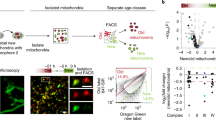
Abstract
A hallmark of mammals is a diploid genome. Despite constraints from dosage compensation and imprinting, haploid embryonic stem cells can be established. However, rapid diploidization is observed in such cultures from mice, rats, and humans, limiting their use and indicating counterselection of a haploid genome. Here, we use metabolic profiling to discover that diploidization is triggered by an imbalance that arises from a smaller cytoplasmic volume and increased mitochondrial density. Reduced respiration causes a change in redox potential, leading to increased NADPH. Conversely, we demonstrate that NADPH oxidation in the mitochondria is sufficient to stabilize the haploid genome. We further show that the redox change leads to reduced AURORA kinase activation on chromosomes, connecting metabolic state to mitotic regulation. Our data, therefore, identify a mitochondrial metabolic imbalance as the root cause of diploidization and connect redox dysregulation to karyotypic instability.
Similar content being viewed by others
Data availability
The RNA seq data of haplod and diploid ESC samples generated in this study have been deposited in the Sequence Read Archive (SRA) database under accession code PRJNA1390253. The raw data underlying the figures are provided as a Source data file. G.D.M. or A.W. may be contacted for additional details on the protocols that support the findings of this study. Source data are provided with this paper.
References
Orr-Weaver, T. L. When bigger is better: the role of polyploidy in organogenesis. Trends Genet. 31, 307–315 (2015).
Morris, J. P., Baslan, T., Soltis, D. E., Soltis, P. S. & Fox, D. T. Integrating the study of polyploidy across organisms, tissues, and disease. Annu. Rev. Genet. 58, 297–318 (2024).
Bogart, J. P. Gynogenetic diploids, tetraploids, or octoploids, and a path to polyploidy in anuran amphibians. Genome 64, 1053–1065 (2021).
Miller, K. E., Cadart, C. & Heald, R. Dodecaploid Xenopus longipes provides insight into the emergence of size scaling relationships during development. Curr. Biol. 33, 1327–1336 (2023).
Mezzasalma, M., Brunelli, E., Odierna, G. & Guarino, F. M. Evolutionary and genomic diversity of true polyploidy in tetrapods. Animals 13, https://doi.org/10.3390/ani13061033 (2023).
Cadart, C., Bartz, J., Oaks, G., Liu, M. Z. & Heald, R. Polyploidy in Xenopus lowers metabolic rate by decreasing total cell surface area. Curr. Biol. 33, 1744–1752 (2023).
Kamel, S., Marsden, J. E. & Pough, F. H. Diploid and tetraploid grey treefrogs (Hyla chrysoscelis and Hyla versicolor) have similar metabolic rates. Comp. Biochem Physiol. A Comp. Physiol. 82, 217–220 (1985).
Glazier, D. S. How metabolic rate relates to cell size. Biology 11, https://doi.org/10.3390/biology11081106 (2022).
Zhou, C. Y. et al. Mitotic chromosomes scale to nuclear-cytoplasmic ratio and cell size in Xenopus. Elife 12, https://doi.org/10.7554/eLife.84360 (2023).
Weeks, A. R., Marec, F. & Breeuwer, J. A. A mite species that consists entirely of haploid females. Science 292, 2479–2482 (2001).
Yaguchi, K. et al. Haploidy-linked cell proliferation defects limit larval growth in zebrafish. Open Biol. 14, 240126 (2024).
Ellinger, M. S. & Murphy, J. A. Cellular morphology in haploid amphibian embryos. J. Embryol. Exp. Morphol. 59, 249–261 (1980).
Ross, L., Davies, N. G. & Gardner, A. How to make a haploid male. Evol. Lett. 3, 173–184 (2019).
Bachtrog, D. et al. Sex determination: why so many ways of doing it? PLoS Biol. 12, e1001899 (2014).
Beukeboom, L. W. et al. Haploid females in the parasitic wasp Nasonia vitripennis. Science 315, 206 (2007).
Wutz, A. Haploid animal cells. Development 141, 1423–1426 (2014).
Leeb, M. et al. Germline potential of parthenogenetic haploid mouse embryonic stem cells. Development 139, 3301–3305 (2012).
Leeb, M. & Wutz, A. Derivation of haploid embryonic stem cells from mouse embryos. Nature 479, 131–134 (2011).
