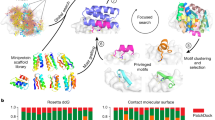
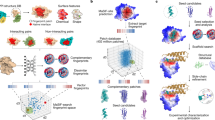
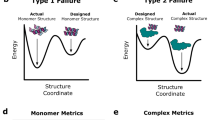
Abstract
The de novo design of small-molecule–binding proteins holds great promise as a potential tool to develop sensors on-demand for arbitrary small molecules. Here we combine deep learning and physics-based methods to generate a family of proteins with diverse and designable pocket geometries, which we employ to computationally design binders for six small-molecule targets. Biophysical characterization of the designed binders reveals nanomolar to low micromolar binding affinities and atomic-level design accuracy. Additionally, we use a cortisol binder to design a chemically induced dimerization (CID) system that enables the construction of a biosensor for cortisol detection. The approach described here demonstrates the potential of the NTF2 fold and deep learning-based protein design in sensor development, paving the way for future platforms to design binders and sensors for small molecules across analytical, environmental, and biomedical applications.
Similar content being viewed by others
Data availability
The X-ray crystallographic structure data generated in this study have been deposited in the Protein Data Bank under the accession IDs 8UQF, 8VFQ, and 8VEZ, corresponding to the cortisol-binding protein and the apixaban-binding proteins, respectively. The deep sequencing data have been deposited in the NCBI database with the accession ID PRJNA1356499. Source data are provided with this paper.
Code availability
The design scripts developed in this study have been deposited on Zenodo [https://doi.org/10.5281/zenodo.17847477].
References
Stanton, B. Z., Chory, E. J. & Crabtree, G. R. Chemically induced proximity in biology and medicine. Science 359 (2018).
Glasgow, A. A. et al. Computational design of a modular protein sense-response system. Science 366, 1024–1028 (2019).
Taylor, N. D. et al. Engineering an allosteric transcription factor to respond to new ligands. Nat. Methods 13, 177–183 (2016).
Tinberg, C. E. et al. Computational design of ligand-binding proteins with high affinity and selectivity. Nature 501, 212–216 (2013).
Bick, M. J. et al. Computational design of environmental sensors for the potent opioid fentanyl. Elife 6 (2017).
Beltrán, J. et al. Rapid biosensor development using plant hormone receptors as reprogrammable scaffolds. Nat. Biotechnol. 40, 1855–1861 (2022).
Dou, J. et al. De novo design of a fluorescence-activating β-barrel. Nature 561, 485–491 (2018).
Polizzi, N. F. et al. De novo design of a hyperstable non-natural protein–ligand complex with sub-Å accuracy. Nat. Chem. 9, 1157–1164 (2017).
Polizzi, N. F. & DeGrado, W. F. A defined structural unit enables de novo design of small-molecule-binding proteins. Science 369, 1227–1233 (2020).
Cochran, F. V. et al. Computational de novo design and characterization of a four-helix bundle protein that selectively binds a nonbiological cofactor. J. Am. Chem. Soc. 127, 1346–1347 (2005).
Farid, T. A. et al. Elementary tetrahelical protein design for diverse oxidoreductase functions. Nat. Chem. Biol. 9, 826–833 (2013).
An, L. et al. Binding and sensing diverse small molecules using shape-complementary pseudocycles. Science 385, 276–282 (2024).
Klima, J. C. et al. Incorporation of sensing modalities into de novo designed fluorescence-activating proteins. Nat. Commun. 12, 856 (2021).
Basanta, B. et al. An enumerative algorithm for de novo design of proteins with diverse pocket structures. Proc. Natl. Acad. Sci. USA. 117, 22135–22145 (2020).
Pan, X. & Kortemme, T. De novo protein fold families expand the designable ligand binding site space. PLoS Comput. Biol. 17, e1009620 (2021).
Dou, J. et al. Sampling and energy evaluation challenges in ligand binding protein design. Protein Sci 26, 2426–2437 (2017).
Eberhardt, R. Y. et al. Filling out the structural map of the NTF2-like superfamily. BMC Bioinformatics 14, 327 (2013).
