Abstract
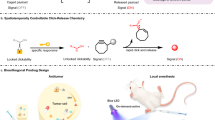
Dynamic and programmable control of therapeutic delivery is a long-standing goal in medicine. Iontronic devices offer precise electronic control over the dosage of bioactive molecules, yet their use has been confined to charged, low-molecular-weight compounds that are electrochemically stable during transport. Here, we present a hybrid delivery platform that integrates iontronic transport with bioorthogonal click-to-release chemistry. In this system, iontronic pumps electrophoretically deliver charged tetrazines as molecular scissors that selectively react with immobilized trans-cyclooctene (TCO)-linked payloads, enabling on-demand bioorthogonal cleavage of the TCO linker and controlled payload release. This approach retains the electronic precision of iontronics while overcoming molecular size, charge, and stability constraints. We demonstrate tunable tetrazine delivery over several days and electronically controlled release of immobilized payloads from small bioactive molecules, such as the antimitotic agent CA4, to the large protein bovine serum albumin. Hence, by integrating bioorthogonal click-to-release strategies, iontronic delivery is extended to biologically relevant macromolecules, providing a foundation for advanced programmable electroceutical devices.
Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the article and its supplementary files. Processed and raw data files generated in this study, including quantitative datasets underlying the figures and raw NMR spectroscopy data, have been deposited in the public institutional repository of TU Wien (TU Wien Research Data) under https://doi.org/10.48436/vxpen-9kg70 (https://doi.org/10.48436/vxpen-9kg70). Any additional requests for information can be directed to and will be fulfilled by the corresponding authors. Source data are provided with this paper.
References
Raijada, D., Wac, K., Greisen, E., Rantanen, J. & Genina, N. Integration of personalized drug delivery systems into digital health. Adv. Drug Deliv. Rev. 176, 113857 (2021).
Badarinadh, K. et al. Intelligent drug delivery: pioneering stimuli-responsive systems to revolutionize disease management- an in-depth exploration. Curr. Drug Deliv. 22, 195–214 (2025).
Li, J. & Mooney, D. J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 1, 16071 (2016).
Patra, J. K. et al. Nano based drug delivery systems: recent developments and future prospects. J. Nanobiotechnology 16, 71 (2018).
Torii, S. et al. Drug-eluting coronary stents: insights from preclinical and pathology studies. Nat. Rev. Cardiol. 17, 37–51 (2020).
Qian, F., Stowe, N., Saidel, G. M. & Gao, J. Comparison of doxorubicin concentration profiles in radiofrequency-ablated rat livers from sustained- and dual-release PLGA millirods. Pharm. Res. 21, 394–399 (2004).
Chiu, B. et al. Surgery combined with controlled-release doxorubicin silk films as a treatment strategy in an orthotopic neuroblastoma mouse model. Br. J. Cancer 111, 708–715 (2014).
Di Mascolo, D. et al. μMESH-enabled sustained delivery of molecular and nanoformulated drugs for glioblastoma treatment. ACS Nano 17, 14572–14585 (2023).
Anselmo, A. C. & Mitragotri, S. An overview of clinical and commercial impact of drug delivery systems. J. Control. Release 190, 15–28 (2014).
Benhabbour, S. R. et al. Ultra-long-acting tunable biodegradable and removable controlled release implants for drug delivery. Nat. Commun. 10, 4324 (2019).
Yoo, J. & Won, Y.-Y. Phenomenology of the initial burst release of drugs from PLGA microparticles. ACS Biomater. Sci. Eng. 6, 6053–6062 (2020).
Yun, Y. H., Lee, B. K. & Park, K. Controlled drug delivery: historical perspective for the next generation. J. Control. Release 219, 2–7 (2015).
Riley, R. S., June, C. H., Langer, R. & Mitchell, M. J. Delivery technologies for cancer immunotherapy. Nat. Rev. Drug Discov. 18, 175–196 (2019).
Delcassian, D., Patel, A. K., Cortinas, A. B. & Langer, R. Drug delivery across length scales. J. Drug Target. 27, 229–243 (2019).
Kearney, C. J. & Mooney, D. J. Macroscale delivery systems for molecular and cellular payloads. Nat. Mater. 12, 1004–1017 (2013).
