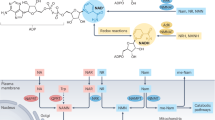
Abstract
NAD+ is a crucial metabolic cofactor whose intracellular levels can influence the progression of multiple metabolic and age-related complications. There is therefore a strong interest in using NAD+ precursors (vitamin B3s) as therapeutic tools, but most current precursors exhibit either poor bioavailability or adverse effects. This study examines the metabolic impact of chronic dietary supplementation with a newly described NAD+ precursor, dihydronicotinamide riboside (NRH), in mice using a comprehensive approach including phenotyping tests, RNA sequencing in different tissues and microbiome analyses. We show that chronic NRH administration at 100 mg/(kg*day) is well tolerated, yet has minimal metabolic effects in mice on a regular diet. However, NRH mitigates high-fat diet-induced metabolic complications when used as a preventive or as a treatment strategy, including improvements in glucose tolerance, increased hepatic expression of lipid catabolism genes and fat redistribution. These results highlight the potential of NRH as a therapeutic agent, although further studies are needed to optimize its use, as higher doses reveal signs of toxicity.
Similar content being viewed by others
Data availability
The transcriptomic data generated in this study have been deposited on ArrayExpress under accession no. E. In addition, all 16S rRNA gene sequencing results have been deposited under accession no. E. All data supporting the findings described in this manuscript are available in the article and in the Supplementary Information (See Source Data File) and from the corresponding author upon request. Source data are provided in this paper.
References
Houtkooper, R. H., Cantó, C., Wanders, R. J. & Auwerx, J. The secret life of NAD+: an old metabolite controlling new metabolic signaling pathways. Endocr. Rev. 31, 194–223 (2010).
Bai, P. Biology of poly(ADP-ribose) polymerases: the factotums of cell maintenance. Mol. Cell 58, 947–958 (2015).
Landry, J., Slama, J. T. & Sternglanz, R. Role of NAD+ in the deacetylase activity of the SIR2-like proteins. Biochem. Biophys. Res. Commun. 278, 685–690 (2000).
Lee, H. C. Cyclic ADP-ribose and nicotinic acid adenine dinucleotide phosphate (NAADP) as messengers for calcium mobilization. J. Biol. Chem. 287, 31633–31640 (2012).
Cercillieux, A., Ciarlo, E. & Canto, C. Balancing NAD+ deficits with nicotinamide riboside: therapeutic possibilities and limitations. Cell. Mol. Life Sci. 79, 463 (2022).
Revollo, J. R. et al. Nampt/PBEF/Visfatin regulates insulin secretion in β cells as a systemic NAD biosynthetic enzyme. Cell Metab. 6, 363–375 (2007).
Bogan, K. L. & Brenner, C. Nicotinic acid, nicotinamide, and nicotinamide riboside: a molecular evaluation of NAD+ precursor vitamins in human nutrition. Annu. Rev. Nutr. 28, 115–130 (2008).
Cantó, C. et al. The NAD(+) precursor nicotinamide riboside enhances oxidative metabolism and protects against high-fat diet-induced obesity. Cell Metab. 15, 838–847 (2012).
Yoshino, J., Mills, K. F., Yoon, M. J. & Imai, S. Nicotinamide mononucleotide, a key NAD(+) intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 14, 528–536 (2011).
Gariani, K. et al. Eliciting the mitochondrial unfolded protein response by nicotinamide adenine dinucleotide repletion reverses fatty liver disease in mice. Hepatology 63, 1190–1204 (2016).
Mitchell, S. J. et al. Nicotinamide Improves Aspects of Healthspan, but Not Lifespan, in Mice. Cell Metab. 27, 667–676.e4 (2018).
Trammell, S. A. J. et al. Nicotinamide riboside opposes type 2 diabetes and neuropathy in mice. Sci. Rep. 6, 26933 (2016).
