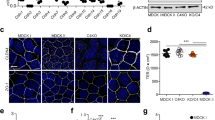
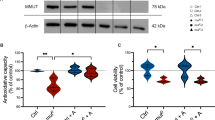
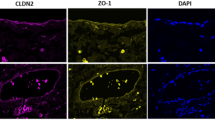
Abstract
Mammals achieve the highest urine concentrations of any vertebrate, a feat that hinges on generating steep osmotic gradients within the renal medulla. Interestingly, the region with the highest osmolality, the inner medulla, is unique to mammals. Among the nephron’s segments, the ascending thin limb (aTL) is the sole element exclusive to this zone and is thought to mediate passive salt reabsorption. However, the architecture and functional impact of the aTL have remained obscure. Here we uncover an unexpected morphogenetic program in the aTL, characterized by extensive apical-junctional interdigitations that greatly increase cell-to-cell contact area. Integrating single-nucleus transcriptomics with high-resolution imaging, we identify claudin-10b, a tight junction protein and paracellular cation pore, as a central driver of this architecture. Inducible deletion of claudin-10b specifically in the aTL abolishes membrane interdigitations and markedly reduces urine-concentrating ability, thereby establishing a direct link between segment-specific epithelial morphology and whole-organ function. Claudin-10b proves necessary for interdigitation formation, acting through transcellular adhesion and interaction with the tight-junction scaffold ZO1. These findings offer definitive evidence that the inner medulla and aTL are essential for maximal urinary concentration, while revealing a non-canonical, morphogenetic role for claudin-10b.
Similar content being viewed by others
Data availability
The single nucleus RNA sequencing data generated in this study have been deposited in the Gene Expression Omnibus database under accession code GSE295103. The processed mean expression data and differential gene expression analysis are available in Supplementary Data 1–7. All other data supporting this study is available from the corresponding author upon request. Source data are provided with this paper.
References
Dantzler, W. H., Layton, A. T., Layton, H. E. & Pannabecker, T. L. Urine-concentrating mechanism in the inner medulla: function of the thin limbs of the loops of Henle. Clin. J. Am. Soc. Nephrol. 9, 1781–1789 (2014).
Sands, J. M. & Layton, H. E. Advances in understanding the urine-concentrating mechanism. Annu. Rev. Physiol. 76, 387–409 (2014).
Rocha, A. S. & Kokko, J. P. Sodium chloride and water transport in the medullary thick ascending limb of Henle. Evidence for active chloride transport. J. Clin. Invest. 52, 612–623 (1973).
Igarashi, P., Vanden Heuvel, G. B., Payne, J. A. & Forbush, B. 3rd Cloning, embryonic expression, and alternative splicing of a murine kidney-specific Na-K-Cl cotransporter. Am. J. Physiol. 269, F405–F418 (1995).
Imai, M. & Kokko, J. P. Sodium chloride, urea, and water transport in the thin ascending limb of Henle. Generation of osmotic gradients by passive diffusion of solutes. J. Clin. Invest. 53, 393–402 (1974).
Takahashi, N. et al. Characterization of Na+ transport across the cell membranes of the ascending thin limb of Henle’s loop. Kidney Int. 47, 789–794 (1995).
Kokko, J. P. & Rector, F. C. Jr. Countercurrent multiplication system without active transport in inner medulla. Kidney Int. 2, 214–223 (1972).
Yoshitomi, K., Kondo, Y. & Imai, M. Evidence for conductive Cl- pathways across the cell membranes of the thin ascending limb of Henle’s loop. J. Clin. Invest. 82, 866–871 (1988).
Uchida, S. & Marumo, F. Severely impaired urine-concentrating ability in mice lacking the CLC-K1 chloride channel. Exp. Nephrol. 8, 361–365 (2000).
Matsumura, Y. et al. Overt nephrogenic diabetes insipidus in mice lacking the CLC-K1 chloride channel. Nat. Genet 21, 95–98 (1999).
Lin, M. H. et al. Impairment in renal medulla development underlies salt wasting in Clc-k2 channel deficiency. JCI Insight https://doi.org/10.1172/jci.insight.151039 (2021).
