Abstract
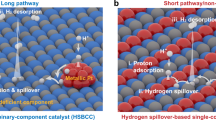
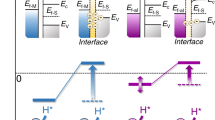
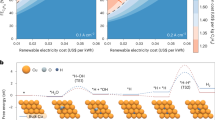
The hydrogen spillover effect offers a promising strategy to overcome the kinetic bottleneck of proton desorption in hydrogen evolution reaction catalysts. However, conventional hydrogen spillover mechanisms rely on interfacial proton transfer between distinct phases and suffer from inherent energy barriers. Here, we show a non-interfacial hydrogen spillover mechanism in a Ni17W3-WO2 heterostructure, engineered through the synergistic creation of a built-in strain gradient and directional electron transfer. This design spatially confines the complete hydrogen evolution process within the Ni17W3 phase, thereby circumventing cross-phase migration and reshaping the hydrogen adsorption energy landscape. Experimental and theoretical analyses confirm the elimination of interfacial barriers and establishment of an optimized proton-migration route. The resulting catalyst achieves a low overpotential of 21 mV at 10 mA cm–2 in 0.5 M H2SO4, along with sustained stability (>1500 hours at 500 mA cm–2) and a Faradaic efficiency of 98.65%. This work demonstrates how tailored heterostructures can bypass interfacial bottlenecks, providing guidance for developing efficient non-precious hydrogen spillover catalysts and advancing sustainable hydrogen production.
Similar content being viewed by others
Data availability
The source data generated in this study are provided in the Source data file. Source data are provided with this paper.
References
Zhang, W. et al. Water electrolysis toward elevated temperature: advances, challenges and frontiers. Chem. Rev. 123, 7119–7192 (2023).
Green, F., Bois von Kursk, O., Muttitt, G. & Pye, S. No new fossil fuel projects: the norm we need. Science 384, 954–957 (2024).
Sha, Q. et al. 10,000-h-stable intermittent alkaline seawater electrolysis. Nature 639, 360–367 (2025).
Guan, D. et al. Hydrogen society: from present to future. Energy Environ. Sci. 16, 4926–4943 (2023).
Glenk, G. & Reichelstein, S. Economics of converting renewable power to hydrogen. Nat. Energy 4, 216–222 (2019).
Zhai, W. et al. Recent progress on the long-term stability of hydrogen evolution reaction electrocatalysts. InfoMat 4, e12357 (2022).
Kosmala, T. et al. Operando visualization of the hydrogen evolution reaction with atomic-scale precision at different metal-graphene interfaces. Nat. Catal. 4, 850–859 (2021).
Liang, J. et al. Electroreduction of alkaline/natural seawater: self-cleaning Pt/carbon cathode and on-site co-synthesis of H2 and Mg hydroxide nanoflakes. Chem 10, 3067–3087 (2024).
Zuo, Y. et al. Ru-Cu nanoheterostructures for efficient hydrogen evolution reaction in alkaline water electrolyzers. J. Am. Chem. Soc. 145, 21419–21431 (2023).
Wang, J., Xu, F., Jin, H., Chen, Y. & Wang, Y. Non-noble metal-based carbon composites in hydrogen evolution reaction: fundamentals to applications. Adv. Mater. 29, 1605838 (2017).
Wei, Z.-W. et al. Reversed charge transfer and enhanced hydrogen spillover in platinum nanoclusters anchored on titanium oxide with rich oxygen vacancies boost hydrogen evolution reaction. Angew. Chem. Int. Ed. 60, 16622–16627 (2021).
Li, J. et al. Boosting electrocatalytic activity of Ru for acidic hydrogen evolution through hydrogen spillover strategy. ACS Energy Lett. 7, 1330–1337 (2022).
Dai, J. et al. Hydrogen spillover in complex oxide multifunctional sites improves acidic hydrogen evolution electrocatalysis. Nat. Commun. 13, 1189 (2022).