Bullock, T. L., Clarkson, D. W., Kent, H. M. & Stewart, M. The 1.6 Å Resolution Crystal Structure of Nuclear Transport Factor 2 (NTF2). J. Mol. Biol. 260, 422–431 (1996).
Yeh, A. H.-W. et al. De novo design of luciferases using deep learning. Nature 614, 774–780 (2023).
Dauparas, J. et al. Robust deep learning-based protein sequence design using ProteinMPNN. Science 378, 49–56 (2022).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Hellhammer, D. H., Wüst, S. & Kudielka, B. M. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology 34, 163–171 (2009).
Hanley, J. P. Warfarin reversal. J. Clin. Pathol. 57, 1132–1139 (2004).
Bom, A. et al. A novel concept of reversing neuromuscular block: chemical encapsulation of rocuronium bromide by a cyclodextrin-based synthetic host. Angew. Chem. Int. Ed Engl. 41, 266–270 (2002).
Lu, G. et al. A specific antidote for reversal of anticoagulation by direct and indirect inhibitors of coagulation factor Xa. Nat. Med. 19, 446–451 (2013).
Mathijssen, R. H. J. et al. Irinotecan pharmacokinetics-pharmacodynamics: the clinical relevance of prolonged exposure to SN-38. Br. J. Cancer 87, 144–150 (2002).
Morejón García, G. et al. Generation of monoclonal antibodies against 17α-hydroxyprogesterone for newborn screening of congenital adrenal hyperplasia. Clin. Chim. Acta 485, 311–315 (2018).
Boyken, S. E. et al. De novo design of protein homo-oligomers with modular hydrogen-bond network-mediated specificity. Science 352, 680–687 (2016).
Basanta, B. et al. Introduction of a polar core into the de novo designed protein Top7. Protein Sci 25, 1299–1307 (2016).
Cao, L. et al. Design of protein-binding proteins from the target structure alone. Nature 605, 551–560 (2022).
Krishna, R. et al. Generalized biomolecular modeling and design with RoseTTAFold All-Atom. Science 384, eadl2528 (2024).
Wicky, B. I. M. et al. Hallucinating symmetric protein assemblies. Science 378, 56–61 (2022).
Laudat, M. H. et al. Salivary cortisol measurement: a practical approach to assess pituitary-adrenal function. J. Clin. Endocrinol. Metab. 66, 343–348 (1988).
Yanovski, J. A., Cutler, G. B. Jr, Chrousos, G. P. & Nieman, L. K. The dexamethasone-suppressed corticotropin-releasing hormone stimulation test differentiates mild Cushing’s disease from normal physiology. J. Clin. Endocrinol. Metab. 83, 348–352 (1998).
Park, H., Zhou, G., Baek, M., Baker, D. & DiMaio, F. Force Field Optimization Guided by Small Molecule Crystal Lattice Data Enables Consistent Sub-Angstrom Protein–Ligand Docking. J. Chem. Theory Comput. 17, 2000–2010 (2021).
Tamara, S., den Boer, M. A. & Heck, A. J. R. High-resolution native mass spectrometry. Chem. Rev. 122, 7269–7326 (2022).
Dixon, A. S. et al. NanoLuc Complementation Reporter Optimized for Accurate Measurement of Protein Interactions in Cells. ACS Chem. Biol. 11, 400–408 (2016).
Landrum, G. RDKit: A software suite for cheminformatics, computational chemistry, and predictive modeling. http://www.rdkit.org/RDKit_Overview.pdf.
Wang, J., Wang, W., Kollman, P. A. & Case, D. A. Automatic atom type and bond type perception in molecular mechanical calculations. J. Mol. Graph. Model. 25, 247–260 (2006).
Bannwarth, C. et al. Extended tight-binding quantum chemistry methods. Wiley Interdiscip. Rev. Comput. Mol. Sci. 11 (2021).
Cole, J. C., Korb, O., McCabe, P., Read, M. G. & Taylor, R. Knowledge-Based Conformer Generation Using the Cambridge Structural Database. J. Chem. Inf. Model. 58, 615–629 (2018).
