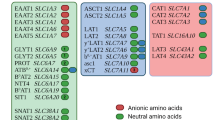
Abstract
The transport of amino acids across cell membranes is essential for metabolism, neuronal signalling, and immune system function. The amino acid polyamine organocation (APC) superfamily controls amino acid transport via mechanisms including amino acid exchange, facilitative diffusion, and sodium- or proton-coupled transport. Although many mammalian APC members functioning as exchangers and sodium-coupled systems have been identified, the mechanisms underlying pH-regulated amino acid transport in mammalian cells remain unclear. Here, we show that the plasma membrane amino acid transporter SLC7A4 is regulated by low extracellular pH and functions as a leucine transporter in human cells. Using Cryo-EM structures of the plant homologue, CAT4, from Arabidopsis thaliana in outward-open apo and L-ornithine-bound states, as well as transport assays and molecular dynamics simulations based on homology models of the human transporter, we identify residues responsible for amino acid selectivity that supports an allosteric mechanism linking ligand recognition to pH regulation. This mechanism is consistent with an evolutionary link to proton-coupled prokaryotic homologues. Overall, our findings provide a structural and functional basis for pH-gated leucine transport by the human SLC7A4 transporter and provides a framework for understanding amino acid selectivity within the wider SLC7 family.
Similar content being viewed by others
Data availability
Atomic coordinates for SLC7A4 have been deposited in the Protein Data Bank under accession codes: 9HJK (Apo with Syb), 9SP8 (L-Orn with Syb) and 9SQH (Apo without Syb). The cryo-EM maps have been deposited in the Electron Microscopy Data Bank (EMDB) under accession codes: EMD-52217, EMD-55065 and EMD-55110. Source data are provided with this paper.
Code availability
The processed molecular dynamics simulations trajectories have been uploaded to Zenodo: (https://doi.org/10.5281/zenodo.17184930).
References
Broer, S. & Gauthier-Coles, G. Amino acid homeostasis in mammalian cells with a focus on amino acid transport. J. Nutr. 152, 16–28 (2022).
Wang, W. & Zou, W. Amino acids and their transporters in T cell immunity and cancer therapy. Mol. Cell 80, 384–395 (2020).
Cui, Z., Joiner, A. M. N., Jansen, R. M. & Hurley, J. H. Amino acid sensing and lysosomal signaling complexes. Curr. Opin. Struct. Biol. 79, 102544 (2023).
Bar-Peled, L. & Sabatini, D. M. Regulation of mTORC1 by amino acids. Trends Cell Biol. 24, 400–406 (2014).
Yuan, H. et al. Lysine catabolism reprograms tumour immunity through histone crotonylation. Nature 617, 818–826 (2023).
Hosios, A. M. et al. Amino acids rather than glucose account for the majority of cell mass in proliferating mammalian cells. Dev. Cell 36, 540–549 (2016).
Lieu, E. L., Nguyen, T., Rhyne, S. & Kim, J. Amino acids in cancer. Exp. Mol. Med 52, 15–30 (2020).
Kandasamy, P., Gyimesi, G., Kanai, Y. & Hediger, M. A. Amino acid transporters revisited: New views in health and disease. Trends Biochem Sci. 43, 752–789 (2018).
Verrey, F. et al. CATs and HATs: the SLC7 family of amino acid transporters. Pflug. Arch. 447, 532–542 (2004).
Jin, C. et al. Pharmacological and structural insights into nanvuranlat, a selective LAT1 (SLC7A5) inhibitor, and its N-acetyl metabolite with implications for cancer therapy. Sci. Rep. 15, 2903 (2025).
Markowicz-Piasecka, M., Huttunen, J., Montaser, A. & Huttunen, K. M. Hemocompatible LAT1-inhibitor can induce apoptosis in cancer cells without affecting brain amino acid homeostasis. Apoptosis 25, 426–440 (2020).
Kim, C. S. et al. BCH, an inhibitor of system L amino acid transporters, induces apoptosis in cancer cells. Biol. Pharm. Bull. 31, 1096–1100 (2008).
Yun, D. W. et al. JPH203, an L-type amino acid transporter 1-selective compound, induces apoptosis of YD-38 human oral cancer cells. J. Pharm. Sci. 124, 208–217 (2014).