Jo, S., Shin, K. H., Kim, E. & Sohn, J. I. High-entropy oxychalcogenide for hydrogen spillover enhanced hydrogen evolution reaction in proton and anion exchange membrane water electrolyzers. Small 21, 2411883 (2025).
Li, J., Ma, Y., Ho, J. C. & Qu, Y. Hydrogen spillover phenomenon at the interface of metal-supported electrocatalysts for hydrogen evolution. Acc. Chem. Res. 57, 895–904 (2024).
Peng, Q. et al. Lattice strain induced Ni0.85Se/WO2.90 heterostructures accelerate dynamic reconstruction for efficient water oxidation. Appl. Catal. B Environ. Energy 384, 126158 (2026).
Lima, N. A. et al. NiWO4 powders prepared via polymeric precursor method for application as ceramic luminescent pigments. J. Adv. Ceram. 9, 55–63 (2020).
Borkar, S. S. & Shetty, M. Elucidating the role of strain in catalysis toward modulating surface-adsorbate interactions and tuning catalytic activity. Preprint at https://doi.org/10.26434/chemrxiv-2024-8ltp5 (2024).
Liang, J. et al. Efficient bubble/precipitate traffic enables stable seawater reduction electrocatalysis at industrial-level current densities. Nat. Commun. 15, 2950 (2024).
Ma, Y. et al. High-entropy metal-organic frameworks for highly reversible sodium storage. Adv. Mater. 33, 2101342 (2021).
Li, Y. et al. Multimetal borides nanochains as efficient electrocatalysts for overall water splitting. Small 15, 1804212 (2019).
Hu, W. et al. Zirconium doping facilitates a vertically aligned NiCoZr-layered hydroxide nanoneedle arrays electrode for hybrid supercapacitors exhibiting a 90,000 cycle durability. J. Energy Storage 97, 112825 (2024).
Zhuo, S. et al. Dual-template engineering of triple-layered nanoarray electrode of metal chalcogenides sandwiched with hydrogen-substituted graphdiyne. Nat. Commun. 9, 3132 (2018).
Jiang, F. et al. A straightforward solvent-pair-enabled multicomponent coassembly approach toward noble-metal-nanoparticle-decorated mesoporous tungsten oxide for trace ammonia sensing. Adv. Mater. 36, 2313547 (2024).
Li, J. et al. Elucidating the critical role of ruthenium single atom sites in water dissociation and dehydrogenation behaviors for robust hydrazine oxidation-boosted alkaline hydrogen evolution. Adv. Funct. Mater. 32, 2109439 (2022).
Li, Y., Hassan, M. S., Zhao, X. & Rogach, A. L. Heterostructured electrocatalysts: from fundamental microkinetic model to electron configuration and interfacial reactive microenvironment. Adv. Mater. 37, 2418146 (2025).
Zhao, G., Rui, K., Dou, S. X. & Sun, W. Heterostructures for electrochemical hydrogen evolution reaction: a review. Adv. Funct. Mater. 28, 1803291 (2018).
Liu, D. et al. Atomically dispersed platinum supported on curved carbon supports for efficient electrocatalytic hydrogen evolution. Nat. Energy 4, 512–518 (2019).
Fan, H. et al. Revealing the role of Ru-O-Ce interface coupling in CeO2-Ru aerogel for boosting hydrogen evolution kinetics. Adv. Energy Mater. 15, 2405681 (2025).
Huynh, T. T., Mai, V. T. T., Nguyen, A. Q. K. & Pham, H. Q. Ni-doped RuPt nanoalloy on acid-treated carbon for pH-universal hydrogen evolution reaction. Adv. Sustain. Syst. 8, 2300380 (2024).
Feng, Y. et al. Electronic metal-support interaction induces hydrogen spillover and platinum utilization in hydrogen evolution reaction. Angew. Chem. Int. Ed. 64, e202413417 (2025).
Zhang, S.-N. et al. Amplifying intermetal bias of Pt-Rh alloy nanolayers for facilitated hydrogen inter-diffusion and evolution. Angew. Chem. Int. Ed. 64, e202512299 (2025).