Méndez-Lara, K. A. et al. Nicotinamide protects against diet-induced body weight gain, increases energy expenditure, and induces white adipose tissue beiging. Mol. Nutr. Food Res. 65, e2100111 (2021).
Li, Q. et al. Improving mitochondrial function in skeletal muscle contributes to the amelioration of insulin resistance by nicotinamide riboside. Int. J. Mol. Sci. 24, 10015 (2023).
Katsyuba, E. et al. De novo NAD+ synthesis enhances mitochondrial function and improves health. Nature 563, 354–359 (2018).
Zhou, C.-C. et al. Hepatic NAD(+) deficiency as a therapeutic target for non-alcoholic fatty liver disease in ageing. Br. J. Pharmacol. 173, 2352–2368 (2016).
Cuenoud, B. et al. Brain NAD is associated with ATP energy production and membrane phospholipid turnover in humans. Front. Aging Neurosci. 12, 609517 (2020).
Janssens, G. E. et al. Healthy aging and muscle function are positively associated with NAD+ abundance in humans. Nat. Aging 2, 254–263 (2022).
Massudi, H. et al. Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. PloS ONE 7, e42357 (2012).
Canto, C. NAD+ Precursors: a questionable redundancy. Metabolites 12, 630 (2022).
Kothawade, P. B., Thomas, A. B. & Chitlange, S. S. Novel niacin receptor agonists: a promising strategy for the treatment of dyslipidemia. Mini Rev. Med. Chem. 21, 2481–2496 (2021).
Lan, S. J. & Henderson, L. M. Uptake of nicotinic acid and nicotinamide by rat erythrocytes. J. Biol. Chem. 243, 3388–3394 (1968).
Sofue, M., Yoshimura, Y., Nishida, M. & Kawada, J. Uptake of nicotinamide by rat pancreatic beta cells with regard to streptozotocin action. J. Endocrinol. 131, 135–138 (1991).
Sadoogh-Abasian, F. & Evered, D. F. Absorption of nicotinic acid and nicotinamide from rat small intestine in vitro. Biochim. Biophys. Acta 598, 385–391 (1980).
Pissios, P. Nicotinamide N -methyltransferase: more than a vitamin B3 clearance enzyme. Trends Endocrinol. Metab. 28, 340–353 (2017).
Bieganowski, P. & Brenner, C. Discoveries of nicotinamide riboside as a nutrient and conserved NRK genes establish a Preiss-Handler independent route to NAD+ in fungi and humans. Cell 117, 495–502 (2004).
Liu, L. et al. Quantitative analysis of NAD synthesis-breakdown fluxes. Cell Metab. 27, 1067–1080 (2018).
Shats, I. et al. Bacteria boost mammalian host NAD metabolism by engaging the deamidated biosynthesis pathway. Cell Metab. 31, 564–579 (2020).
Ratajczak, J. et al. NRK1 controls nicotinamide mononucleotide and nicotinamide riboside metabolism in mammalian cells. Nat. Commun. 7, 13103 (2016).
Giroud-Gerbetant, J. et al. A reduced form of nicotinamide riboside defines a new path for NAD+ biosynthesis and acts as an orally bioavailable NAD+ precursor. Mol. Metab. 30, 192–202 (2019).
Yang, Y., Mohammed, F. S., Zhang, N. & Sauve, A. A. Dihydronicotinamide riboside is a potent NAD+ concentration enhancer in vitro and in vivo. J. Biol. Chem. 294, 9295–9307 (2019).
Yang, Y., Zhang, N., Zhang, G. & Sauve, A. A. NRH salvage and conversion to NAD+ requires NRH kinase activity by adenosine kinase. Nat. Metab. 2, 364–379 (2020).
Wu, K. et al. NADH and NRH as potential dietary supplements or pharmacological agents for early liver injury caused by acute alcohol exposure. J. Funct. Foods 87, 104852 (2021).