Wigle, J. T. & Oliver, G. Prox1 function is required for the development of the murine lymphatic system. Cell 98, 769–778 (1999).
Kim, Y.-M. et al. Role of Prox1 in the transforming ascending thin limb of Henle’s loop during mouse kidney development. PLoS ONE 10, e0127429 (2015).
Srinivasan, R. S. et al. Lineage tracing demonstrates the venous origin of the mammalian lymphatic vasculature. Genes Dev. 21, 2422–2432 (2007).
Uchida, S. et al. Localization and functional characterization of rat kidney-specific chloride channel, ClC-K1. J. Clin. Invest. 95, 104–113 (1995).
Chakraborty, T. et al. Light-sheet microscopy of cleared tissues with isotropic, subcellular resolution. Nat. Methods 16, 1109–1113 (2019).
Sonntag, S. R. et al. Diuretic state affects ascending thin limb tight junctions. Am. J. Physiol. Ren. Physiol. 314, F190–F195 (2018).
Ransick, A. et al. Single-cell profiling reveals sex, lineage, and regional diversity in the mouse kidney. Dev. Cell 51, 399–413 e397 (2019).
CZI Cell Science Program, et al. CZ CELLxGENE Discover: a single-cell data platform for scalable exploration, analysis and modeling of aggregated data. Nucleic Acids Res. 53, D886–D900 (2025).
Van Itallie, C. M. & Anderson, J. M. Claudins and epithelial paracellular transport. Annu. Rev. Physiol. 68, 403–429 (2006).
Itoh, M. et al. Direct binding of three tight junction-associated MAGUKs, ZO-1, ZO-2, and ZO-3, with the COOH termini of claudins. J. Cell Biol. 147, 1351–1363 (1999).
Van Itallie, C. M. et al. Two splice variants of claudin-10 in the kidney create paracellular pores with different ion selectivities. Am. J. Physiol. Ren. Physiol. 291, F1288–F1299 (2006).
Gunzel, D. et al. Claudin-10 exists in six alternatively spliced isoforms that exhibit distinct localization and function. J. Cell Sci. 122, 1507–1517 (2009).
Otani, T. et al. Claudins and JAM-A coordinately regulate tight junction formation and epithelial polarity. J. Cell Biol. 218, 3372–3396 (2019).
Miyazaki, S. et al. Mechanism of interdigitation formation at apical boundary of MDCK cell. iScience 26, 106594 (2023).
Furuse, M. et al. Reconstitution of functional tight junctions with individual claudin subtypes in epithelial cells. Cell Struct. Funct. 48, 1–17 (2023).
Morita, K., Furuse, M., Fujimoto, K. & Tsukita, S. Claudin multigene family encoding four-transmembrane domain protein components of tight junction strands. Proc. Natl. Acad. Sci. USA 96, 511–516 (1999).
Krause, G. et al. Structure and function of extracellular claudin domains. Ann. N. Y. Acad. Sci. 1165, 34–43 (2009).
Hempel, C. et al. Assembly of tight junction strands: claudin-10b and claudin-3 form homo-tetrameric building blocks that polymerise in a channel-independent manner. J. Mol. Biol. 432, 2405–2427 (2020).
Piontek, J. et al. Formation of tight junction: determinants of homophilic interaction between classic claudins. FASEB J. 22, 146–158 (2008).
Yu, A. S. et al. Molecular basis for cation selectivity in claudin-2-based paracellular pores: identification of an electrostatic interaction site. J. Gen. Physiol. 133, 111–127 (2009).
Van Itallie, C., Rahner, C. & Anderson, J. M. Regulated expression of claudin-4 decreases paracellular conductance through a selective decrease in sodium permeability. J. Clin. Invest. 107, 1319–1327 (2001).
Van Itallie, C. M., Lidman, K. F., Tietgens, A. J. & Anderson, J. M. Newly synthesized claudins but not occludin are added to the basal side of the tight junction. Mol. Biol. Cell 30, 1406–1424 (2019).
