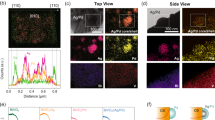
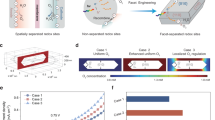
Abstract
Improvement of solar-to-chemical energy conversion in photocatalytic CO2 reduction remains fundamentally constrained by insufficient utilization of solar energy, particularly low-energy photons. Here we report a nanoscale greenhouse structure (Bi@Fe2O3) that enables cascaded utilization of full solar spectrum. The Bi nanocore primarily absorbs low-energy photons, generating localized nanoheating via non-radiative heating through localized surface plasmon resonance effects and energetic hot electrons. Meanwhile, the oxygen-vacancy-rich loose Fe2O3 shell absorbs high-energy photons and serves as the catalytic bed, where injected hot electrons and confined heat synergistically promote CO2 activation and deep hydrogenation. Benefiting from the interplay between photochemical and photothermal effects, the system achieves a CH4 production rate of 273.81 μmol g–1 h–1 with 98.60% selectivity and an apparent quantum efficiency of 0.64% at 850 nm illumination without any external heating or sacrificial agents. This work paves a way for the efficient utilization of the entire solar spectrum.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information files. Data are available from the corresponding authors upon request. Source data are provided with this paper.
References
Lei, T. et al. Global iron and steel plant CO2 emissions and carbon-neutrality pathways. Nature 622, 514–520 (2023).
Bonchio, M. et al. Best practices for experiments and reporting in photocatalytic CO2 reduction. Nat. Catal. 6, 657–665 (2023).
Wu, Y. A. et al. Facet-dependent active sites of a single Cu2O particle photocatalyst for CO2 reduction to methanol. Nat. Energy 4, 957–968 (2019).
Kang, X., He, Z., Wang, F., Liu, Y. & Guo, L. Decrypting the controlled product selectivity over tunable Ni─Co bimetallic alloy for photoreduction CO2. Adv. Funct. Mater. 35, 2419802 (2024).
An, J., Ge, S., Wang, G. & Fu, H. Photocontrolled heterojunctions constructed from holmium single atom modified Mg1.2Ti1.8O5/g-C3N4 with enhanced photocatalytic CO2 conversion. Energy Environ. Sci. 17, 5039–5047 (2024).
Ma, Y. et al. Selective photocatalytic CO2 reduction in aerobic environment by microporous Pd-porphyrin-based polymers coated hollow TiO2. Nat. Commun. 13, 1400 (2022).
Feng, X. et al. Unlocking bimetallic active sites via a desalination strategy for photocatalytic reduction of atmospheric carbon dioxide. Nat. Commun. 13, 2146 (2022).
Li, Y., Li, B., Zhang, D., Cheng, L. & Xiang, Q. Crystalline carbon nitride supported copper single atoms for photocatalytic CO2 reduction with nearly 100% CO selectivity. ACS Nano 14, 10552–10561 (2020).
Jiang, L. et al. An innovative CuxAg50−x/UiO66-NH2 photocatalyst prepared using a dual ship bottling strategy for photocatalytic CO2 reduction: controlled product selectivity and pathways. Energy Environ. Sci. 17, 8228–8242 (2024).
Karmakar, S. et al. Confining charge-transfer complex in a metal-organic framework for photocatalytic CO2 reduction in water. Nat. Commun. 14, 4508 (2023).
Liu, Y. & Zhou, Y. Synergistic mechanisms of dual-metal single-atoms catalysts for promoting CO2 photoreduction: bifunctional, intersite distance, and electronic effects. ACS Catal. 15, 10957–10970 (2025).
Wang, J., Li, L., Wei, R. & Dong, R. Quantum dot-based micromotors with NIR-I light photocatalytic propulsion and NIR-II fluorescence. ACS Appl. Mater. Interfaces 14, 48967–48975 (2022).
Kamath, A., Melnychuk, C. & Guyot-Sionnest, P. Toward bright mid-infrared emitters: thick-shell n-type HgSe/CdS nanocrystals. J. Am. Chem. Soc. 143, 19567–19575 (2021).
Jain, P. K., Huang, X., El-Sayed, I. H. & El-Sayed, M. A. Noble metals on the nanoscale: optical and photothermal properties and some applications in imaging, sensing, biology, and medicine. Acc. Chem. Res. 41, 1578–1586 (2008).
Eustis, S. & El-Sayed, M. A. Why gold nanoparticles are more precious than pretty gold: noble metal surface plasmon resonance and its enhancement of the radiative and nonradiative properties of nanocrystals of different shapes. Chem. Soc. Rev. 35, 209–217 (2006).
Olson, J. et al. Optical characterization of single plasmonic nanoparticles. Chem. Soc. Rev. 44, 40–57 (2015).
Jiang, X. et al. Plasmonic active “Hot Spots”-confined photocatalytic CO2 reduction with high selectivity for CH4 production. Adv. Mater. 34, 2109330 (2022).