Stockwell, B. R. et al. Ferroptosis: a regulated cell death nexus linking metabolism, redox biology, and disease. Cell 171, 273–285 (2017).
Tarlungeanu, D. C. et al. Impaired amino acid transport at the blood brain barrier is a cause of autism spectrum disorder. Cell 167, 1481–1494 e18 (2016).
Puris, E., Gynther, M., Auriola, S. & Huttunen, K. M. L-Type amino acid transporter 1 as a target for drug delivery. Pharm. Res 37, 88 (2020).
Closs, E. I., Boissel, J. P., Habermeier, A. & Rotmann, A. Structure and function of cationic amino acid transporters (CATs). J. Membr. Biol. 213, 67–77 (2006).
Vastermark, A., Wollwage, S., Houle, M. E., Rio, R. & Saier, M. H. Jr Expansion of the APC superfamily of secondary carriers. Proteins 82, 2797–2811 (2014).
Shi, Y. Common folds and transport mechanisms of secondary active transporters. Annu Rev. Biophys. 42, 51–72 (2013).
Yamashita, A., Singh, S. K., Kawate, T., Jin, Y. & Gouaux, E. Crystal structure of a bacterial homologue of Na+/Cl-dependent neurotransmitter transporters. Nature 437, 215–223 (2005).
Forrest, L. R. Structural symmetry in membrane proteins. Annu Rev. Biophys. 44, 311–337 (2015).
Lee, Y. et al. Structural basis of anticancer drug recognition and amino acid transport by LAT1. Nat. Commun. 16, 1635 (2025).
Li, Y. et al. Cryo-EM structure of the human Asc-1 transporter complex. Nat. Commun. 15, 3036 (2024).
Dai, L. et al. Structural basis for the substrate recognition and transport mechanism of the human y(+)LAT1-4F2hc transporter complex. Sci. Adv. 11, eadq0558 (2025).
Dai, L. et al. Structural insights into the human system y(+)L amino acid transporter complex. Structure 33, 1224–1232 e5 (2025).
Yan, R. et al. Cryo-EM structure of the human heteromeric amino acid transporter b(0,+)AT-rBAT. Sci. Adv. 6, eaay6379 (2020).
Yan, R., Zhou, J., Li, Y., Lei, J. & Zhou, Q. Structural insight into the substrate recognition and transport mechanism of the human LAT2-4F2hc complex. Cell Discov. 6, 82 (2020).
Yan, R., Zhao, X., Lei, J. & Zhou, Q. Structure of the human LAT1-4F2hc heteromeric amino acid transporter complex. Nature 568, 127–130 (2019).
Parker, J. L. et al. Molecular basis for redox control by the human cystine/glutamate antiporter system xc-. Nat. Commun. 12, 7147 (2021).
Rosell, A. et al. Structural bases for the interaction and stabilization of the human amino acid transporter LAT2 with its ancillary protein 4F2hc. Proc. Natl. Acad. Sci. USA 111, 2966–2971 (2014).
Boll, M., Daniel, H. & Gasnier, B. The SLC36 family: proton-coupled transporters for the absorption of selected amino acids from extracellular and intracellular proteolysis. Pflügers Arch.: Eur. J. Physiol. 447, 776–779 (2004).
Fan, S. J. & Goberdhan, D. C. I. PATs and SNATs: amino acid sensors in disguise. Front Pharm. 9, 640 (2018).
Jungnickel, K. E. J., Parker, J. L. & Newstead, S. Structural basis for amino acid transport by the CAT family of SLC7 transporters. Nat. Commun. 9, 550 (2018).
Wolf, S. et al. Expression of solute carrier 7A4 (SLC7A4) in the plasma membrane is not sufficient to mediate amino acid transport activity. Biochem J. 364, 767–775 (2002).
Wu, Z., Alibay, I., Newstead, S. & Biggin, P. C. Proton Control of Transitions in an Amino Acid Transporter. Biophys. J. 117, 1342–1351 (2019).
Tao, Y. et al. Structure of a eukaryotic SWEET transporter in a homotrimeric complex. Nature 527, 259–263 (2015).
Vekony, N., Wolf, S., Boissel, J. P., Gnauert, K. & Closs, E. I. Human cationic amino acid transporter hCAT-3 is preferentially expressed in peripheral tissues. Biochemistry 40, 12387–12394 (2001).