Fenton, O. S., Olafson, K. N., Pillai, P. S., Mitchell, M. J. & Langer, R. Advances in biomaterials for drug delivery. Adv. Mater. 30, 1705328 (2018).
Farra, R. et al. First-in-human testing of a wirelessly controlled drug delivery microchip. Sci. Transl. Med. 4, 122ra121–122ra121 (2012).
Stevenson, C. L., Santini, J. T. & Langer, R. Reservoir-based drug delivery systems utilizing microtechnology. Adv. Drug Deliv. Rev. 64, 1590–1602 (2012).
Lee, S. H. et al. Implantable system for chronotherapy. Sci. Adv. 7, eabj4624 (2021).
Dagdeviren, C. et al. Miniaturized neural system for chronic, local intracerebral drug delivery. Sci. Transl. Med. 10, eaan2742 (2018).
Patroklou, G. et al. pH-responsive hydrogels: recent advances in pharmaceutical applications. Polymers 17, 1451 (2025).
Fu, D., Zhong, L., Xu, J., Mo, A. & Yang, M. Hydrazone-functionalized nanoscale covalent organic frameworks as a nanocarrier for pH-responsive drug delivery enhanced anticancer activity. RSC Adv. 14, 20799–20808 (2024).
Parshad, B. et al. Towards precision medicine using biochemically triggered cleavable conjugation. Commun. Chem. 8, 100 (2025).
Cong, X., Chen, J. & Xu, R. Recent progress in bio-responsive drug delivery systems for tumor therapy. Front. Bioeng. Biotechnol. 10, 916952 (2022).
Zhang, Z., Lu, Z., Yuan, Q., Zhang, C. & Tang, Y. ROS-responsive and active targeted drug delivery based on conjugated polymer nanoparticles for synergistic chemo-/photodynamic therapy. J. Mater. Chem. B 9, 2240–2248 (2021).
Chen, J. et al. The application of phenylboronic acid pinacol ester functionalized ROS-responsive multifunctional nanoparticles in the treatment of Periodontitis. J. Nanobiotechnology 22, 181 (2024).
Rinaldi, A. et al. Applications of the ROS-responsive thioketal linker for the production of smart nanomedicines. Polymers 14, 687 (2022).
Acharya, B. & Chikan, V. Pulse magnetic fields induced drug release from gold coated magnetic nanoparticle decorated liposomes. Magnetochemistry 6, 52 (2020).
Shi, J., Zhang, H., Jackson, J., Shademani, A. & Chiao, M. A robust and refillable magnetic sponge capsule for remotely triggered drug release. J. Mater. Chem. B 4, 7415–7422 (2016).
Carrelo, H. et al. Injectable thermoresponsive microparticle/hydrogel system with superparamagnetic nanoparticles for drug release and magnetic hyperthermia applications. Gels https://doi.org/10.3390/gels9120982 (2023).
Zhang, Z. Q. & Song, S. C. Multiple hyperthermia-mediated release of TRAIL/SPION nanocomplex from thermosensitive polymeric hydrogels for combination cancer therapy. Biomaterials 132, 16–27 (2017).
Desai, O. et al. Ultrasound-triggered drug release in vivo from antibubble-loaded macrophages. J. Control. Release 378, 365–376 (2025).
Chen, X. et al. Ultrasound-triggered on demand lidocaine release relieves postoperative pain. Front. Bioeng. Biotechnol. 10, 925047 (2022).
Almadani, I. F., Almadani, M. F., AlSawaftah, N., Abuwatfa, W. H. & Husseini, G. A. Nanocarriers responsive to light—A review. Micro 4, 827–844 (2024).
Kim, M. et al. Wireless, battery-free, remote photoactivation of caged-morphine for photopharmacological pain modulation without side effects. Biosens. Bioelectron. 281, 117440 (2025).
Xing, Y., Zeng, B. & Yang, W. Light responsive hydrogels for controlled drug delivery. Front. Bioeng. Biotechnol. 10, 1075670 (2022).
Proctor, C. M. et al. Electrophoretic drug delivery for seizure control. Sci. Adv. 4, eaau1291 (2018).
Arbring Sjöström, T. et al. A decade of iontronic delivery devices. Adv. Mater. Technol. 3, 1700360 (2018).
Strakosas, X., Seitanidou, M., Tybrandt, K., Berggren, M. & Simon, D. T. An electronic proton-trapping ion pump for selective drug delivery. Sci. Adv. 7, eabd8738 (2021).