He, H. et al. Facile synthesis of high areal density and stable Pt single-atom electrocatalysts by arc discharge atomization and coni trapping. Adv. Sci. 12, e11806 (2025).
Siva, P. & Vasu, K. Enhanced hydrogen evolution and symmetric supercapacitor performance of a Ru-doped multiphase WS2 electrode. J. Mater. Chem. A 12, 961–967 (2024).
Zhang, J. et al. Face-centered cubic structured RuCu hollow urchin-like nanospheres enable remarkable hydrogen evolution catalysis. Sci. China Chem. 65, 87–95 (2022).
Zhang, X. et al. Chlorine coordination engineering optimizes hydrogen evolution reaction kinetics on Ru(100) facet. Adv. Funct. Mater. 35, 202508730 (2025).
Majumdar, A. et al. Strain engineering of Ru-Co2Ni nanoalloy encapsulated with carbon nanotubes for efficient anion and proton exchange membrane water electrolysis. Adv. Funct. Mater. 35, 2420517 (2025).
Liu, Q. et al. Nearly hollow Ru-Cu-MoO2 octahedrons consisting of clusters and nanocrystals for high efficiency hydrogen evolution reaction. J. Mater. Chem. A 10, 12341–12349 (2022).
Li, Y. et al. Arming Ru with oxygen-vacancy-enriched RuO2 sub-nanometer skin activates unique bifunctionality for pH-universal overall water splitting. Adv. Mater. 35, 2206351 (2023).
Qiang, L. et al. Carbon dot-driven spatial and electronic modulation of Ru on graphene for pH-universal hydrogen evolution reaction electrocatalysts. Green Chem. 27, 12364–12377 (2025).
Wang, J., Zhang, G., Liu, H., Wang, L. & Li, Z. Ru regulated electronic structure of PdxCuy nanosheets for efficient hydrogen evolution reaction in wide pH range. Small 20, 2310277 (2024).
Zhao, W. et al. Pt-Ru dimer electrocatalyst with electron redistribution for hydrogen evolution reaction. ACS Catal. 12, 5540–5548 (2022).
Zhu, T. et al. Amorphous ruthenium-selenium nanoparticles as a pH-universal catalyst for enhanced hydrogen evolution reaction. ACS Catal. 14, 1914–1921 (2024).
Liu, H. et al. Single-atom nickel encapsulated in nanosheet-coiled rGo-CTAB-MoS2 nanoflowers for high-efficiency and long-term hydrogen evolution in acidic medium. Adv. Funct. Mater. 35, 2425826 (2025).
Siva, P. & Vasu, K. Efficient hydrogen evolution from degenerate-doped p-type WS2 electrocatalysts over a wide pH range. J. Mater. Chem. A 12, 11101–11108 (2024).
Ma, Y.-Y. et al. Highly efficient hydrogen evolution triggered by a multi-interfacial Ni/WC hybrid electrocatalyst. Energy Environ. Sci. 11, 2114–2123 (2018).
Naseem, M. et al. Tuning the catalytic activity of MoS2−x-NbSx heterostructure nanosheets for bifunctional acidic water splitting. Small 21, 2501464 (2025).
Wu, R. et al. Dopant triggered atomic configuration activates water splitting to hydrogen. Nat. Commun. 14, 2306 (2023).
Li, J. et al. Hollow bimetallic cobalt-based selenide polyhedrons for efficient hydrogen production in practical PEM electrolysis. ACS Sustain. Chem. Eng. 12, 10020–10032 (2024).
Chen, Z. et al. Metallic W/WO2 solid-acid catalyst boosts hydrogen evolution reaction in alkaline electrolyte. Nat. Commun. 14, 5363 (2023).
Kwon, J. et al. Tailored electronic structure of Ir in high entropy alloy for highly active and durable bifunctional electrocatalyst for water splitting under an acidic environment. Adv. Mater. 35, 2300091 (2023).
Chen, J. et al. Reversible hydrogen spillover in Ru-WO3-x enhances hydrogen evolution activity in neutral pH water splitting. Nat. Commun. 13, 5382 (2022).
