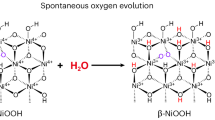
Abstract
The realization of multi-energy water oxidation systems is impeded by the challenge of integrating multiple energy inputs. Here, we overcome this limitation via ultrasonic pre-treatment of the electrolyte, which triggers a mechano-electrochemical coupling effect through piezoelectric polarization. This process promotes a Grotthuss-type OH− state that weakens O-H bonds and increases the interfacial OH− concentration, thereby influencing the electrochemical reconstruction of Ni(OH)2 to NiOOH and modifying water electrolysis pathways. These changes enhance Ni-O covalency and synergistically activate two low-energy water oxidation pathways on NiOOH involving lattice oxygen: one couples lattice oxygen with adsorbed oxygen, while the other facilitates direct lattice oxygen-oxygen coupling. Both routes bypass the high-energy *OOH intermediate typical of the conventional adsorbate evolution mechanism (*OH → *O → *OOH → O2), with the latter also avoiding *O adsorption entirely. Notably, just one minute of ultrasonic stimulation reduces the overpotential by 222 mV at 100 mA cm-2. This pulsed-energy strategy thus offers an efficient and scalable approach to realizing multi-energy-enhanced water splitting.
Similar content being viewed by others
Data availability
The source data generated in this study are provided in the Source Data file. Source data are provided with this paper.
References
Turner, J. A. Sustainable hydrogen production. Science 305, 972–975 (2004).
Shah, A. H. et al. The role of alkali metal cations and platinum-surface hydroxyl in the alkaline hydrogen evolution reaction. Nat. Catal. 5, 923–933 (2022).
You, B. et al. Enhancing electrocatalytic water splitting by strain engineering. Adv. Mater. 31, 1807001 (2019).
Song, J. et al. A review on fundamentals for designing oxygen evolution electrocatalysts. Chem. Soc. Rev. 49, 2196–2214 (2020).
Suen, N. T. et al. Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives. Chem. Soc. Rev. 46, 337–365 (2017).
Zhang, K. & Zou, R. Advanced transition metal-based OER electrocatalysts: current status, opportunities, and challenges. Small 17, 2100129 (2021).
Song, F. et al. Transition metal oxides as electrocatalysts for the oxygen evolution reaction in alkaline solutions: an application-inspired renaissance. J. Am. Chem. Soc. 140, 7748–7759 (2018).
Zhai, P. et al. Regulating electronic states of nitride/hydroxide to accelerate kinetics for oxygen evolution at large current density. Nat. Commun. 14, 1874 (2023).
Hu, C. et al. Advanced catalyst design strategies and in-situ characterization techniques for enhancing electrocatalytic activity and stability of oxygen evolution reaction. Electrochem. Energy Rev. 7, 1–20 (2024).
Hong, W. T. et al. Charge-transfer-energy-dependent oxygen evolution reaction mechanisms for perovskite oxides. Energy Environ. Sci. 10, 2190–2200 (2017).
Liao, Y. et al. Lattice distortion induced Ce-doped NiFe-LDH for efficient oxygen evolution. Chem. Eng. J. 464, 142669 (2023).
Liu, R., Wang, Y., Liu, D., Zou, Y. & Wang, S. Water-plasma-enabled exfoliation of ultrathin layered double hydroxide nanosheets with multivacancies for water oxidation. Adv. Mater. 29, 1701546 (2017).
Zhang, D. et al. Optimal electrocatalyst design strategies for acidic oxygen evolution. Adv. Sci. 11, 2401975 (2024).
Lu, M., Du, Y., Yan, S., Yu, T. & Zou, Z. Thermally stimulated spin switching accelerates water electrolysis. Phys. Rev. Lett. 133, 258001 (2024).
Lu, M., Du, Y., Yan, S., Yu, T. & Zou, Z. Thermal suppression of charge disproportionation accelerates interface electron transfer of water electrolysis. Proc. Natl. Acad. Sci. USA 121, e2316054120 (2024).
