Abstract
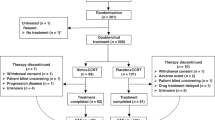
Induction chemotherapy (IC) followed by concurrent chemoradiotherapy (CCRT) is the standard treatment for locoregionally advanced nasopharyngeal carcinoma (LA-NPC); however, patients with a suboptimal response to IC, defined as detectable Epstein–Barr virus DNA and/or stable or progressive disease after IC, remain at high risk of treatment failure. Here we report an open-label, randomised, phase 2 trial evaluating whether adding nimotuzumab, a humanised anti–epidermal growth factor receptor antibody, to CCRT improves outcomes in this high-risk population. A total of 246 patients with untreated, non-keratinising, stage II–IVA LA-NPC were randomly assigned (1:1) to receive CCRT with or without nimotuzumab. The primary endpoint was 2-year progression-free survival (PFS); secondary endpoints included overall survival, distant metastasis–free survival, locoregional relapse–free survival, short-term response rate, and safety. At a median follow-up of 47 months, the 2-year PFS was 81.0% (90% confidence interval [CI], 74.3–86.1) in the nimotuzumab plus CCRT group and 80.8% (90% CI, 74.2–85.7) in the CCRT-alone group (hazard ratio, 0.93 [90% CI, 0.63–1.37]; p = 0.70). Survival outcomes were similar between groups, while low-grade rash occurred more frequently with nimotuzumab. These findings indicate that adding nimotuzumab to CCRT does not improve survival in patients with LA-NPC with a suboptimal response to IC, underscoring the need for predictive biomarkers and alternative therapeutic strategies. Trial registration: NCT04223024.
Similar content being viewed by others
Data availability
The deidentified individual participant data generated and analyzed in this study cannot be made publicly available due to patient privacy restrictions and ethical regulations. These data, along with the statistical analysis plan (included in Supplementary information) and a data dictionary, may be made available to qualified researchers upon reasonable request for non-commercial academic purposes upon reasonable request, subject to approval by the study steering committee and the institutional review board of Sun Yat-sen University Cancer Centre. Access will require a signed data-sharing agreement. Requests should be submitted in writing to the corresponding author (H.-Q.M.) and will be initially reviewed within 12 weeks. If approved, data provision will be arranged within 6 weeks after the execution of the data-sharing agreement, unless otherwise specified in the agreement. The study protocol is included in the Supplementary Information file. All other data supporting this work are included in the main article, supplementary, or source data file. Source data for figures and tables are provided with this paper. Source data are provided with this paper.
References
Zhang, Y. et al. Gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma. N. Engl. J. Med. 381, 1124–1135 (2019).
Sun, Y. et al. Induction chemotherapy plus concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: a phase 3, multicentre, randomised controlled trial. Lancet Oncol. 17, 1509–1520 (2016).
Chen, Y. P. et al. Chemotherapy in combination with radiotherapy for definitive-intent treatment of Stage II-IVA nasopharyngeal carcinoma: CSCO and ASCO Guideline. J. Clin. Oncol. 39, 840–859 (2021).
NCCN. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology: Head and Neck Cancers (Version 1.2025). Available at: https://www.nccn.org (accessed January 2025) (2025).
Liu, L. T. et al. The prognostic value of plasma Epstein-Barr Viral DNA and tumor response to neoadjuvant chemotherapy in advanced-stage nasopharyngeal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 93, 862–869 (2015).
Huang, C. L. et al. Plasma Epstein-Barr Virus DNA load after induction chemotherapy predicts outcome in locoregionally advanced nasopharyngeal carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 104, 355–361 (2019).
Lv, J. et al. Liquid biopsy tracking during sequential chemo-radiotherapy identifies distinct prognostic phenotypes in nasopharyngeal carcinoma. Nat. Commun. 10, 3941 (2019).
Ciardiello, F. & Tortora, G. A novel approach in the treatment of cancer: targeting the epidermal growth factor receptor. Clin. Cancer Res. 7, 2958–2970 (2001).
Mendelsohn, J. Targeting the epidermal growth factor receptor for cancer therapy. J. Clin. Oncol. 20, 1s–13s (2002).
Vermorken, J. B. et al. Platinum-based chemotherapy plus cetuximab in head and neck cancer. N. Engl. J. Med. 359, 1116–1127 (2008).
Guigay, J. et al. Cetuximab, docetaxel, and cisplatin versus platinum, fluorouracil, and cetuximab as first-line treatment in patients with recurrent or metastatic head and neck squamous-cell carcinoma (GORTEC 2014-01 TPExtreme): a multicentre, open-label, randomised, phase 2 trial. Lancet Oncol. 22, 463–475 (2021).
Zhang, M. et al. Long-term survival of patients with chemotherapy-naïve metastatic nasopharyngeal carcinoma receiving cetuximab plus docetaxel and cisplatin regimen. Front. Oncol. 10, 1011 (2020).