Jonsson, A., Sjöström, T. A., Tybrandt, K., Berggren, M. & Simon, D. T. Chemical delivery array with millisecond neurotransmitter release. Sci. Adv. 2, e1601340 (2016).
Jonsson, A. et al. Therapy using implanted organic bioelectronics. Sci. Adv. 1, e1500039 (2015).
Poxson, D. J. et al. Regulating plant physiology with organic electronics. Proc. Natl. Acad. Sci. USA 114, 4597–4602 (2017).
Bernacka-Wojcik, I. et al. Implantable organic electronic ion pump enables ABA hormone delivery for control of stomata in an intact tobacco plant. Small 15, 1902189 (2019).
Li, H. et al. Remote-controlled wireless bioelectronics for fluoxetine therapy to promote wound healing in a porcine model. Adv. Mater. Technol. https://doi.org/10.1002/admt.202500040 (2024).
Seitanidou, M., Blomgran, R., Pushpamithran, G., Berggren, M. & Simon, D. T. Modulating inflammation in monocytes using capillary fiber organic electronic ion pumps. Adv. Healthc. Mater. 8, 1900813 (2019).
Waldherr, L. et al. Targeted chemotherapy of glioblastoma spheroids with an iontronic pump. Adv. Mater. Technol. 6, 2001302 (2021).
Handl, V. et al. Continuous iontronic chemotherapy reduces brain tumor growth in embryonic avian in vivo models. J. Control. Release 369, 668–683 (2024).
Yoo, H. et al. Ionic diode-based drug delivery system. Adv. Mater. 37, 2412377 (2025).
Seitanidou, M., Tybrandt, K., Berggren, M. & Simon, D. T. Overcoming transport limitations in miniaturized electrophoretic delivery devices. Lab Chip 19, 1427–1435 (2019).
Wang, J., Wang, X., Fan, X. & Chen, P. R. Unleashing the power of bond cleavage chemistry in living systems. ACS Cent. Sci. 7, 929–943 (2021).
Versteegen, R. M., Rossin, R., ten Hoeve, W., Janssen, H. M. & Robillard, M. S. Click to release: instantaneous doxorubicin elimination upon tetrazine ligation. Angew. Chem. Int. Ed. 52, 14112–14116 (2013).
Rossin, R. et al. Triggered drug release from an antibody-drug conjugate using fast “click-to-release” chemistry in mice. Bioconjug. Chem. 27, 1697–1706 (2016).
Ligthart, N. A. M. et al. A lysosome-targeted tetrazine for organelle-specific click-to-release chemistry in antigen presenting cells. J. Am. Chem. Soc. 145, 12630–12640 (2023).
Czuban, M. et al. Bio-orthogonal chemistry and reloadable biomaterial enable local activation of antibiotic prodrugs and enhance treatments against Staphylococcus aureus infections. ACS Cent. Sci. 4, 1624–1632 (2018).
Keppel, P., Hecko, S. & Mikula, H. Click-triggered bioorthogonal bond-cleavage reactions. Top. Curr. Chem. 383, 25 (2025).
Sarris, A. J. C. et al. Fast and pH-independent elimination of trans-cyclooctene by using aminoethyl-functionalized tetrazines. Chem. A Eur. J. 24, 18075–18081 (2018).
Poxson, D. J. et al. Capillary-fiber based electrophoretic delivery device. ACS Appl. Mater. Interfaces 11, 14200–14207 (2019).
Jia, M., Luo, L. & Rolandi, M. Correlating ionic conductivity and microstructure in polyelectrolyte hydrogels for bioelectronic devices. Macromol. Rapid Commun. 43, 2100687 (2022).
Keppel, P. et al. Tetrazine-triggered bioorthogonal cleavage of trans-cyclooctene-caged phenols using a minimal self-immolative linker strategy. ChemBioChem 23, e202200363 (2022).