Closs, E. I., Graf, P., Habermeier, A., Cunningham, J. M. & Forstermann, U. Human cationic amino acid transporters hCAT-1, hCAT-2A, and hCAT-2B: three related carriers with distinct transport properties. Biochemistry 36, 6462–6468 (1997).
Su, Y. H., Frommer, W. B. & Ludewig, U. Molecular and functional characterization of a family of amino acid transporters from Arabidopsis. Plant Physiol. 136, 3104–3113 (2004).
Schiöth, H. B., Roshanbin, S., Hägglund, M. G. A. & Fredriksson, R. Evolutionary origin of amino acid transporter families SLC32, SLC36 and SLC38 and physiological, pathological and therapeutic aspects. Mol. Asp. Med. 34, 571–585 (2013).
Hammes, U. Z., Nielsen, E., Honaas, L. A., Taylor, C. G. & Schachtman, D. P. AtCAT6, a sink-tissue-localized transporter for essential amino acids in Arabidopsis. Plant J. 48, 414–426 (2006).
Carter, C. et al. The vegetative vacuole proteome of Arabidopsis thaliana reveals predicted and unexpected proteins. Plant Cell 16, 3285–3303 (2004).
Yang, H., Postel, S., Kemmerling, B. & Ludewig, U. Altered growth and improved resistance of Arabidopsis against Pseudomonas syringae by overexpression of the basic amino acid transporter AtCAT1. Plant Cell Environ. 37, 1404–1414 (2014).
Okumoto, S. & Pilot, G. Amino acid export in plants: a missing link in nitrogen cycling. Mol. Plant 4, 453–463 (2011).
Hammes, U. Z., Meier, S., Dietrich, D., Ward, J. M. & Rentsch, D. Functional properties of the arabidopsis peptide transporters AtPTR1 and AtPTR5. J. Biol. Chem. 285, 39710–39717 (2010).
Sperandeo, M. P. et al. The gene encoding a cationic amino acid transporter (SLC7A4) maps to the region deleted in the velocardiofacial syndrome. Genomics 49, 230–236 (1998).
Xia, L. et al. Structural insights into cationic amino acid transport and viral receptor engagement by CAT1. Nat. Commun. 17, 1108 (2025).
Ye, M. et al. Amino acid and viral binding by the high-affinity Cationic Amino acid Transporter 1 (CAT1) from Mus musculus. Nat. Commun. https://doi.org/10.1038/s41467-026-69421-0 (2026).
Xie, Y. et al. Structures and an activation mechanism of human potassium-chloride cotransporters. Sci. Adv. 6, eabc5883 (2020).
Yang, X., Wang, Q. & Cao, E. Structure of the human cation-chloride cotransporter NKCC1 determined by single-particle electron cryo-microscopy. Nat. Commun. 11, 1016 (2020).
Jeckelmann, J. M. et al. Structure, function and pharmacology of SLC7 family members and homologues. Chim. (Aarau) 76, 1011–1018 (2022).
Dickens, D. et al. Modulation of LAT1 (SLC7A5) transporter activity and stability by membrane cholesterol. Sci. Rep. 7, 43580 (2017).
Scanga, R. et al. Impact of 7-ketocholesterol on the function and stability of the LAT1 transporter. Biochem Pharm. 239, 117075 (2025).
Hutchinson, K. & Schlessinger, A. Comprehensive characterization of LAT1 cholesterol-binding sites. J. Chem. Theory Comput 20, 3349–3358 (2024).
Lee, Y. et al. Cryo-EM structure of the human L-type amino acid transporter 1 in complex with glycoprotein CD98hc. Nat. Struct. Mol. Biol. 26, 510–517 (2019).
Yan, R. et al. Mechanism of substrate transport and inhibition of the human LAT1-4F2hc amino acid transporter. Cell Discov. 7, 16 (2021).
Yang, H. et al. Structural insights into the substrate transport mechanism of the amino acid transporter complex. J. Biol. Chem. 301, 110569 (2025).
Krishnamurthy, H. & Gouaux, E. X-ray structures of LeuT in substrate-free outward-open and apo inward-open states. Nature 481, 469–474 (2012).
Gao, X. et al. Mechanism of substrate recognition and transport by an amino acid antiporter. Nature 463, 828–832 (2010).