Cortie, M. B. & McDonagh, A. M. Synthesis and optical properties of hybrid and alloy plasmonic nanoparticles. Chem. Rev. 111, 3713–3735 (2011).
Skrabalak, S. E., Au, L., Li, X. & Xia, Y. Facile synthesis of Ag nanocubes and Au nanocages. Nat. Protoc. 2, 2182–2190 (2007).
Hu, C. et al. Surface plasmon enabling nitrogen fixation in pure water through a dissociative mechanism under mild conditions. J. Am. Chem. Soc. 141, 7807–7814 (2019).
Guo, J. et al. Boosting hot electrons in hetero-superstructures for plasmon-enhanced catalysis. J. Am. Chem. Soc. 139, 17964–17972 (2017).
Zhang, X. et al. Product selectivity in plasmonic photocatalysis for carbon dioxide hydrogenation. Nat. Commun. 8, 14542 (2017).
Zheng, Z., Tachikawa, T. & Majima, T. Plasmon-enhanced formic acid dehydrogenation using anisotropic Pd–Au nanorods studied at the single-particle level. J. Am. Chem. Soc. 137, 948–957 (2015).
Mukherjee, S. et al. Hot electrons do the impossible: plasmon-induced dissociation of H2 on Au. Nano Lett. 13, 240–247 (2012).
Yu, S. & Jain, P. K. Plasmonic photosynthesis of C1–C3 hydrocarbons from carbon dioxide assisted by an ionic liquid. Nat. Commun. 10, 2022 (2019).
Yu, S., Wilson, A. J., Heo, J. & Jain, P. K. Plasmonic control of multi-electron transfer and C–C coupling in visible-light-driven CO2 reduction on Au nanoparticles. Nano Lett. 18, 2189–2194 (2018).
Zhang, Y. et al. Surface-plasmon-driven hot electron photochemistry. Chem. Rev. 118, 2927–2954 (2017).
Polte, J. Nucleation and growth of gold nanoparticles studied via in situ small angle X-ray scattering at millisecond time resolution. ACS Nano 4, 1076–1082 (2010).
Zhan, C. et al. From plasmon-enhanced molecular spectroscopy to plasmon-mediated chemical reactions. Nat. Rev. Chem. 2, 216–230 (2018).
Clavero, C. Plasmon-induced hot-electron generation at nanoparticle/metal-oxide interfaces for photovoltaic and photocatalytic devices. Nat. Photonics 8, 95–103 (2014).
Swearer, D. F. et al. Heterometallic antenna−reactor complexes for photocatalysis. Proc. Natl. Acad. Sci. USA 113, 8916–8920 (2016).
Mubeen, S. et al. An autonomous photosynthetic device in which all charge carriers derive from surface plasmons. Nat. Nanotechnol. 8, 247–251 (2013).
Griffiths, J. et al. Resolving sub-angstrom ambient motion through reconstruction from vibrational spectra. Nat. Commun. 12, 6759 (2021).
Lin, Q. Optical suppression of energy barriers in single molecule-metal binding. Sci. Adv. 8, eabp9285 (2022).
Cai, M. et al. Greenhouse-inspired supra-photothermal CO2 catalysis. Nat. Energy 6, 807–814 (2021).
Zhang, N. et al. Enhanced selective catalytic reduction of NO with NH3 over homoatomic dinuclear sites in defective α-Fe2O3. Chem. Eng. J. 426, 131845 (2021).
Cui, X. et al. Photothermal nanomaterials: a powerful light-to-heat converter. Chem. Rev. 123, 6891–6952 (2023).
Gao, W. et al. Photo-driven syngas conversion to lower olefins over oxygen-decorated Fe5C2 catalyst. Chem. 4, 2917–2928 (2018).
Chen, Y.-Z. et al. Singlet oxygen-engaged selective photo-oxidation over Pt nanocrystals/porphyrinic MOF: the roles of photothermal effect and Pt electronic state. J. Am. Chem. Soc. 139, 2035–2044 (2017).
Mao, C. et al. Beyond the thermal equilibrium limit of ammonia synthesis with dual temperature zone catalyst powered by solar light. Chem. 5, 2702–2717 (2019).
Wang, K. et al. Modeling the effect of Cu doped TiO2 with carbon dots on CO2 methanation by H2O in a photo-thermal system. Appl. Catal. B Environ. 256, 117780 (2019).
Li, J. et al. Sunlight induced photo-thermal synergistic catalytic CO2 conversion via localized surface plasmon resonance of MoO3−x. J. Mater. Chem. A 7, 2821–2830 (2019).
Wang, W. et al. Photocatalytic C–C coupling from carbon dioxide reduction on copper oxide with mixed-valence copper(I)/copper(II). J. Am. Chem. Soc. 143, 2984–2993 (2021).
