Abstract
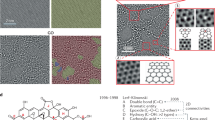
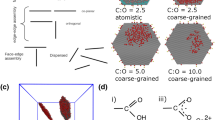
Graphene oxide (GO) has become an increasingly important industrial chemical and useful two-dimensional material. The complexity in both functional groups and heterostructure of GO offers its rich chemistry yet complicates its stability, dispersibility and processing property. Interactions with functional groups have been pioneered to explain these confusing properties of GO. However, the critical role of structural heterogeneity keeps elusive. Here, we report that the irreversible dispersibility of GO solid origins from the interlayer π-π stacking and the accessibility between conjugated domains. Experiments and simulations reveal that the exclusion of interlayer water leads to irreversible π-π stacking. This insight into the π-π stacking mechanism informs the design of selective gelation paths, enabling the scalable, continuous production of highly conductive graphene-based hydrogel for neural probes. Our work unveils a general mechanism for confusing dispersibility of GO and opens supramolecular interactions modulating methods for assembled structures and materials.
Similar content being viewed by others
Data availability
All other data supporting the findings of this study are available in the main text and the Supplementary Information. The data generated in this study are provided in the Source Data file. Source data are provided with this paper.
Code availability
All simulation input scripts, data files, and key output datasets generated in this study have been deposited in Figshare and are publicly available at https://doi.org/10.6084/m9.figshare.30880817.
References
Brodie, B. C. On the atomic weight of graphite. Philos. Trans. R. Soc. London. 149, 249–259 (1859).
Gao, W., Alemany, L. B., Ci, L. & Ajayan, P. M. New insights into the structure and reduction of graphite oxide. Nat. Chem. 1, 403–408 (2009).
Bagri, A. et al. Structural evolution during the reduction of chemically derived graphene oxide. Nat. Chem. 2, 581–587 (2010).
Kim, S. et al. Room-temperature metastability of multilayer graphene oxide films. Nat. Mater. 11, 544–549 (2012).
Kumar, P. V. et al. Scalable enhancement of graphene oxide properties by thermally driven phase transformation. Nat. Chem. 6, 151–158 (2014).
Wu, J., Lin, H., Moss, D. J., Loh, K. P. & Jia, B. Graphene oxide for photonics, electronics and optoelectronics. Nat. Rev. Chem. 7, 162–183 (2023).
Ren, W. & Cheng, H. M. The global growth of graphene. Nat. Nanotechnol. 9, 726–730 (2014).
Georgakilas, V. et al. Noncovalent functionalization of graphene and graphene oxide for energy materials, biosensing, catalytic, and biomedical applications. Chem. Rev. 116, 5464–5519 (2016).
Yousefi, N., Lu, X., Elimelech, M. & Tufenkji, N. Environmental performance of graphene-based 3D macrostructures. Nat. Nanotechnol. 14, 107–119 (2019).
Dreyer, D. R., Park, S., Bielawski, C. W. & Ruoff, R. S. The chemistry of graphene oxide. Chem. Soc. Rev. 39, 228–240 (2010).
Erickson, K. et al. Determination of the local chemical structure of graphene oxide and reduced graphene oxide. Adv. Mater. 22, 4467–4472 (2010).
Wang, Y. et al. Conformational phase map of two-dimensional macromolecular graphene oxide in solution. Matter 3, 230–245 (2020).
Gao, Y. et al. Falling-leaves stacking aggregation of two-dimensional macromolecular graphene oxide in solution. Langmuir 39, 12166–12173 (2023).
Yeh, C. N., Raidongia, K., Shao, J., Yang, Q. H. & Huang, J. On the origin of the stability of graphene oxide membranes in water. Nat. Chem. 7, 166–170 (2015).
Huang, H., Park, H. & Huang, J. Self-crosslinking of graphene oxide sheets by dehydration. Chem 8, 2432–2441 (2022).
Kim, J. et al. Graphene oxide sheets at interfaces. J. Am. Chem. Soc. 132, 8180–8186 (2010).
Lodge, T. P. & Ueki, T. Mechanically tunable, readily processable ion gels by self-assembly of block copolymers in ionic liquids. Acc. Chem. Res. 49, 2107–2114 (2016).
Gabriel, J. C. P. et al. Swollen liquid-crystalline lamellar phase based on extended solid-like sheets. Nature 413, 504–508 (2001).
Ming, X. et al. 2D-topology-seeded graphitization for highly thermally conductive carbon fibers. Adv. Mater. 34, 2201867 (2022).
Grimme, S. Do special noncovalent π-π stacking interactions really exist? Angew. Chem. Int. Ed. 47, 3430–3434 (2008).
Meyer, J. C. et al. The structure of suspended graphene sheets. Nature 446, 60–63 (2007).
Zhang, J. et al. Free folding of suspended graphene sheets by random mechanical stimulation. Phys. Rev. Lett. 104, 166805 (2010).
Cao, Y. et al. New structural insights into densely assembled reduced graphene oxide membranes. Adv. Funct. Mater. 32, 2201535 (2022).
Li, M., Zeng, F., Chang, H., Xu, B. & Wang, W. Aggregate structure evolution of low-rank coals during pyrolysis by in-situ X-ray diffraction. Int. J. Coal Geol. 116, 262–269 (2013).
Eda, G. et al. Blue photoluminescence from chemically derived graphene oxide. Adv. Mater. 22, 505–509 (2010).
Chien, C. T. et al. Tunable photoluminescence from graphene oxide. Angew. Chem. Int. Ed. 51, 6662–6666 (2012).