Fang, J. et al. A reduced form of nicotinamide riboside protects the cochlea against aminoglycoside-induced ototoxicity by SIRT1 activation. Biomed. Pharmacother. 150, 113071 (2022).
Chini, C. C. S. et al. Dihydronicotinamide riboside is a potent NAD+ precursor promoting a pro-inflammatory phenotype in macrophages. Front. Immunol. 13, 840246 (2022).
Li, J. et al. NAD+ bioavailability mediates PARG inhibition-induced replication arrest, intra S-phase checkpoint and apoptosis in glioma stem cells. NAR Cancer 3, zcab044 (2021).
Chellappa, K. et al. NAD precursors cycle between host tissues and the gut microbiome. Cell Metab. 34, 1947–1959.e5 (2022).
Ren, Z. et al. NAD+ and its possible role in gut microbiota: Insights on the mechanisms by which gut microbes influence host metabolism. Anim. Nutr. 10, 360–371 (2022).
Peluso, A. A. et al. Oral supplementation of nicotinamide riboside alters intestinal microbial composition in rats and mice, but not humans. Npj Aging 9, 7 (2023).
Kim, L. et al. Host–microbiome interactions in nicotinamide mononucleotide (NMN) deamidation. FEBS Lett. 597, 2196–2220 (2023).
Koren, O. et al. A guide to enterotypes across the human body: meta-analysis of microbial community structures in human microbiome datasets. PLoS Comput. Biol. 9, e1002863 (2013).
Spellerberg, I. F. & Fedor, P. J. A tribute to Claude Shannon (1916–2001) and a plea for more rigorous use of species richness, species diversity and the ‘Shannon–Wiener’ Index. Glob. Ecol. Biogeogr. 12, 177–179 (2003).
Sonavane, M., Hayat, F., Makarov, M., Migaud, M. E. & Gassman, N. R. Dihydronicotinamide riboside promotes cell-specific cytotoxicity by tipping the balance between metabolic regulation and oxidative stress. PLOS ONE 15, e0242174 (2020).
Giner, M. P. et al. A method to monitor the NAD+ metabolome—from mechanistic to clinical applications. Int. J. Mol. Sci. 22, 10598 (2021).
Vacca, M. et al. An unbiased ranking of murine dietary models based on their proximity to human metabolic dysfunction-associated steatotic liver disease (MASLD). Nat. Metab. 6, 1178–1196 (2024).
Farrell, G. et al. Mouse models of nonalcoholic steatohepatitis: toward optimization of their relevance to human nonalcoholic steatohepatitis. Hepatology 69, 2241–2257 (2019).
Xiao, L. et al. High-fat feeding rather than obesity drives taxonomical and functional changes in the gut microbiota in mice. Microbiome 5, 43 (2017).
Walker, A. et al. Distinct signatures of host–microbial meta-metabolome and gut microbiome in two C57BL/6 strains under high-fat diet. ISME J. 8, 2380–2396 (2014).
Zachara, M. et al. Mammalian adipogenesis regulator (Areg) cells use retinoic acid signalling to be non- and anti-adipogenic in age-dependent manner. EMBO J. 41, e108206 (2022).
Yu, J. et al. Redox biology in adipose tissue physiology and obesity. Adv. Biol. 7, 2200234 (2023).
Conze, D. B., Crespo-Barreto, J. & Kruger, C. L. Safety assessment of nicotinamide riboside, a form of vitamin B3. Hum. Exp. Toxicol. 35, 1149–1160 (2016).
Marinescu, A. G. et al. Safety assessment of high-purity, synthetic nicotinamide riboside (NR-E) in a 90-day repeated dose oral toxicity study, with a 28-day recovery arm. Int. J. Toxicol. 39, 307–320 (2020).
Koczor, C. A. et al. Temporal dynamics of base excision/single-strand break repair protein complex assembly/disassembly are modulated by the PARP/NAD+/SIRT6 axis. Cell Rep. 37, 109917 (2021).
