1355–1360 (2018).
Peri, A. et al. The landscape of T cell antigens for cancer immunotherapy. Nat. Cancer 4, 937–954 (2023).
Chesney, J. A. et al. Randomized, double-blind, placebo-controlled, global phase III trial of talimogene laherparepvec combined with pembrolizumab for advanced melanoma. J. Clin. Oncol. 41, 528–540 (2023).
Liang, F. et al. Vaccine priming is restricted to draining lymph nodes and controlled by adjuvant-mediated antigen uptake. Sci. Transl. Med. 9, eaal2094 (2017).
Roth, G. A. et al. Designing spatial and temporal control of vaccine responses. Nat. Rev. Mater. 7, 174–195 (2022).
Xia, H. et al. Computational prediction of MHC anchor locations guides neoantigen identification and prioritization. Sci. Immunol. 8, eabg2200 (2023).
Li, J. et al. The screening, identification, design and clinical application of tumor-specific neoantigens for TCR-T cells. Mol. Cancer 22, 141 (2023).
Pearlman, A. H. et al. Targeting public neoantigens for cancer immunotherapy. Nat. Cancer 2, 487–497 (2021).
Manno, M., Bongiovanni, A., Margolis, L., Bergese, P. & Arosio, P. The physico-chemical landscape of extracellular vesicles. Nat. Rev. Bioeng. 3, 68–82 (2025).
Tkach, M. & Théry, C. Communication by extracellular vesicles: where we are and where we need to go. Cell 164, 1226–1232 (2016).
Cocozza, F., Grisard, E., Martin-Jaular, L., Mathieu, M. & Théry, C. Snapshot: extracellular vesicles. Cell 182, 262–262.e1 (2020).
Naseri, M., Bozorgmehr, M., Zöller, M., Pirmardan, E. R. & Madjd, Z. Tumor-derived exosomes: the next generation of promising cell-free vaccines in cancer immunotherapy. Oncoimmunology 9, 1779991 (2020).
Zhou, W. et al. Exosomes derived from immunogenically dying tumor cells as a versatile tool for vaccination against pancreatic cancer. Biomaterials 280, 121306 (2022).
Wiklander, O. P. B., Brennan, M. Á, Lötvall, J., Breakefield, X. O. & Andaloussi, S. E. Advances in therapeutic applications of extracellular vesicles. Sci. Transl. Med. 11, eaav8521 (2019).
Zhang, C. et al. Tissue-derived extracellular vesicles: isolation, purification, and multiple roles in normal and tumor tissues. Life Sci. 321, 121624 (2023).
Han, J. et al. Novel personalized cancer vaccine using tumor extracellular vesicles with attenuated tumorigenicity and enhanced immunogenicity. Adv. Sci. 11, e2308662 (2024).
Han, L. et al. Removing the stumbling block of exosome applications in clinical and translational medicine: expand production and improve accuracy. Stem Cell Res. Ther. 14, 57 (2023).
Li, S. et al. Ex vivo expansion and hydrogel-mediated in vivo delivery of tissue-resident memory T cells for immunotherapy. Sci. Adv. 10, eadm7928 (2024).
Barlek, M. H. et al. Systemic peptide amphiphile nanofiber delivery following subcutaneous injection. Biomaterials 303, 122401 (2023).
Hernandez, A., Hartgerink, J. D. & Young, S. Self-assembling peptides as immunomodulatory biomaterials. Front. Bioeng. Biotechnol. 11, 1139782 (2023).
Gong, N. et al. Enhancing in situ cancer vaccines using delivery technologies. Nat. Rev. Drug Discov. 23, 607–625 (2024).
Liu, T. et al. Injectable nano-in-gel vaccine for spatial and temporal control of vaccine kinetics and breast cancer postsurgical therapy. ACS Nano 18, 3087–3100 (2024).
Kim, J. et al. Injectable, spontaneously assembling, inorganic scaffolds modulate immune cells in vivo and increase vaccine efficacy. Nat. Biotechnol. 33, 64–72 (2015).
Falcone, N. et al. Peptide hydrogels as immunomaterials and their use in cancer immunotherapy delivery. Adv. Healthc. Mater. 12, e2301096 (2023).
Wang, F. et al. Supramolecular filament hydrogel as a universal immunomodulator carrier for immunotherapy combinations. ACS Nano 17, 10651–10664 (2023).
Wang, F. et al. Tumour sensitization via the extended intratumoural release of a STING agonist and camptothecin from a self-assembled hydrogel. Nat. Biomed. Eng. 4, 1090–1101 (2020).
Wang, F. et al. Supramolecular prodrug hydrogelator as an immune booster for checkpoint blocker-based immunotherapy. Sci. Adv. 6, eaaz8985 (2020).
Fan, Q. et al. An implantable blood clot–based immune niche for enhanced cancer vaccination. Sci. Adv. 6, eabb4639 (2020).
Shah, N. J. et al. A biomaterial-based vaccine eliciting durable tumour-specific responses against acute myeloid leukaemia. Nat. Biomed. Eng. 4, 40–51 (2020).
Makarov, D. V., Loeb, S., Getzenberg, R. H. & Partin, A. W. Biomarkers for prostate cancer. Annu. Rev. Med. 60, 139–151 (2009).