Hu, C., Dong, Y., Shi, Q., Long, R. & Xiong, Y. Catalysis under electric-/magnetic-/electromagnetic-field coupling. Chem. Soc. Rev. 54, 524559 (2025).
Xie, F., Du, Y., Lu, M., Yan, S. & Zou, Z. Thermal-stimulated spin disordering accelerates water electrolysis. Energy Environ. Sci. 18, 1972–1983 (2025).
Fujishima, A. & Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972).
Garces-Pineda, F. A., Blasco-Ahicart, M., Nieto-Castro, D., Lopez, N. & Ramon Galan-Mascaros, J. Direct magnetic enhancement of electrocatalytic water oxidation in alkaline media. Nat. Energy 4, 519–525 (2019).
Du, Y. et al. Continuous strain tuning of oxygen evolution catalysts with anisotropic thermal expansion. Nat. Commun. 15, 1780 (2024).
Liu, D. et al. Heat-triggered ferri-to-paramagnetic transition accelerates redox couple-mediated electrocatalytic water oxidation. Adv. Funct. Mater. 32, 2111234 (2022).
Marx, D., Chandra, A. & Tuckerman, M. E. Aqueous basic solutions: hydroxide solvation, structural diffusion, and comparison to the hydrated proton. Chem. Rev. 110, 2174–2216 (2010).
Li, Y. et al. Facilitating the rapid Grotthuss diffusion of OH⁻ in electrolyte for accelerated oxygen evolution reaction. J. Water Process Eng. 66, 106062 (2024).
Payaka, A., Yotmanee, P. & Tongraar, A. Characteristics of the “hypercoordination” of hydroxide (OH-) in water: a comparative study of HF/MM and B3LYP/MM MD simulations. J. Mol. Liq. 188, 89–95 (2013).
Kronberg, R. & Laasonen, K. Dynamics and surface propensity of H+ and OH- within rigid interfacial water: implications for electrocatalysis. J. Phys. Chem. Lett. 12, 10128–10134 (2021).
Chandra, A., Tuckerman, M. E. & Marx, D. Connecting solvation shell structure to proton transport kinetics in hydrogen-bonded networks via population correlation functions. Phys. Rev. Lett. 99, 145901 (2007).
Yang, Y. et al. Flexible piezoelectric pressure sensor based on polydopamine-modified BaTiO3/PVDF composite film for human motion monitoring. Sens. Actuators A Phys. 301, 111789 (2020).
Kulkarni, N. D. & Kumari, P. Role of rGO on mechanical, thermal, and piezoelectric behaviour of PVDF-BTO nanocomposites for energy harvesting applications. J. Polym. Res. 30, 3449 (2023).
Liu, Y. et al. Enhanced piezo-catalytic performance of BaTiO3 nanorods combining highly exposed active crystalline facets and superior deformation capability: water purification and activation mechanism. Chem. Eng. J. 488, 150768 (2024).
Davis, J. G., Gierszal, K. P., Wang, P. & Ben-Amotz, D. Water structural transformation at molecular hydrophobic interfaces. Nature 491, 582–585 (2012).
Wang, Y.-H. et al. In situ Raman spectroscopy reveals the structure and dissociation of interfacial water. Nature 600, 81–85 (2021).
Tao, X. et al. Iron and oxygen vacancies co-modulated adsorption evolution and lattice oxygen dual-path mechanism for water oxidation. Nat. Commun. 16, 8788 (2025).
Wu, F. et al. Engineering lattice oxygen regeneration of NiFe layered double hydroxide enhances oxygen evolution catalysis durability. Angew. Chem. Int. Ed. 64, e202413250 (2025).
Bediako, D. K. et al. Structure-activity correlations in a nickel-borate oxygen evolution catalyst. J. Am. Chem. Soc. 134, 6801–6809 (2012).