Tosco, P., Stiefl, N. & Landrum, G. Bringing the MMFF force field to the RDKit: implementation and validation. J. Cheminform. 6, 37 (2014).
Zubatyuk, R., Smith, J. S., Leszczynski, J. & Isayev, O. Accurate and transferable multitask prediction of chemical properties with an atoms-in-molecules neural network. Sci Adv 5, eaav6490 (2019).
Maguire, J. B. et al. Perturbing the energy landscape for improved packing during computational protein design. Proteins 89, 436–449 (2021).
Lee, G. R. et al. Design scripts for “Small-molecule binding and sensing with a designed protein family”. Zenodo. https://doi.org/10.5281/zenodo.17847477 (2025).
Leaver-Fay, A. et al. ROSETTA3: an object-oriented software suite for the simulation and design of macromolecules. Methods Enzymol 487, 545–574 (2011).
Tyka, M. D. et al. Alternate states of proteins revealed by detailed energy landscape mapping. J. Mol. Biol. 405, 607–618 (2011).
Klein, J. C. et al. Multiplex pairwise assembly of array-derived DNA oligonucleotides. Nucleic Acids Res 44, e43 (2016).
Benatuil, L., Perez, J. M., Belk, J. & Hsieh, C.-M. An improved yeast transformation method for the generation of very large human antibody libraries. Protein Eng. Des. Sel. 23, 155–159 (2010).
Gietz, R. D. & Schiestl, R. H. Large-scale high-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nat. Protoc. 2, 38–41 (2007).
Jacobs, T. M., Yumerefendi, H., Kuhlman, B. & Leaver-Fay, A. SwiftLib: rapid degenerate-codon-library optimization through dynamic programming. Nucleic Acids Res 43, e34 (2015).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30, 614–620 (2014).
Rubin, A. F. et al. Correction to: A statistical framework for analyzing deep mutational scanning data. Genome Biol 19, 17 (2018).
Qian, J. et al. Accelerating protein design by scaling experimental characterization. bioRxiv https://doi.org/10.1101/2025.08.05.668824 (2025).
Dang, B. et al. SNAC-tag for sequence-specific chemical protein cleavage. Nat. Methods 16, 319–322 (2019).
Kabsch, W. XDS. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr D Struct Biol 75, 861–877 (2019).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Williams, C. J. et al. MolProbity: More and better reference data for improved all-atom structure validation. Protein Sci 27, 293–315 (2018).
Zhang, Y. & Skolnick, J. TM-align: a protein structure alignment algorithm based on the TM-score. Nucleic Acids Res 33, 2302–2309 (2005).
Bennett, N. R. et al. Improving de novo protein binder design with deep learning. Nat. Commun. 14, 2625 (2023).
Marty, M. T. et al. Bayesian deconvolution of mass and ion mobility spectra: from binary interactions to polydisperse ensembles. Anal. Chem. 87, 4370–4376 (2015).