Hou, S. et al. Ru single atoms tailoring the acidity of metallic tungsten dioxide for a boosted alkaline hydrogen evolution reaction. ACS Catal. 14, 8238–8251 (2024).
Zhao, H. et al. Raw biomass electroreforming coupled to green hydrogen generation. Nat. Commun. 12, 2008 (2021).
Du, H. et al. Unlocking interfacial electron transfer of ruthenium phosphides by homologous core-shell design toward efficient hydrogen evolution and oxidation. Adv. Mater. 34, 2204624 (2022).
Yan, Y. et al. H-buffer effects boosting H-spillover for efficient hydrogen evolution reaction. Energy Environ. Sci. 17, 6024–6033 (2024).
Yuan, C. et al. KxNayH(2-x-y)Ti2O5 for high-performance lithium-ion battery anode with Li dendrites-free mechanism, zero-strain characteristic. Adv. Energy Mater. 13, 2302015 (2023).
Wang, Q. et al. 2D metastable-phase hafnium oxide triggers hydrogen spillover for boosting hydrogen production. Adv. Mater. 37, 2415978 (2025).
Bastians, S., Crump, G., Griffith, W. P. & Withnall, R. Raspite and studtite: Raman spectra of two unique minerals. J. Raman Spectrosc. 35, 726–731 (2004).
Ross-Medgaarden, E. I. & Wachs, I. E. Structural determination of bulk and surface tungsten oxides with UV-vis diffuse reflectance spectroscopy and Raman spectroscopy. J. Phys. Chem. C. 111, 15089–15099 (2007).
Guo, P. et al. Heterojunction-induced rapid transformation of Ni3+/Ni2+ sites which mediates urea oxidation for energy-efficient hydrogen production. Adv. Mater. 36, 2311766 (2024).
Wang, Y. et al. Defect-induced Zn-Co pair active site for high-efficiency electrosynthesis of H2O2. Matter 8, 102479 (2025).
You, S. et al. Quantifying the conductivity of a single polyene chain by lifting with an STM tip. Nat. Commun. 15, 6475 (2024).
Liu, J. et al. Optimizing hydrogen adsorption by d-d orbital modulation for efficient hydrogen evolution catalysis. Adv. Energy Mater. 12, 2103301 (2022).
Wang, J. et al. Switch Volmer-Heyrovsky to Volmer-Tafel pathway for efficient acidic electrocatalytic hydrogen evolution by correlating Pt single atoms with clusters. Small 20, 2309427 (2024).
Jin, J. et al. Atomic sulfur filling oxygen vacancies optimizes H absorption and boosts the hydrogen evolution reaction in alkaline media. Angew. Chem. Int. Ed. 60, 14117–14123 (2021).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558–561 (1993).
Noerskov, J. K. et al. Trends in the exchange current for hydrogen evolution. J. Electrochem. Soc. 152, J23 (2005).
Yang, J. et al. Ultrahigh-current-density niobium disulfide catalysts for hydrogen evolution. Nat. Mater. 18, 1309–1314 (2019).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (nos. 52002294 and 22379116), Hubei Provincial Natural Science Foundation of China (no. JCZRLH202600799), and the Open and Innovation Fund of Hubei Three Gorges Laboratory (no. SC250002). The authors also extend their appreciation to the Deanship of Scientific Research, Islamic University of Madinah, Saudi Arabia, for funding this research work.
Author information
Authors and Affiliations
Contributions
X.S., X.P., and S.X. designed this research and wrote the manuscript. S.X., H.D., and S.C. performed material synthesis and performance tests. Y.M. conducted theoretical calculations. L.X., B.G., X.Z., and Y.Y. performed material characterizations. I.S. polished the manuscript. X.S., X.P. supervised the research. All authors contributed to and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Huilong Fei, Hyuksu Han and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xie, S., Dong, H., Cao, S. et al. Engineering non-interfacial hydrogen spillover in a Ni17W3-WO2 heterostructure. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70976-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70976-1