Elling, U. et al. Forward and reverse genetics through derivation of haploid mouse embryonic stem cells. Cell Stem Cell 9, 563–574 (2011).
Sagi, I. et al. Derivation and differentiation of haploid human embryonic stem cells. Nature 532, 107–111 (2016).
Zhong, C. et al. Generation of human haploid embryonic stem cells from parthenogenetic embryos obtained by microsurgical removal of male pronucleus. Cell Res. 26, 743–746 (2016).
Li, W. et al. Androgenetic haploid embryonic stem cells produce live transgenic mice. Nature 490, 407–411 (2012).
Li, W. et al. Genetic modification and screening in rat using haploid embryonic stem cells. Cell Stem Cell 14, 404–414 (2014).
Yang, H. et al. Generation of haploid embryonic stem cells from Macaca fascicularis monkey parthenotes. Cell Res 23, 1187–1200 (2013).
Wang, H. et al. Genetic screening and multipotency in rhesus monkey haploid neural progenitor cells. Development 145, https://doi.org/10.1242/dev.160531 (2018).
Essletzbichler, P. et al. Megabase-scale deletion using CRISPR/Cas9 to generate a fully haploid human cell line. Genome Res. 24, 2059–2065 (2014).
Kotecki, M., Reddy, P. S. & Cochran, B. H. Isolation and characterization of a near-haploid human cell line. Exp. Cell Res. 252, 273–280 (1999).
Freimann, R. & Wutz, A. A fast and efficient size separation method for haploid embryonic stem cells. Biomicrofluidics 11, 054117 (2017).
Qu, C. et al. Haploid embryonic stem cells can be enriched and maintained by simple filtration. J. Biol. Chem. 293, 5230–5235 (2018).
Sun, S. et al. BCL2 is a major regulator of haploidy maintenance in murine embryonic stem cells. Cell Prolif. 56, e13498 (2023).
Zhang, W. et al. Inhibition of apoptosis reduces diploidization of haploid mouse embryonic stem cells during differentiation. Stem Cell Rep. 15, 185–197 (2020).
Olbrich, T. et al. A p53-dependent response limits the viability of mammalian haploid cells. Proc. Natl. Acad. Sci. USA 114, 9367–9372 (2017).
Olbrich, T. et al. A chemical screen identifies compounds capable of selecting for haploidy in mammalian cells. Cell Rep. 28, 597–604 (2019).
He, Z. Q. et al. Generation of mouse haploid somatic cells by small molecules for genome-wide genetic screening. Cell Rep. 20, 2227–2237 (2017).
Takahashi, S. et al. Induction of the G2/M transition stabilizes haploid embryonic stem cells. Development 141, 3842–3847 (2014).
He, W. et al. Reduced self-diploidization and improved survival of semi-cloned mice produced from androgenetic haploid embryonic stem cells through overexpression of Dnmt3b. Stem Cell Rep. 10, 477–493 (2018).
Zhang, G. et al. A genetic screen identifies Etl4-deficiency capable of stabilizing the haploidy in embryonic stem cells. Stem Cell Rep. 16, 29–38 (2021).
Yaguchi, K. et al. Uncoordinated centrosome cycle underlies the instability of non-diploid somatic cells in mammals. J. Cell Biol. 217, 2463–2483 (2018).
York, A. G. et al. Instant super-resolution imaging in live cells and embryos via analog image processing. Nat. Methods 10, 1122–1126 (2013).
Xie, N. et al. NAD(+) metabolism: pathophysiologic mechanisms and therapeutic potential. Signal Transduct. Target Ther. 5, 227 (2020).
Hayes, J. D., Dinkova-Kostova, A. T. & Tew, K. D. Oxidative stress in cancer. Cancer Cell 38, 167–197 (2020).
Xiao, W., Wang, R. S., Handy, D. E. & Loscalzo, J. NAD(H) and NADP(H) redox couples and cellular energy metabolism. Antioxid. Redox Signal. 28, 251–272 (2018).
Mullen, A. R. et al. Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature 481, 385–388 (2011).
Metallo, C. M. et al. Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia. Nature 481, 380–384 (2011).
Zhang, D. X., Zou, A. P. & Li, P. L. Ceramide-induced activation of NADPH oxidase and endothelial dysfunction in small coronary arteries. Am. J. Physiol. Heart Circ. Physiol. 284, H605–H612 (2003).