Yuan, W. Z. et al. Changing the behavior of chromophores from aggregation-caused quenching to aggregation-induced emission: development of highly efficient light emitters in the solid state. Adv. Mater. 22, 2159–2163 (2010).
Birks, J. B. Photophysics of Aromatic Molecules (Wiley, 1970).
Lakowicz, J. R. Principles of Fluorescence Spectroscopy (Springer, 2006).
Rafiee, J. et al. Wetting transparency of graphene. Nat. Mater. 11, 217–222 (2012).
Li, Z. et al. Effect of airborne contaminants on the wettability of supported graphene and graphite. Nat. Mater. 12, 925–931 (2013).
Compton, O. C. et al. Tuning the mechanical properties of graphene oxide paper and its associated polymer nanocomposites by controlling cooperative intersheet hydrogen bonding. ACS Nano 6, 2008–2019 (2012).
Qin, Z. & Buehler, M. Bioinspired design of functionalised graphene. Mol. Simulat. 38, 695–703 (2012).
Ling, S. et al. Design and function of biomimetic multilayer water purification membranes. Sci. Adv. 3, e1601939 (2017).
Liu, L., Zhang, R., Liu, Y., Tan, W. & Zhu, G. Insight into hydrogen bonds and characterization of interlayer spacing of hydrated graphene oxide. J. Mol. Model. 24, 137 (2018).
Medhekar, N. V., Ramasubramaniam, A., Ruoff, R. S. & Shenoy, V. B. Hydrogen bond networks in graphene oxide composite paper: structure and mechanical properties. ACS Nano 4, 2300–2306 (2010).
Do, K. U., Conner, A. V. & Wheeler, S. E. Making Sense of Heteroatom Effects in π–π Interactions. J. Am. Chem. Soc. 147, 32273–32286 (2025).
Zhao, X. et al. Polyelectrolyte-stabilized graphene oxide liquid crystals against salt, pH, and serum. Langmuir 30, 3715–3722 (2014).
Ciesielski, A. & Samorì, P. Supramolecular approaches to graphene: from self-assembly to molecule-assisted liquid-phase exfoliation. Adv. Mater. 28, 6030–6051 (2016).
Flory, P. J. & Rehner, J. Statistical mechanics of cross-linked polymer networks II. Swelling. J. Chem. Phys. 11, 521–526 (1943).
Flory, P. J. Principles of Polymer Chemistry (Cornell University Press, 1953).
Chang, D. et al. Reversible fusion and fission of graphene oxide-based fibers. Science 372, 614–617 (2021).
Sun, Z., Fang, S. & Hu, Y. H. 3D graphene materials: from understanding to design and synthesis control. Chem. Rev. 120, 10336–10453 (2020).
Yuk, H., Lu, B. & Zhao, X. Hydrogel bioelectronics. Chem. Soc. Rev. 48, 1642–1667 (2019).
Wu, J. et al. Adhesive anti-fibrotic interfaces on diverse organs. Nature 630, 360–367 (2024).
Won, D. et al. Laser-induced wet stability and adhesion of pure conducting polymer hydrogels. Nat. Electron. 7, 475–486 (2024).
Han, I. K. et al. Electroconductive, adhesive, non-swelling, and viscoelastic hydrogels for bioelectronics. Adv. Mater. 35, 2203431 (2023).
Li, G. et al. Highly conducting and stretchable double-network hydrogel for soft bioelectronics. Adv. Mater. 34, 2200261 (2022).
Acknowledgements
This work is financially supported by the National Key Research and Development Program of China (2022YFA1205300 and 2022YFA1205301 to Z.X.), National Natural Science Foundation of China (Nos. 52122301 to Z.X., 52090030 to C.G., 52272046 to Y.J.L., 12425201 to Z.P.X. and 52303284 to Y.W.), “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2023C01190 to Z.X.), the Fundamental Research Funds for the Central Universities (226-2024-00074 to Z.X. and 226-2023-00023 to Y.J.L.), International Research Center for X Polymers, International Campus, Zhejiang University (No. 130000-171207723/001/014 to Y.W.) and the fellowship of China National Postdoctoral Program for Innovative Talents (BX20230309 to D.C.). We thank staffs from Testing and Analysis Center of Department of Polymer Science and Engineering at Zhejiang University: Yi Guo for SEM, Yingying Zhang for CLSM and fluorescence spectrophotometer, Xinning Zhang for AFM and TEM.
Author information
Authors and Affiliations
Contributions
Conceptualization, Y. Gao, X. Ming, Z. Xu, Z. P. Xu; Methodology, Y. Gao, Y. Wang, Y. C. Liao, X. Ming, R. M. Li, Z. Xu, Z. P. Xu and X. L. Zhao; Investigation, Y. Gao, Y. C. Liao, Y. Wang, X. Ming, R. M. Li, D. Chang, L. Peng, H. Li, K. W. Li and M. Cao; Writing-Original Draft, Y. Gao, Y. C. Liao, Y. Wang, X. Ming, Z. Xu, Z. P. Xu, Y. J. Liu, C. Gao and R. M. Li; Writing-Review & Editing, Y. Gao, X. Ming, Y. C. Liao, Z. Xu, and Z. P. Xu; Funding Acquisition, Z. Xu and Z. P. Xu; Supervision, Z. Xu, Z. P. Xu and X. Ming. All authors participated in the discussion and comments of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks In Woo Cheong, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gao, Y., Wang, Y., Liao, Y. et al. π-π Stacking origin of irreversible dispersibility of graphene oxide. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71003-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71003-z