Acknowledgements
This work has received funding from the European Union’s Horizon Europe Research and Innovation Programme under Grant Agreement 101099963 (bioSWITCH—M.B., D.T.S., R.S., L.W., H.M., J.B.) and the European Research Council under Grant Agreements 101117736 (Time2SWITCH—J.B.) and 101042881 (bioTARGET—H.M.). This research was funded in part by the Austrian Science Fund (FWF) [10.55776/J4304—J.B., 10.55776/I4623—H.M., 10.55776/Y1443—H.M.]. Additional funding was provided by the Knut and Alice Wallenberg Foundation (M.B., D.T.S., T.A.S.). We gratefully acknowledge the support of the PhD program Molecular Medicine (MOLMED) at Medical University of Graz (C.B.). L.W. received funding from the BioTechMed Graz Young Research Groups. We wish to thank Aneta Marková, Loïc Baudoin, and Peter Nadj for their assistance in the early development of multi-well iontronics, and Maria Seitanidou, Tobias Abrahamsson and David J. Poxson for assistance with preliminary tetrazine delivery experiments, and Adam El-Zoghbi for help with tetrazine synthesis. The authors acknowledge TU Wien Bibliothek for financial support through its Open Access Funding Programme.
Author information
Authors and Affiliations
Contributions
S.H. synthesized, characterized, and applied compounds for immobilization and the subsequent iontronic click-to-release (C2R) of CA4 and BSA, optimized prodrug activation and protein release assays, and drafted the manuscript, including figure preparation. M.E.J.V. optimized ion exchange membrane composition, developed and optimized protocols for ion exchange membrane fabrication, assembled iontronic devices, performed and optimized tetrazine delivery and step-function experiments, conducted assay quantification, data analysis, figure preparation, and contributed to manuscript writing. C.B. performed iontronic click-to-release (C2R) experiments with sulfo-cTCO-DMEDA-CA4 in solution and on beads, assisted with BSA release experiments, and contributed to data analysis, figure preparation, and manuscript writing. D.B. designed, fabricated, and optimized custom 3D-printed components, implemented device optimization strategies, performed tetrazine delivery experiments, supported data analysis and figure preparation, and contributed to manuscript writing. M.E.H. improved and implemented IEM fabrication protocols, assembled iontronic devices, developed and improved characterization setups, conducted tetrazine delivery and step-function experiments, and contributed to data analysis, figure preparation, and manuscript writing. N.P. synthesized and characterized fluorogenic rTCO probes and tetrazines, carried out iontronic tetrazine delivery experiments, assay development, and cyclic voltammetry measurements. R.B. supported 3D-printed component fabrication, device optimization, and tetrazine delivery, performed step-function tuning, and contributed to data analysis, figure preparation, and manuscript writing. P.K. synthesized and characterized cTCO-prodrugs and conducted all HPLC-MS-based release studies and contributed to manuscript writing. A.L. performed kinetic measurements for the C2R reaction. W.K. synthesized the multi-step cTCO linker. H.S.U. designed ion exchange membrane composition, developed iontronic device fabrication methods, designed and fabricated initial custom 3D-printed components. I.B.W. designed ion exchange membrane composition, developed and supervised iontronic device fabrication, designed initial characterization setup. T.A.S. designed ion exchange membrane composition, designed initial characterization setups, provided supervision, and secured funding. M.B. provided project supervision, secured funding, and contributed to manuscript writing. D.T.S. provided project supervision, secured funding, and contributed to manuscript writing. R.S. provided project supervision and contributed to manuscript writing. L.W. performed preliminary iontronic in vitro C2R experiments with sulfo-cTCO-DMEDA-CA4, designed in vitro studies, provided project supervision, and contributed to manuscript writing. H.M. conceived the study, co-led project supervision, developed the bioorthogonal chemistry and synthetic strategy, secured funding, and co-wrote the manuscript. J.B. conceived the study, co-led project supervision, developed the iontronic-C2R delivery concept, performed early C2R iontronic experiments, secured funding, and co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
I.B.W., T.A.S., M.B., D.T.S., H.M., and J.B. are shareholders in OBOE IPR AB, which owns patents related to this research and is the parent company of Iontronics AB. OBOE IPR AB is the applicant of the European patent EP-4114508 B1 (granted), which covers iontronic delivery systems relevant to the work reported in this manuscript. J.B., H.M., D.T.S., and M.B. are named inventors on this patent. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ismail Eş and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hecko, S., Vleugels, M.E.J., Bayer, C. et al. Iontronic click-to-release enables electrically controlled delivery of drugs and biomolecules beyond charge and size limitations. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70985-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70985-0