Lo, Y. & Hwang, B. In situ Raman studies on cathodically deposited nickel hydroxide films and electroless Ni-P electrodes in 1 M KOH solution. Langmuir 14, 944–950 (1998).
Chen, D. et al. Probing structural evolution and charge storage mechanism of NiO₂Hₓ electrode materials using in operando resonance Raman spectroscopy. Adv. Sci. 3, 1600045 (2016).
Klaus, S., Cai, Y., Louie, M. W., Trotochaud, L. & Bell, A. T. Effects of Fe electrolyte impurities on Ni(OH)₂/NiOOH structure and oxygen evolution activity. J. Phys. Chem. C. 119, 7243–7254 (2015).
Zhou, Y.-N., Li, F.-T., Dong, B. & Chai, Y.-M. Double self-reinforced coordination modulation constructing stable Ni4+ for water oxidation. Energy Environ. Sci. 17, 1468–1481 (2024).
Yin, Z.-H. et al. Ir single atoms boost metal-oxygen covalency on selenide-derived NiOOH for direct intramolecular oxygen coupling. J. Am. Chem. Soc. 146, 6846–6855 (2024).
Wu, Y. et al. Triggering lattice oxygen activation of single-atomic Mo sites anchored on Ni-Fe oxyhydroxides nanoarrays for electrochemical water oxidation. Adv. Mater. 34, 2202523 (2022).
Pandya, K. I., Hoffman, R. W., McBreen, J. & O’Grady, W. E. In situ X-ray absorption spectroscopic studies of nickel oxide electrodes. J. Electrochem. Soc. 137, 383–383 (1990).
Ke, L. et al. Carbonate electrolytes manipulate lattice oxygen dynamics of oxyhydroxides toward efficient and durable water oxidation. Nat. Commun. 16, 11241 (2025).
Zhang, N. & Chai, Y. Lattice oxygen redox chemistry in solid-state electrocatalysts for water oxidation. Energy Environ. Sci. 14, 4647–4671 (2021).
He, L., Song, F., Cao, Y. & Chen, Q. Covalency regulation of high entropy oxyfluoride-rich metal-oxygen bond enhances high rate and long-life performance of sodium-ion batteries under high voltage. Energy Storage Mater. 80, 104370 (2025).
Zhao, Y. et al. Designing local electron delocalization in 2D covalent organic frameworks for enhanced sunlight-driven photocatalytic activity. ACS Catal. 14, 3556–3564 (2024).
Wang, Y. et al. Gd-induced oxygen vacancy creation activates lattice oxygen oxidation for water electrolysis. Adv. Funct. Mater. 35, 2500118 (2025).
Adamu, H. et al. Tuning OER electrocatalysts toward LOM pathway through the lens of multi-descriptor feature selection by artificial intelligence-based approach. ACS Mater. Lett. 5, 299–320 (2022).
Dong, H. et al. Spin magnetic effect activates dual-site intramolecular O-O bridging for nickel-iron hydroxide enhanced oxygen evolution catalysis. Adv. Sci. 12, 2415525 (2025).
Li, J. et al. Activated Ni-O-Ir enhanced electron transfer for boosting oxygen evolution reaction activity of LaNi1-xIrxO3. Small 18, 2204723 (2022).
Lu, Q. et al. Breaking the activity-stability trade-off of RuO2 via metallic Ru bilateral regulation for acidic oxygen evolution reaction. Angew. Chem. Int. Ed. 64, e202503733 (2025).
Zhai, P. et al. Regulating electronic states of nitride/hydroxide to accelerate kinetics for oxygen evolution at large current density. Nat. Commun. 14, 1873 (2023).
Grimaud, A. et al. Activating lattice oxygen redox reactions in metal oxides to catalyse oxygen evolution. Nat. Chem. 9, 457–465 (2017).
Ren, X., Zhai, Y., Yang, N., Wang, B. & Liu, S. F. Lattice oxygen redox mechanisms in the alkaline oxygen evolution reaction. Adv. Funct. Mater. 34, 2401610 (2024).