Bonner, J. A. et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 354, 567–578 (2006).
Gillison, M. L. et al. Radiotherapy plus cetuximab or cisplatin in human papillomavirus-positive oropharyngeal cancer (NRG Oncology RTOG 1016): a randomised, multicentre, non-inferiority trial. Lancet 393, 40–50 (2019).
Ang, K. K. et al. Randomized phase III trial of concurrent accelerated radiation plus cisplatin with or without cetuximab for stage III to IV head and neck carcinoma: RTOG 0522. J. Clin. Oncol. 32, 2940–2950 (2014).
Mesía, R. et al. Chemoradiotherapy with or without panitumumab in patients with unresected, locally advanced squamous-cell carcinoma of the head and neck (CONCERT-1): a randomised, controlled, open-label phase 2 trial. Lancet Oncol. 16, 208–220 (2015).
Chua, D. T., Nicholls, J. M., Sham, J. S. & Au, G. K. Prognostic value of epidermal growth factor receptor expression in patients with advanced stage nasopharyngeal carcinoma treated with induction chemotherapy and radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 59, 11–20 (2004).
Ma, B. B. Y. et al. A phase II study of concurrent cetuximab-cisplatin and intensity-modulated radiotherapy in locoregionally advanced nasopharyngeal carcinoma. Ann. Oncol. 23, 1287–1292 (2012).
Ramakrishnan, M. S. et al. Nimotuzumab, a promising therapeutic monoclonal for treatment of tumors of epithelial origin. mAbs 1, 41–48 (2009).
Diaz Miqueli, A. et al. Biological activity in vitro of anti-epidermal growth factor receptor monoclonal antibodies with different affinities. Hybridoma 26, 423–431 (2007).
Patil, V. M. et al. A randomized phase 3 trial comparing nimotuzumab plus cisplatin chemoradiotherapy versus cisplatin chemoradiotherapy alone in locally advanced head and neck cancer. Cancer 125, 3184–3197 (2019).
Zhao, C. et al. Anti-epidermal growth factor receptor (EGFR) monoclonal antibody combined with cisplatin and 5-fluorouracil in patients with metastatic nasopharyngeal carcinoma after radical radiotherapy: a multicentre, open-label, phase II clinical trial. Ann. Oncol. 30, 637–643 (2019).
Liu, Z. G. et al. Nimotuzumab combined with concurrent chemoradiotherapy in locally advanced nasopharyngeal carcinoma: a retrospective analysis. Oncotarget 7, 24429–24435 (2016).
Fangzheng, W. et al. Long-term use of nimotuzumab in combination with intensity-modulated radiotherapy and chemotherapy in the treatment of locoregionally advanced nasopharyngeal carcinoma: experience of a single institution. Oncol. Res. 26, 277–287 (2018).
Yao, J. J. et al. Comparing treatment outcomes of concurrent chemoradiotherapy with or without nimotuzumab in patients with locoregionally advanced nasopharyngeal carcinoma. Cancer Biol. Ther. 19, 1102–1107 (2018).
Cai, Z. et al. Concurrent chemoradiotherapy combined with nimotuzumab in stage III-IVa nasopharyngeal carcinoma: a retrospective analysis. J. Cancer Res. Clin. Oncol. 149, 2327–2344 (2023).
Wang, R. J., Ke, R. Q., Yu, Y. F., Lu, G. Z. & Wu, S. G. Addition of nimotuzumab to concurrent chemoradiotherapy after induction chemotherapy improves outcomes of patients with locally advanced nasopharyngeal carcinoma. Front. Pharmacol. 15, 1366853 (2024).
Huang, J. F. et al. Induction chemotherapy followed by concurrent chemoradiation and nimotuzumab for locoregionally advanced nasopharyngeal carcinoma: preliminary results from a phase II clinical trial. Oncotarget 8, 2457–2465 (2017).
Niu, X. et al. Anti-epidermal growth factor receptor (EGFR) monoclonal antibody combined with chemoradiotherapy for induction chemotherapy resistant locally advanced nasopharyngeal carcinoma: a prospective phase II study. Transl. Oncol. 39, 101797 (2024).
Caudell, J. J. et al. NCCN Guidelines® Insights: Head and Neck Cancers, Version 1.2022. J. Natl. Compr. Cancer Netw. 20, 224–234 (2022).
Zhang, Y. et al. Final overall survival analysis of gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma: a multicenter, randomized phase III trial. J. Clin. Oncol. 40, 2420–2425 (2022).
Petit, C. et al. Role of chemotherapy in patients with nasopharynx carcinoma treated with radiotherapy (MAC-NPC): an updated individual patient data network meta-analysis. Lancet Oncol. 24, 611–623 (2023).