Titov, D. V. et al. Complementation of mitochondrial electron transport chain by manipulation of the NAD+/NADH ratio. Science 352, 231–235 (2016).
Cracan, V., Titov, D. V., Shen, H., Grabarek, Z. & Mootha, V. K. A genetically encoded tool for manipulation of NADP(+)/NADPH in living cells. Nat. Chem. Biol. 13, 1088–1095 (2017).
Yang, R. et al. Identification of purine biosynthesis as an NADH-sensing pathway to mediate energy stress. Nat. Commun. 13, 7031 (2022).
Tsogtbaatar, E., Landin, C., Minter-Dykhouse, K. & Folmes, C. D. L. Energy metabolism regulates stem cell pluripotency. Front. Cell Dev. Biol. 8, 87 (2020).
Handy, D. E. & Loscalzo, J. Redox regulation of mitochondrial function. Antioxid. Redox Signal. 16, 1323–1367 (2012).
Donthamsetty, S. et al. Mitochondrial genome regulates mitotic fidelity by maintaining centrosomal homeostasis. Cell Cycle 13, 2056–2063 (2014).
Cheng, A. et al. A mitotic NADPH upsurge promotes chromosome segregation and tumour progression in aneuploid cancer cells. Nat. Metab. 5, 1141–1158 (2023).
Hochegger, H., Hegarat, N. & Pereira-Leal, J. B. Aurora at the pole and equator: overlapping functions of aurora kinases in the mitotic spindle. Open Biol. 3, 120185 (2013).
Brand, M. D. & Nicholls, D. G. Assessing mitochondrial dysfunction in cells. Biochem. J. 435, 297–312 (2011).
Dikalova, A. E. et al. Therapeutic targeting of mitochondrial superoxide in hypertension. Circ. Res. 107, 106–116 (2010).
Lagouge, M. et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 127, 1109–1122 (2006).
Borra, M. T., Smith, B. C. & Denu, J. M. Mechanism of human SIRT1 activation by resveratrol. J. Biol. Chem. 280, 17187–17195 (2005).
Glazier, D. S. Metabolic level and size scaling of rates of respiration and growth in unicellular organisms. Funct. Ecol. 23, 963–968 (2009).
Cadart, C. & Heald, R. Scaling of biosynthesis and metabolism with cell size. Mol. Biol. Cell 33, https://doi.org/10.1091/mbc.E21-12-0627 (2022).
Miettinen, T. P. & Bjorklund, M. Mitochondrial function and cell size: an allometric relationship. Trends Cell Biol. 27, 393–402 (2017).
Faccenda, D. & Campanella, M. Molecular regulation of the mitochondrial F(1)F(o)-ATPsynthase: physiological and pathological significance of the inhibitory factor 1 (IF(1)). Int. J. Cell Biol. 2012, 367934 (2012).
Rydstrom, J. Mitochondrial NADPH, transhydrogenase and disease. Biochim. Biophys. Acta 1757, 721–726 (2006).
Kampjut, D. & Sazanov, L. A. Structure and mechanism of mitochondrial proton-translocating transhydrogenase. Nature 573, 291–295 (2019).
Hoek, J. B. & Rydstrom, J. Physiological roles of nicotinamide nucleotide transhydrogenase. Biochem. J. 254, 1–10 (1988).
Wise, D. R. et al. Hypoxia promotes isocitrate dehydrogenase-dependent carboxylation of alpha-ketoglutarate to citrate to support cell growth and viability. Proc. Natl. Acad. Sci. USA 108, 19611–19616 (2011).
Lewis, C. A. et al. Tracing compartmentalized NADPH metabolism in the cytosol and mitochondria of mammalian cells. Mol. Cell 55, 253–263 (2014).
Sazanov, L. A. & Jackson, J. B. Proton-translocating transhydrogenase and NAD- and NADP-linked isocitrate dehydrogenases operate in a substrate cycle which contributes to fine regulation of the tricarboxylic acid cycle activity in mitochondria. FEBS Lett. 344, 109–116 (1994).
Niu, X. et al. Cytosolic and mitochondrial NADPH fluxes are independently regulated. Nat. Chem. Biol. 19, 837–845 (2023).
Galli, M. & Morgan, D. O. Cell size determines the strength of the spindle assembly checkpoint during embryonic development. Dev. Cell 36, 344–352 (2016).