Tokuda, S., Higashi, T. & Furuse, M. ZO-1 knockout by TALEN-mediated gene targeting in MDCK cells: involvement of ZO-1 in the regulation of cytoskeleton and cell shape. PLoS ONE 9, e104994 (2014).
Hancock, J. F., Paterson, H. & Marshall, C. J. A polybasic domain or palmitoylation is required in addition to the CAAX motif to localize p21ras to the plasma membrane. Cell 63, 133–139 (1990).
Klar, J. et al. Altered paracellular cation permeability due to a rare CLDN10B variant causes anhidrosis and kidney damage. PLoS Genet. 13, e1006897 (2017).
Milatz, S. A novel claudinopathy based on claudin-10 mutations. Int. J. Mol. Sci. https://doi.org/10.3390/ijms20215396 (2019).
Fu, Y., Gu, Y., Zheng, Z., Wasteneys, G. & Yang, Z. Arabidopsis interdigitating cell growth requires two antagonistic pathways with opposing action on cell morphogenesis. Cell 120, 687–700 (2005).
Schoofs, H. et al. Dynamic cytoskeletal regulation of cell shape supports resilience of lymphatic endothelium. Nature 641, 465–475 (2025).
Ansel, M., Ramachandran, K., Dey, G. & Brunet, T. Origin and evolution of microvilli. Biol. Cell 116, e2400054 (2024).
Derderian, C., Canales, G. I. & Reiter, J. F. Seriously cilia: a tiny organelle illuminates evolution, disease, and intercellular communication. Dev. Cell 58, 1333–1349 (2023).
Mattila, P. K. & Lappalainen, P. Filopodia: molecular architecture and cellular functions. Nat. Rev. Mol. Cell Biol. 9, 446–454 (2008).
Yamada, E. The fine structure of the renal glomerulus of the mouse. J. Biophys. Biochem. Cytol. 1, 551–566 (1955).
Scott, R. P. & Quaggin, S. E. Review series: the cell biology of renal filtration. J. Cell Biol. 209, 199–210 (2015).
Holzman, L. B. et al. Nephrin localizes to the slit pore of the glomerular epithelial cell. Kidney Int. 56, 1481–1491 (1999).
Gunzel, D. & Yu, A. S. Claudins and the modulation of tight junction permeability. Physiol. Rev. 93, 525–569 (2013).
Kubota, K. et al. Ca(2+)-independent cell-adhesion activity of claudins, a family of integral membrane proteins localized at tight junctions. Curr. Biol. 9, 1035–1038 (1999).
Quintanova, C. et al. Unrecognized role of claudin-10b in basolateral membrane infoldings of the thick ascending limb. Ann. N. Y. Acad. Sci. 1517, 266–278 (2022).
van der Veen, R. E. et al. Claudin-4 polymerizes after a small extracellular claudin-3-like substitution. J. Biol. Chem. 300, 107693 (2024).
Spadaro, D. et al. Tension-dependent stretching activates ZO-1 to control the junctional localization of its interactors. Curr. Biol. 27, 3783–3795 e3788 (2017).
Schwayer, C. et al. Mechanosensation of tight junctions depends on ZO-1 phase separation and flow. Cell 179, 937–952 e918 (2019).
Quadros, R. M. et al. Easi-CRISPR: a robust method for one-step generation of mice carrying conditional and insertion alleles using long ssDNA donors and CRISPR ribonucleoproteins. Genome Biol. 18, 92 (2017).
Lun, A. T. L., Pages, H. & Smith, M. L. beachmat: a bioconductor C++ API for accessing high-throughput biological data from a variety of R matrix types. PLoS Comput. Biol. 14, e1006135 (2018).
Lun, A. T., Bach, K. & Marioni, J. C. Pooling across cells to normalize single-cell RNA sequencing data with many zero counts. Genome Biol. 17, 75 (2016).
Xu, C. & Su, Z. Identification of cell types from single-cell transcriptomes using a novel clustering method. Bioinformatics 31, 1974–1980 (2015).