Liu, Y. et al. Reduced nicotinamide mononucleotide (NMNH) potently enhances NAD+ and suppresses glycolysis, the TCA cycle, and cell growth. J. Proteome Res. 20, 2596–2606 (2021).
Xiao, W. & Loscalzo, J. Metabolic responses to reductive stress. Antioxid. Redox Signal. 32, 1330–1347 (2020).
Zhang, J. & Chen, F. Integrated transcriptome and metabolome study reveal the therapeutic effects of nicotinamide riboside and nicotinamide mononucleotide on nonalcoholic fatty liver disease. Biomed. Pharmacother. 175, 116701 (2024).
Okur, M. N. et al. Long-term NAD + supplementation prevents the progression of age-related hearing loss in mice. Aging Cell 22, e13909 (2023).
Kiesworo, K. et al. Nicotinamide mononucleotide restores impaired metabolism, endothelial cell proliferation and angiogenesis in old sedentary male mice. iScience 28, 111656 (2025).
Zeng, X. et al. NRH, a potent NAD+ enhancer, improves glucose homeostasis and lipid metabolism in diet-induced obese mice through an active adenosine kinase pathway. Metabolism 164, 156110 (2025).
Shulman, G. I. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N. Engl. J. Med. 371, 1131–1141 (2014).
Pellegrinelli, V., Carobbio, S. & Vidal-Puig, A. Adipose tissue plasticity: how fat depots respond differently to pathophysiological cues. Diabetologia 59, 1075–1088 (2016).
Sakers, A., De Siqueira, M. K., Seale, P. & Villanueva, C. J. Adipose-tissue plasticity in health and disease. Cell 185, 419–446 (2022).
Hotamisligil, G. S. Inflammation and endoplasmic reticulum stress in obesity and diabetes. Int. J. Obes. 32, S52–S54 (2008).
Özcan, U. et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306, 457–461 (2004).
Castro, J. P., Grune, T. & Speckmann, B. The two faces of reactive oxygen species (ROS) in adipocyte function and dysfunction. Biol. Chem. 397, 709–724 (2016).
Tormos, K. V. et al. Mitochondrial complex III ROS regulate adipocyte differentiation. Cell Metab. 14, 537–544 (2011).
Lee, H., Lee, Y. J., Choi, H., Ko, E. H. & Kim, J. Reactive oxygen species facilitate adipocyte differentiation by accelerating mitotic clonal expansion. J. Biol. Chem. 284, 10601–10609 (2009).
Baldini, F. et al. Adipocyte hypertrophy parallels alterations of mitochondrial status in a cell model for adipose tissue dysfunction in obesity. Life Sci. 265, 118812 (2021).
Grenier-Larouche, T., Galinier, A., Casteilla, L., Carpentier, A. C. & Tchernof, A. Omental adipocyte hypertrophy relates to coenzyme Q10 redox state and lipid peroxidation in obese women. J. Lipid Res. 56, 1985–1992 (2015).
Lapatto, H. A. K. et al. Nicotinamide riboside improves muscle mitochondrial biogenesis, satellite cell differentiation, and gut microbiota in a twin study. Sci. Adv. 9, eadd5163 (2023).
Schreiber, S. et al. Nicotinamide modulates gut microbial metabolic potential and accelerates recovery in mild-to-moderate COVID-19. Nat. Metab. 7, 1136–1149 (2025).
Utzschneider, K. M., Kratz, M., Damman, C. J. & Hullar, M. Mechanisms linking the gut microbiome and glucose metabolism. J. Clin. Endocrinol. Metab. 101, 1445–1454 (2016).
Dao, M. C. et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut 65, 426–436 (2016).
Han, Y. & Xiao, H. Whole food–based approaches to modulating gut microbiota and associated diseases. Annu. Rev. Food Sci. Technol. 11, 119–143 (2020).