Acknowledgements
We thank B. Basanta for useful discussions on generating NTF2 scaffolds for binder design. This research was supported by the National Institute on Aging (R01AG063845 to I.G., N.R., and B.C.;R01CA240339 to I.G. and N.R.;R0AI160052 to A.B.); the Open Philanthropy Project Improving Protein Design Fund (to G.R.L., S.J.P., D.T., J.D., A.B., H.N., I.G., and B.C.); the Washington Research Foundation, Innovation Fellows Program (to G.R.L.); a Washington Research Foundation Fellowship (S.J.P.); the NIH Pathway to Independence Award R00EB031913 (A.H.-W.Y.); Department of Defense, Defense Threat Reduction Agency (HDTRA1-21-1-0007 to I.A.;HDTRA1-21-1-0038 to I.G.;HDTRA1-19-1-0003 to S.J.P., G.R.L., and D.T.); the Audacious Project at the Institute for Protein Design (A.K., A.B., H.N., H.H.); Microsoft (D.T., J.D., and I.A.); Howard Hughes Medical Institute (G.R.L., D.B., A.B., I.A., and D.R.L.); the Air Force Office of Scientific Research (FA9550-18-1-0297 to S.P.); Novo Nordisk Foundation (NNF18OC0030446 to C.N.); the National Institute of Allergy and Infectious Diseases (NIAID) (Contract No. 75N93022C00036 and HHSN272201700059C to I.A.); the Defense Advanced Research Projects Agency program Harnessing Enzymatic Activity for Lifesaving Remedies (HEALR) under award HR0011-21-2-0012 (to A.B. and I.G.); National Science Foundation grant CHE-1629214 (A.B.); Spark Therapeutics (I.A.); Bill and Melinda Gates Foundation (#OPP1156262 to A.K. and H.N.); AMGEN (I.G.); NovoNordisk (I.G.); the Nordstrom Barrier Institute for Protein Design Directors Fund (I.G.); Dr. Eric and Ms. Wendy Schmidt, and Schmidt Futures funding from Eric and Wendy Schmidt by recommendation of the Schmidt Futures program (I.G.); R35GM118062 (D.R.L); R01EB031172 (D.R.L.); R01EB027793 (D.R.L.); a Ruth L. Kirchstein National Science Research Award Postdoctoral Fellowship (F32 GM133088 to J.A.M.M.). Crystallographic data were collected at the APS and ALS. Advanced Photon Source (APS) Northeastern Collaborative Access Team beamline 24ID-C, is funded by the National Institute of General Medical Sciences from the National Institutes of Health (P30 GM124165). This research used resources of the Advanced Photon Source, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357. The Advanced Light Source (ALS) is supported by the Director, Office of Science, Office of 20 Basic Energy Sciences and US Department of Energy under contract number DE-AC02- 05CH11231. The Berkeley Center for Structural Biology is supported in part by the National Institutes of Health (NIH), National Institute of General Medical Sciences. Native mass spectrometry measurements were provided by the NIH-funded Resource for Native Mass Spectrometry Guided Structural Biology at The Ohio State University (NIH P41 GM128577 awarded to Vicki Wysocki).
Author information
Authors and Affiliations
Contributions
D.B. supervised research. G.R.L., S.J.P., and C.N. conceptualized the small-molecule binder design project and performed analysis on binder screening and characterization data. G.R.L. contributed computational metrics for design approach 1, developed the design approach 2 pipeline, experimentally characterized the designs, and participated in LigandMPNN development. S.J.P. redesigned NTF2 scaffolds, formulated design approach 1, and experimentally characterized the designs. C.N. developed the design approach 1 pipeline. A.H-W.Y. conceptualized and characterized the design of chemical-induced dimerizations. S.J.P. and G.R.L. assisted with designing and characterizing cortisol-induced dimers. G.R.L., S.J.P., A.H-W.Y., and D.B. wrote the manuscript. D.T. and I.A. contributed to NTF2 scaffold design. J.D. provided LigandMPNN. J.A.M.M. performed the synthesis of biotinylated ligands under D.R.L.’s supervision. S.J.P., A.K., A.B., and H.N. performed crystallography experiments, data collection, and determined the crystal structure of the cortisol binder. G.R.L., S.J.P., A.K., A.B., E.B., and B.S. performed crystallography experiments, data collection, and determined the crystal structure of the apixaban binder. I.G., D.V., and N.R. helped with yeast library assembly and deep sequencing. H.L.H. assisted with protein purification and BLI experiments. B.C. provided guidance on miniprotein design for the cortisol-induced dimerization system. H.K.H. helped with NGS data analysis.
Corresponding authors
Ethics declarations
Competing interests
G.R.L., S.J.P., C.N., A.H-W.Y., and D.B. are co-inventors on several provisional patent applications submitted by the University of Washington pertaining to the designed protein sequences generated in this work (application numbers: US 63/599,194 and PCT 18/945,961). The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, G.R., Pellock, S.J., Norn, C. et al. Small-molecule binding and sensing with a designed protein family. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70953-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70953-8