Haghverdi, L., Buttner, M., Wolf, F. A., Buettner, F. & Theis, F. J. Diffusion pseudotime robustly reconstructs lineage branching. Nat. Methods 13, 845–848 (2016).
van Dijk, D. et al. Recovering gene interactions from single-cell data using data diffusion. Cell 174, 716–729 e727 (2018).
Chaney, C. P., Drake, K. A. & Carroll, T. J. Integration of multiple, diverse methods to identify biologically significant marker genes. J. Mol. Biol. 434, 167754 (2022).
Finak, G. et al. MAST: a flexible statistical framework for assessing transcriptional changes and characterizing heterogeneity in single-cell RNA sequencing data. Genome Biol. 16, 278 (2015).
Liberzon, A. et al. The molecular signatures database (MSigDB) hallmark gene set collection. Cell Syst. 1, 417–425 (2015).
Sergushichev, A. A. An algorithm for fast preranked gene set enrichment analysis using cumulative statistic calculation. Preprint at bioRxiv https://doi.org/10.1101/060012 (2016).
Aibar, S. et al. SCENIC: single-cell regulatory network inference and clustering. Nat. Methods 14, 1083–1086 (2017).
Marciano, D. K. et al. p120 catenin is required for normal renal tubulogenesis and glomerulogenesis. Development 138, 2099–2109 (2011).
Mostov, K. E. & Deitcher, D. L. Polymeric immunoglobulin receptor expressed in MDCK cells transcytoses IgA. Cell 46, 613–621 (1986).
Belardi, B. et al. A weak link with actin organizes tight junctions to control epithelial permeability. Dev. Cell 54, 792–804 e797 (2020).
Yu, A. S. Electrophysiological characterization of claudin ion permeability using stably transfected epithelial cell lines. Methods Mol. Biol. 762, 27–41 (2011).
Gao, L. et al. Afadin orients cell division to position the tubule lumen in developing renal tubules. Development 144, 3511–3520 (2017).
Liu, H. et al. Heterozygous mutation of Vegfr3 reduces renal lymphatics without renal dysfunction. J. Am. Soc. Nephrol. 32, 3099–3113 (2021).
Acknowledgements
We thank the UTSW O’Brien Kidney Research Core, CRI Transgenic Core, McDermott Sequencing Core, and Mineral Metabolism Core. We thank Elizabeth Chen, D.J. Pan, Josh Mendell, and Don Hilgemann (UTSW) for helpful suggestions. We thank Mikio Furuse (National Institute for Physiological Sciences) and Tetsu Otani (Tokyo Metropolitan University) for cell lines and suggestions. This work was funded by NIH R01DK118032 (D.K.M.), R01DK141873 (D.K.M.), RM1GM145399 (K.M.D.) and R35GM133522 (R.F.).
Author information
Authors and Affiliations
Contributions
J.N.W., M.T.D., T.J.C., and D.K.M. conceptualized the study and designed experiments. J.N.W., S.O., B.L.F., J.B.P., C.H., K.G., H.L., A.M.R., and J.M.W. conducted experiments. J.N.W., B.L.F., J.B.P., K.G., and A.M.R. executed mouse studies. J.N.W. and S.O. generated and analyzed cell lines. J.N.W. conducted immunofluorescence and confocal imaging. J.N.W., S.O., C.H., R.W., S.Z., and D.K.M. analyzed data. C.P.C. executed the single nucleus RNA sequencing bioinformatic analysis. B.L.F., H.L., K.M.D., and R.P.F. collected light sheet microscopy images. J.N.W. and D.K.M. wrote the manuscript. All authors reviewed and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
K.M.D. and R.P.F. hold a patent for the axially scanned light-sheet microscope that is currently licensed by Intelligent Imaging Innovations, Inc. and subsequently sub-licensed by Life Canvas Technologies, but do not have any investment interests or financial stakes in these companies. K.M.D. has an investment interest in Discovery Imaging Systems, LLC. All other authors have no competing interests.
Peer review
Peer review information
Nature Communications thanks Vidhi Dalal, Benjamin Humphreys, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Warshaw, J.N., Oh, S., Chaney, C.P. et al. An epithelial morphogenetic program for maximal urine concentration. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70938-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70938-7