Reiter, R. E. et al. Prostate stem cell antigen: a cell surface marker overexpressed in prostate cancer. Proc. Natl. Acad. Sci. USA. 95, 1735–1740 (1998).
Wüstemann, T., Haberkorn, U., Babich, J. & Mier, W. Targeting prostate cancer: prostate-specific membrane antigen based diagnosis and therapy. Med. Res. Rev. 39, 40–69 (2019).
Le, D. T. et al. Results from a phase IIb, randomized, multicenter study of GVAX pancreas and CRS-207 compared with chemotherapy in adults with previously treated metastatic pancreatic adenocarcinoma (ECLIPSE study). J. Am. Assoc. Cancer Res. 25, 5493–5502 (2019).
Hewitt, D. B. et al. A Phase 3 randomized clinical trial of chemotherapy with or without algenpantucel-L (hyperacute-pancreas) immunotherapy in subjects with borderline resectable or locally advanced unresectable pancreatic cancer. Ann. Surg. 275, 45–53 (2022).
Batista-Duharte, A., Portuondo, D., Pérez, O. & Carlos, I. Z. Systemic immunotoxicity reactions induced by adjuvanted vaccines. Int. Immunopharmacol. 20, 170–180 (2014).
Lynn, G. M. et al. In vivo characterization of the physicochemical properties of polymer-linked TLR agonists that enhance vaccine immunogenicity. Nat. Biotechnol. 33, 1201–1210 (2015).
Francica, J. R. et al. Thermoresponsive polymer nanoparticles co-deliver RSV F trimers with a TLR-7/8 adjuvant. Bioconjug. Chem. 27, 2372–2385 (2016).
Li, J. et al. Dendritic cell derived exosomes loaded neoantigens for personalized cancer immunotherapies. J. Control. Release 353, 423–433 (2023).
Huang, L. et al. Engineered exosomes as an in situ DC-primed vaccine to boost antitumor immunity in breast cancer. Mol. Cancer 21, 45 (2022).
Zhang, Y. et al. Complete remission of tumors in mice with neoantigen-painted exosomes and anti-PD-1 therapy. Mol. Ther. 31, 3579–3593 (2023).
Meng, Y. et al. Extracellular vesicles-based vaccines: emerging immunotherapies against cancer. J. Control. Release 378, 438–459 (2025).
Shi, X. et al. Antitumor efficacy of interferon-γ-modified exosomal vaccine in prostate cancer. Prostate 80, 811–823 (2020).
Rao, Q. et al. Tumor-derived exosomes elicit tumor suppression in murine hepatocellular carcinoma models and humans in vitro. Hepatology 64, 456–472 (2016).
Liu, H. et al. Co-delivery of tumor-derived exosomes with alpha-galactosylceramide on dendritic cell-based immunotherapy for glioblastoma. Cancer Lett. 411, 182–190 (2017).
Gu, X., Erb, U., Büchler, M. W. & Zöller, M. Improved vaccine efficacy of tumor exosome compared to tumor lysate loaded dendritic cells in mice. Int. J. Cancer 136, E74–E84 (2015).
Mahaweni, N. M. et al. Tumour-derived exosomes as antigen delivery carriers in dendritic cell-based immunotherapy for malignant mesothelioma. J. Extracell. Vesicles 2, 24 (2013).
Zitvogel, L. et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 4, 594–600 (1998).
Peinado, H. et al. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat. Med. 18, 883–891 (2012).
Zhao, S. et al. Tumor-derived exosomal miR-934 induces macrophage M2 polarization to promote liver metastasis of colorectal cancer. J. Hematol. 13, 156 (2020).
Acknowledgements
This study was supported by National Natural Science Foundation of China (82473851 to F.W., 82373358 and 82472142 to L.D., 82227801 to W.X., 82573527 to J.P.); National Key Research and Development Program of China (2025ZD01903300 and 2022YFC3501905 to F.W.); Major Natural Science Research Projects of Shanghai Municipal Education Commission (2023ZKZD23 to W.X.); Scientific Research Specialization on Post-market Clinical Research of Innovative Drugs of National Healthcare Commission (WKZX2023CX100002 to J.P.); Shanghai Top Priority Research Center Project (2023ZZ02014 to W.X.); Shanghai High-level Local Universities Construction Program; Shanghai Rising-Star Program (23QA1405900 to L.D.); National Outstanding Young Physician of China (2022YQ014 to L.D.); Shanghai Rising-Star Young Medical Talents Program (to L.D.); Physician-Scientist Development Award from Shanghai Immune Therapy Institute (to L.D.); Shanghai Jiao Tong University 2030 Program (to L.D.).
Author information
Authors and Affiliations
Contributions
J.P., F.W., L.D., and W.X. supervised the study; Q.C., C.J., F.W., and L.D. designed the experiments; X.D. constructed prostate orthotopic model and breast cancer metastasis model; M.T., Q.S., C.H., and Z.P. assisted in animal experiments; Q.C. completed analysis of transcriptome sequencing data; Q.C., F.W., L.D., and J.P. analyzed data and organized figures; J.P., W.X., L.D., and F.W. provided funding; Q.C., F.W., and J.P. wrote the manuscript. All authors revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Shuguang Tan, Theresa Whiteside and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Q., Jiang, C., Du, X. et al. Spatiotemporally engineered tumor-derived extracellular vesicle-based scaffold vaccine for personalized cancer immunotherapy. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70924-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70924-z