Welburn, J. P. et al. Aurora B phosphorylates spatially distinct targets to differentially regulate the kinetochore-microtubule interface. Mol. Cell 38, 383–392 (2010).
DeHart, D. N., Lemasters, J. J. & Maldonado, E. N. Erastin-like anti-warburg agents prevent mitochondrial depolarization induced by free tubulin and decrease lactate formation in cancer cells. SLAS Discov. 23, 23–33 (2018).
Vander Heiden, M. G. et al. Outer mitochondrial membrane permeability can regulate coupled respiration and cell survival. Proc. Natl. Acad. Sci. USA 97, 4666–4671 (2000).
Brenner, C. et al. Bcl-2 and Bax regulate the channel activity of the mitochondrial adenine nucleotide translocator. Oncogene 19, 329–336 (2000).
Dube, H. et al. A mitochondrial-targeted cyclosporin A with high binding affinity for cyclophilin D yields improved cytoprotection of cardiomyocytes. Biochem J. 441, 901–907 (2012).
Abd Radzak, S. M. et al. Insights regarding mitochondrial DNA copy number alterations in human cancer (review). Int. J. Mol. Med. 50, https://doi.org/10.3892/ijmm.2022.5160 (2022).
Jornayvaz, F. R. & Shulman, G. I. Regulation of mitochondrial biogenesis. Essays Biochem. 47, 69–84 (2010).
Holzner, M. et al. The scramblases VMP1 and TMEM41B are required for primitive endoderm specification by targeting WNT signaling. Cell Death Differ https://doi.org/10.1038/s41418-024-01435-x (2024).
Monfort, A., Di Minin, G. & Wutz, A. Screening for factors involved in X chromosome inactivation using haploid ESCs. Methods Mol. Biol. 1861, 1–18 (2018).
Aizawa, E. et al. Haploid mouse germ cell precursors from embryonic stem cells reveal Xist activation from a single X chromosome. Stem Cell Rep. 17, 43–52 (2022).
Fuhrer, T., Heer, D., Begemann, B. & Zamboni, N. High-throughput, accurate mass metabolome profiling of cellular extracts by flow injection-time-of-flight mass spectrometry. Anal. Chem. 83, 7074–7080 (2011).
Buescher, J. M., Moco, S., Sauer, U. & Zamboni, N. Ultrahigh performance liquid chromatography-tandem mass spectrometry method for fast and robust quantification of anionic and aromatic metabolites. Anal. Chem. 82, 4403–4412 (2010).
Pang, Z. et al. MetaboAnalyst 6.0: towards a unified platform for metabolomics data processing, analysis and interpretation. Nucleic Acids Res. 52, W398–W406 (2024).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3, RESEARCH0034 (2002).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Kim, D., Paggi, J. M., Park, C., Bennett, C. & Salzberg, S. L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 37, 907–915 (2019).
Putri, G. H., Anders, S., Pyl, P. T., Pimanda, J. E. & Zanini, F. Analysing high-throughput sequencing data in Python with HTSeq 2.0. Bioinformatics 38, 2943–2945 (2022).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Liu, T. et al. Multi-color live-cell STED nanoscopy of mitochondria with a gentle inner membrane stain. Proc. Natl. Acad. Sci. USA 119, e2215799119 (2022).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Acknowledgements
We acknowledge the Functional Genomics Center Zurich (FGCZ) and the Flow Cytometry Core Facility (FCCF) for technical support. We thank C. Ebner for the help with reagent preparations. We thank F. Mair, I. Vgenopoulou, A. Grison, and J. Corn for their helpful discussions. GDM was supported by the ETH Zurich Postdoctoral Fellowship Program as well as the Marie Curie Actions for People COFUND Program. This work was supported by grants from the Swiss National Science Foundation (SNF grants 31003A_152814/1 and 31003A_175643/1). T.K. was supported by the SNF grant 310030_215716.
Author information
Authors and Affiliations
Contributions
G.D.M., A.W. designed the research. G.D.M., A.R., K.H., T.F., T.K., and N.Z. collected the data. G.D.M., T.F., T.K., and N.Z. conducted software analysis. G.D.M., A.W. supervised the work. G.D.M., A.R., K.H., T.F., T.K., N.Z., and A.W. prepared and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Di Minin, G., Rüegg, A.B., Halter, K. et al. Mitochondrial metabolic imbalance drives diploidization in mouse haploid embryonic stem cells via NADPH overload. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70939-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70939-6