Boutant, M. et al. SIRT1 enhances glucose tolerance by potentiating brown adipose tissue function. Mol. Metab. 4, 118–131 (2015).
Makarov, M. V. et al. Chemical and biochemical reactivity of the reduced forms of nicotinamide riboside. ACS Chem. Biol. 16, 604–614 (2021).
Membrez, M. et al. Trigonelline is an NAD+ precursor that improves muscle function during ageing and is reduced in human sarcopenia. Nat. Metab. 6, 433–447 (2024).
Bankhead, P. et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 7, 16878 (2017).
Cantó, C. & Garcia-Roves, P. M. High-resolution respirometry for mitochondrial characterization of ex vivo mouse tissues. Curr. Protoc. Mouse Biol. 5, 135–153 (2015).
Schmidt, U., Weigert, M., Broaddus, C. & Myers, G. Cell detection with star-convex polygons. in Medical Image Computing and Computer Assisted Intervention – MICCAI 2018(Springer International Publishing, Cham, 2018).
Alpern, D. et al. BRB-seq: ultra-affordable high-throughput transcriptomics enabled by bulk RNA barcoding and sequencing. Genome Biol. 20, 71 (2019).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90–W97 (2016).
Zhou, Y. et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 10, 1523 (2019).
Callahan, B. J. et al. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 13, 581–583 (2016).
McMurdie, P. J. & Holmes, S. phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8, e61217 (2013).
Acknowledgements
The authors thank Jessica Dessimoz at the Histology Platform from the Ecole Polytechnique Fédérale de Lausanne (EPFL) for technical and scientific support in the performance of histology analyses. We would also like to thank Roy Combe and the team at the phenotyping unit of the Centre for Phenogenomics at EPFL for technical assistance during indirect calorimetry and blood biochemistry analyses. We are also grateful to the Gene Expression Core Facility at EPFL for their support. Finally, we would like to thank Camille L.G. Lambert and Constantin Goeldel for their help and contribution in adapting the StarDist tool. This research was funded by Nestlé Research, by the EU Horizon Europe research and innovation programme MSCA-Doctoral Network NADIS (no. 101073251; to R.H.H., C.C., K.T.V., L.v.G.). We also thank NIH (NCCIH R21 grant AT009908-01) and the Mitchell Cancer Institute, F.P. Whiddon College of Medicine, University of South Alabama, for financial support to M.V.M., F.H. and M.E.M.
Author information
Authors and Affiliations
Contributions
M.R., S.M., M.E.M., R.H.H., B.D. and C.C. designed the different studies. F.H., M.V.M. and M.E.M. synthesised labelled and unlabelled forms of NRH. M.R., L.v.G., H.H., J.R., C.S., M.J., J.G.G., J.L.S.G. and C.C. performed the animal interventions, phenotyping and tissue collection. K.T.V., B.V.S., M.P.G., S.C., S.M. and R.H.H. performed the MS-based analyses of NAD+ metabolites. M.R. and V.G. performed gut microbiome analyses. M.R. and G.v.M. performed transcriptomic analyses. M.R., J.R. and C.C. performed all other analyses on tissue and blood samples. M.R., B.D. and C.C. wrote the manuscript, and all other authors contributed to its editing.
Corresponding author
Ethics declarations
Competing interests
M.R., M.J., J.G.G., S.M. and C.C. performed part of this research while they were employed by Nestlé Research, a part of the Société des Produits Nestlé S.A (SPN). M.P.G., S.C. and J.L.S.G. are employees of the SPN. M.E.M. is the inventor of intellectual property on the synthesis of NRH; M.E.M. and S.P.N. have filed patents related to therapeutic uses of NRH. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Jonas Treebak, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Rumpler, M., van Mierlo, G., Vinten, K.T. et al. Therapeutic potential of dihydronicotinamide riboside (NRH) on obesity and glucose intolerance in mice. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70965-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70965-4