Wang, X., Zhong, H., Xi, S., Lee, W. S. V. & Xue, J. Understanding of oxygen redox in the oxygen evolution reaction. Adv. Mater. 34, 2107956 (2022).
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
Bayly, C. I., Cieplak, P., Cornell, W. & Kollman, P. A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J. Phys. Chem. 97, 10269–10280 (1993).
Hess, B., Kutzner, C., van der Spoel, D. & Lindahl, E. GROMACS 4: Algorithms for highly efficient, load-balanced, and scalable molecular simulation. J. Chem. Theory Comput. 4, 435–447 (2008).
Berendsen, H. J. C., Postma, J. P. M., Van Gunsteren, W. F., DiNola, A. & Haak, J. R. Molecular dynamics with coupling to an external bath. J. Chem. Phys. 81, 3684–3690 (1984).
Essmann, U., Perera, L., Berkowitz, M. L., Darden, T., Lee, H. & Pedersen, L. G. A smooth particle mesh Ewald method. J. Chem. Phys. 103, 8577–8593 (1995).
Astrakas, L. G., Gousias, C. & Tzaphlidou, M. Structural destabilization of chignolin under the influence of oscillating electric fields. J. Appl. Phys. 111, 074702 (2012).
Hess, B., Bekker, H., Berendsen, H. J. C. & Fraaije, J. G. E. M. LINCS: a linear constraint solver for molecular simulations. J. Comput. Chem. 18, 1463–1472 (1997).
Van Gunsteren, W. F. & Berendsen, H. J. C. A leap-frog algorithm for stochastic dynamics. Mol. Simul. 1, 173–185 (1988).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Elbaz, Y., Furman, D. & Toroker, M. C. Hydrogen transfer through different crystal phases of nickel oxy/hydroxide. Phys. Chem. Chem. Phys. 20, 25169–25178 (2018).
Friebel, D. et al. Identification of highly active Fe sites in (Ni,Fe)OOH for electrocatalytic water splitting. J. Am. Chem. Soc. 137, 1305–1313 (2015).
Dronskowski, R. & Blöchl, P. E. Crystal orbital Hamilton populations (COHP): energy-resolved visualization of chemical bonding in solids based on density-functional calculations. J. Phys. Chem. 97, 8617–8624 (1993).
Maintz, S., Deringer, V. L., Tchougréeff, A. L. & Dronskowski, R. LOBSTER: a tool to extract chemical bonding from plane-wave based DFT. J. Comput. Chem. 37, 1030–1035 (2016).
Acknowledgements
This work was primarily supported by the National Natural Science Foundation of China [Grant Nos. 22409089 (Y. L.), 52272082 (J. K.), 52272217 (S. Y.), 51872135 (S. Y.), 51572121 (S. Y.) and 21633004 (S. Y.)], the Natural Science Foundation of Jiangsu Province (No. BK20250031, J. K.), the Scientific and Technological Innovation Project of Carbon Emission Peak and Carbon Neutrality of Jiangsu Province (No. BE2022028-1, S. Y.), and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD, C. L.). The numerical calculations in this paper have been performed on the computing facilities in the High Performance Computing Center (HPCC) of Nanjing University.
Author information
Authors and Affiliations
Contributions
Y.L., S.Y., and J.K. conceived and designed the research. C.L., J.K., and S.Y. supervised and coordinated the project. Y.L., S.W., and S.Y. wrote the manuscript with critical input from all co-authors. Y.D. performed the theoretical calculations. S.W., M.Y., Y.C., and T.M. performed the experimental work and data collection under Y.L.’s guidance. Y.L., S.W., and M.Y. conducted data analysis and interpretation. All authors participated in manuscript revision and approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Siyu Lu, Shichun Mu, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Y., Wang, S., Yuan, M. et al. Piezoelectric activation of dual lattice-oxygen mechanism through OH− Grotthuss transport in water electrolysis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70979-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70979-y