Errasti-Murugarren, E. et al. L amino acid transporter structure and molecular bases for the asymmetry of substrate interaction. Nat. Commun. 10, 1807 (2019).
Bansal, M., Kumar, S. & Velavan, R. HELANAL: a program to characterize helix geometry in proteins. J. Biomol. Struct. Dyn. 17, 811–819 (2000).
Wiedmer, T. et al. Metabolic mapping of the human solute carrier superfamily. Mol. Syst. Biol. 21, 560–598 (2025).
Yang, H., Krebs, M., Stierhof, Y. D. & Ludewig, U. Characterization of the putative amino acid transporter genes AtCAT2, 3 &4: the tonoplast localized AtCAT2 regulates soluble leaf amino acids. J. Plant Physiol. 171, 594–601 (2014).
Winter, G., Todd, C. D., Trovato, M., Forlani, G. & Funck, D. Physiological implications of arginine metabolism in plants. Front Plant Sci. 6, 534 (2015).
Blazquez, M. A. Polyamines: their role in plant development and stress. Annu Rev. Plant Biol. 75, 95–117 (2024).
Shaffer, P. L., Goehring, A., Shankaranarayanan, A. & Gouaux, E. Structure and mechanism of a Na+-independent amino acid transporter. Sci. (N. Y., NY) 325, 1010–1014 (2009).
Kratochvil, H. T. et al. Transient water wires mediate selective proton transport in designed channel proteins. Nat. Chem. 15, 1012–1021 (2023).
Nayak, S. R. et al. Cryo-EM structure of GABA transporter 1 reveals substrate recognition and transport mechanism. Nat. Struct. Mol. Biol. 30, 1023–1032 (2023).
Fang, Y. et al. Structure of a prokaryotic virtual proton pump at 3.2 A resolution. Nature 460, 1040–1043 (2009).
Olsson, M. H. M., Sondergaard, C. R., Rostkowski, M. & Jensen, J. H. PROPKA3: consistent treatment of internal and surface residues in empirical pK(a) predictions. J. Chem. Theory Comput. 7, 525–537 (2011).
Rullo-Tubau, J. et al. Structure and mechanisms of transport of human Asc1/CD98hc amino acid transporter. Nat. Commun. 15, 2986 (2024).
Wiedmer, T. et al. Accelerating SLC transporter research: streamlining knowledge and validated tools. Clin. Pharm. Ther. 112, 439–442 (2022).
Goldmann, U. et al. Data- and knowledge-derived functional landscape of human solute carriers. Mol. Syst. Biol. 21, 599–631 (2025).
Jansson, T. Amino acid transporters in the human placenta. Pediatr. Res 49, 141–147 (2001).
Christensen, H. L. et al. The choroid plexus sodium-bicarbonate cotransporter NBCe2 regulates mouse cerebrospinal fluid pH. J. Physiol. 596, 4709–4728 (2018).
Bernardino, R. L., Carrageta, D. F., Sousa, M., Alves, M. G. & Oliveira, P. F. pH and male fertility: making sense on pH homeodynamics throughout the male reproductive tract. Cell Mol. Life Sci. 76, 3783–3800 (2019).
Parker, J. L. & Newstead, S. Method to increase the yield of eukaryotic membrane protein expression in Saccharomyces cerevisiae for structural and functional studies. Protein Sci. 23, 1309–1314 (2014).
Drew, D. et al. GFP-based optimization scheme for the overexpression and purification of eukaryotic membrane proteins in Saccharomyces cerevisiae. Nat. Protoc. 3, 784–798 (2008).
Zimmermann, I. et al. Generation of synthetic nanobodies against delicate proteins. Nat. Protoc. 15, 1707–1741 (2020).
Caesar, J. et al. SIMPLE 3.0. Stream single-particle cryo-EM analysis in real time. J. Struct. Biol. X 4, 100040 (2020).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. Elife 7, e42166 (2018).
Mirdita, M. et al. ColabFold: making protein folding accessible to all. Nat. Methods 19, 679–682 (2022).
Emsley, P. Tools for ligand validation in Coot. Acta Crystallogr D. Struct. Biol. 73, 203–210 (2017).
Afonine, P. V. et al. Real-space refinement in PHENIX for cryo-EM and crystallography. Acta Crystallogr D. Struct. Biol. 74, 531–544 (2018).