Yang, J. et al. Dynamic behavior of single-atom catalysts in electrocatalysis: identification of Cu–N3 as an active site for the oxygen reduction reaction. J. Am. Chem. Soc. 143, 14530–14539 (2021).
Wang, L. et al. Selective reduction of CO to acetaldehyde with CuAg electrocatalysts. Proc. Natl. Acad. Sci. USA 117, 12572–12575 (2020).
Chang, X., Malkani, A., Yang, X. & Xu, B. Mechanistic Insights into electroreductive C–C coupling between CO and acetaldehyde into multicarbon products. J. Am. Chem. Soc. 142, 2975–2983 (2020).
Garza, A. J., Bell, A. T. & Head-Gordon, M. Mechanism of CO2 reduction at copper surfaces: pathways to C2 products. ACS Catal. 8, 1490–1499 (2018).
Peng, B., Zhang, K., Sun, Y., Han, B. & He, M. Role of water in green carbon science. J. Am. Chem. Soc. 147, 13083–13100 (2025).
Zhu, J. et al. Asymmetric triple-atom sites confined in ternary oxide enabling selective CO2 photothermal reduction to acetate. J. Am. Chem. Soc. 143, 131845 (2021).
Shangguan, W. et al. Molecular-level insight into photocatalytic CO2 reduction with H2O over Au nanoparticles by interband transitions. Nat. Commun. 13, 3894 (2022).
Wu, X. et al. Identification of the active sites on metallic MoO2−x nano-sea-urchin for atmospheric CO2 photoreduction under UV, visible, and near-infrared light illumination. Angew. Chem. Int. Ed. 62, e202213124 (2022).
Ma, Z. et al. Selectivity reversal of photocatalytic CO2 reduction by Pt loading. Catal. Sci. Technol. 8, 5129–5132 (2018).
Zhang, R. et al. Photocatalytic oxidative dehydrogenation of ethane using CO2 as a soft oxidant over Pd/TiO2 catalysts to C2H4 and syngas. ACS Catal. 8, 9280–9286 (2018).
Cui, Y. et al. A chiral mesostructured photocatalyst for efficient solar-driven CO2 reduction to ethanol. Nat. Synth. 5, 117–128 (2025).
Gurudayal, G. et al. Efficient solar-driven electrochemical CO2 reduction to hydrocarbons and oxygenates. Energy Environ. Sci. 10, 2222–2230 (2017).
Furthmu¨ller, G. K. J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 169–186 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Joubert, G. K. D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Hammer, B., Hansen, L. B. & Nørskov, J. K. Improved adsorption energetics within density-functional theory using revised Perdew–Burke–Ernzerhof functionals. Phys. Rev. B 59, 7413–7421 (1999).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Wang, V., Xu, N., Liu, J.-C., Tang, G. & Geng, W.-T. VASPKIT: a user-friendly interface facilitating high-throughput computing and analysis using VASP code. Comput. Phys. Commun. 267, 108033 (2021).
Zhang, Z. et al. Cavity-confined Au@Cu2O yolk-shell nanoreactors enable switchable CH4/C2H4 selectivity. Nat. Commun. 16, 7559 (2025).
Tsao, C.-W. et al. Dual-plasmonic Au@Cu7S4 yolk@shell nanocrystals for photocatalytic hydrogen production across visible to near infrared spectral region. Nat. Commun. 15, 413 (2024).
Sun, Z. et al. Atomic dispersed Co on NC@Cu core-shells for solar seawater splitting. Adv. Mater. 36, 2406088 (2024).
Xu, Z. et al. Exploring the mechanisms of charge transfer and identifying active sites in the hydrogen evolution reaction using hollow C@MoS2-Au@CdS nanostructures as photocatalysts. Adv. Mater. 37, 2501091 (2025).
Wang, X. et al. Efficient electrically powered CO2-to-ethanol via suppression of deoxygenation. Nat. Energy 5, 478–486 (2020).
Acknowledgements
This work was supported by the Basic Science Center Program for Ordered Energy Conversion of the National Natural Science Foundation of China (No. 52488201) and the 2025 Annual Science and Technology Support Project of Daqingshan Laboratory (No. 2025KYPT0188). The authors also acknowledge the support from the Computing Center in Xi'an, as well as the Instrumental Analysis Center and HPC Platform of Xi’an Jiaotong University.
Author information
Authors and Affiliations
Contributions
X.K. designed research, carried out the experiments, analyzed the experimental data, and wrote the manuscript. M.J. performed DFT calculations and analyzed data. J.L. performed COMSOL Multiphysics simulations and analyzed data. C.L., X.D., F.W., and S.B. helped analyze data. Y.L. and L.G. helped revise the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Peer review
Peer review information
Nature Communications thanks Hong Chen, Lingzhi Wang, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kang, X., Jiang, M., Lv, J. et al. Nanoscale greenhouse effect for promoting solar-driven CO2 reduction with water to CH4. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70960-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70960-9