Long, F. et al. AceDRG: a stereochemical description generator for ligands. Acta Crystallogr D. Struct. Biol. 73, 112–122 (2017).
Croll, T. I. ISOLDE: a physically realistic environment for model building into low-resolution electron-density maps. Acta Crystallogr D. Struct. Biol. 74, 519–530 (2018).
Pettersen, E. F. et al. UCSF ChimeraX: structure visualization for researchers, educators, and developers. Protein Sci. 30, 70–82 (2021).
Parker, J. L., Mindell, J. A. & Newstead, S. Thermodynamic evidence for a dual transport mechanism in a POT peptide transporter. Elife 3, e04273 (2014).
Eswar, N. et al. Comparative protein structure modeling using Modeller. Curr. Protoc. Bioinformatics. 5, Unit-5 6 (2006).
Wu, E. L. et al. CHARMM-GUI Membrane Builder toward realistic biological membrane simulations. J. Comput Chem. 35, 1997–2004 (2014).
Lindorff-Larsen, K. et al. Improved side-chain torsion potentials for the Amber ff99SB protein force field. Proteins 78, 1950–1958 (2010).
Jämbeck, J. P. M. & Lyubartsev, A. P. An extension and further validation of an all-atomistic force field for biological membranes. J. Chem. Theory Comput. 8, 2938–2948 (2012).
Horn, A. H. A consistent force field parameter set for zwitterionic amino acid residues. J. Mol. Model 20, 2478 (2014).
Sousa da Silva, A. W. & Vranken, W. F. ACPYPE - AnteChamber PYthon Parser interfacE. BMC Res Notes 5, 367 (2012).
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79, 926–935 (1983).
Abraham, M. J. et al. GROMACS: high performance molecular simulations through multi-level parallelism from laptops to supercomputers. SoftwareX 1-2, 19–25 (2015).
Essmann, U. et al. A smooth particle mesh Ewald method. J. Chem. Phys. 103, 8577–8593 (1995).
Berendsen, H. J., Postma, J. P. M., van Gunsteren, W. F., DiNola, A. & Haak, J. R. Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684–3690 (1984).
Parrinello, M. & Rahman, A. Polymorphic transitions in single crystals: a new molecular dynamics method. J. Appl. Phys. 52, 7182–7190 (1981).
Bussi, G., Donadio, D. & Parrinello, M. Canonical sampling through velocity rescaling. J. Chem. Phys. 126, 014101(2007).
Michaud-Agrawal, N., Denning, E. J., Woolf, T. B. & Beckstein, O. MDAnalysis: a toolkit for the analysis of molecular dynamics simulations. J. Comput Chem. 32, 2319–2327 (2011).
Bouysset, C. & Fiorucci, S. ProLIF: a library to encode molecular interactions as fingerprints. J. Cheminform 13, 72 (2021).
Acknowledgements
This research was supported by Wellcome awards (215519/Z/19/Z & 219531/Z/19/Z) to SN and UKRI BBSRC award BB/Z517215/1 to JLP and SN. Computing was supported via the Advanced Research Computing facility, Oxford, the EPSRC ARCHER2 UK National Supercomputing Service and JADE (EP/X035603/1), granted via the High-End Computing Consortium for Biomolecular Simulation (HECBioSim-https://www.hecbiosim.ac.uk), supported by EPSRC (EP/X035603/1) to PCB. DK was supported by a BBSRC studentship (BB/ M011224/1) and an Onassis Foundation PhD scholarship award (F ZO 035-1/2018-2019). AB was supported by a BBSRC studentship (BB/T008784/1). The authors gratefully acknowledge the Micron Bioimaging Facility for their support & assistance in this work.
Author information
Authors and Affiliations
Contributions
D.K., J.L.P. and S.N. conceived the project. D.K. and A.B. performed all cloning, protein preparation and transport assays. D.K., T.K. and Y.C.Z performed all cryo-EM sample processing, data collection and image analysis. D.K. and S.N. constructed the atomic models with assistance from Y.C.Z. D.K., S.L. and P.C.B. performed all molecular dynamics simulations and analysis. D.K. and S.N. wrote the manuscript and prepared figures with contributions and discussions from J.L.P.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Marcel Bermudez and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kolokouris, D., Bothra, A., Kato, T. et al. Structural basis for pH-responsive amino acid transport via SLC7A4.. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70956-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70956-5