Peng, H. et al. Optimize the cycle of neoadjuvant chemotherapy for locoregionally advanced nasopharyngeal carcinoma treated with intensity-modulated radiotherapy: a propensity score matching analysis. Oral Oncol. 62, 78–84 (2016).
Guo, S. S. et al. Reduced-dose radiotherapy for Epstein-Barr virus DNA selected staged III nasopharyngeal carcinoma: a single-arm, phase 2 trial. Eur. J. Cancer 194, 113336 (2023).
Hui, E. P. et al. Randomized phase II trial of concurrent cisplatin-radiotherapy with or without neoadjuvant docetaxel and cisplatin in advanced nasopharyngeal carcinoma. J. Clin. Oncol. 27, 242–249 (2009).
Liu, T. et al. Management of suboptimal response to induction chemotherapy in locoregionally advanced nasopharyngeal carcinoma: Re-induction therapy or direct to Radiotherapy? Radiother. Oncol. 163, 185–191 (2021).
Lv, J. et al. Longitudinal on-treatment circulating tumor DNA as a biomarker for real-time dynamic risk monitoring in cancer patients: the EP-SEASON study. Cancer Cell 42, 1401–1414.e1404 (2024).
Guo, S. S. et al. Induction vs adjuvant chemoradiotherapy in patients with high-risk N2 to N3 nasopharyngeal carcinoma: a phase 3 randomized clinical trial. JAMA Oncol. 11, 864–873 (2025).
Rehman, S. K. & O’Brien, C. A. Persister cells that survive chemotherapy are pinpointed. Nature 608, 675–676 (2022).
Zhou, X. et al. Survival impact of increasing time to IMRT initiation following induction chemotherapy in nasopharyngeal carcinoma: a propensity score-matched analysis. Oral. Oncol. 122, 105506 (2021).
Bourhis, J. et al. Rapid tumor cell proliferation after induction chemotherapy in oropharyngeal cancer. Laryngoscope 104, 468–472 (1994).
Chen, X., Liang, R. & Zhu, X. Anti-EGFR therapies in nasopharyngeal carcinoma. Biomed. Pharmacother. 131, 110649 (2020).
Fan, Z., Baselga, J., Masui, H. & Mendelsohn, J. Antitumor effect of anti-epidermal growth factor receptor monoclonal antibodies plus cis-diamminedichloroplatinum on well established A431 cell xenografts. Cancer Res. 53, 4637–4642 (1993).
Huang, S. M., Bock, J. M. & Harari, P. M. Epidermal growth factor receptor blockade with C225 modulates proliferation, apoptosis, and radiosensitivity in squamous cell carcinomas of the head and neck. Cancer Res. 59, 1935–1940 (1999).
Ciardiello, F. et al. Antitumor activity of sequential treatment with topotecan and anti-epidermal growth factor receptor monoclonal antibody C225. Clin. Cancer Res. 5, 909–916 (1999).
Inoue, K. et al. Paclitaxel enhances the effects of the anti-epidermal growth factor receptor monoclonal antibody ImClone C225 in mice with metastatic human bladder transitional cell carcinoma. Clin. Cancer Res. 6, 4874–4884 (2000).
Prewett, M. C. et al. Enhanced antitumor activity of anti-epidermal growth factor receptor monoclonal antibody IMC-C225 in combination with irinotecan (CPT-11) against human colorectal tumor xenografts. Clin. Cancer Res. 8, 994–1003 (2002).
Chan, A. T. et al. Multicenter, phase II study of cetuximab in combination with carboplatin in patients with recurrent or metastatic nasopharyngeal carcinoma. J. Clin. Oncol. 23, 3568–3576 (2005).
Tang, L. Q. et al. Prospective study of tailoring whole-body dual-modality [18F]fluorodeoxyglucose positron emission tomography/computed tomography with plasma Epstein-Barr virus DNA for detecting distant metastasis in endemic nasopharyngeal carcinoma at initial staging. J. Clin. Oncol. 31, 2861–2869 (2013).
Shao, J. Y. et al. Comparison of plasma Epstein-Barr virus (EBV) DNA levels and serum EBV immunoglobulin A/virus capsid antigen antibody titers in patients with nasopharyngeal carcinoma. Cancer 100, 1162–1170 (2004).
An, X. et al. Plasma Epstein-Barr virus DNA level strongly predicts survival in metastatic/recurrent nasopharyngeal carcinoma treated with palliative chemotherapy. Cancer 117, 3750–3757 (2011).
Liu, S. L. et al. Liposomal paclitaxel versus docetaxel in induction chemotherapy using Taxanes, cisplatin and 5-fluorouracil for locally advanced nasopharyngeal carcinoma. BMC Cancer 18, 1279 (2018).
Wang, Y. et al. A triplet chemotherapy regimen of cisplatin, fluorouracil and paclitaxel for locoregionally recurrent nasopharyngeal carcinoma cases contraindicated for re-irradiation/surgery. Expert Opin. Pharmacother. 17, 1585–1590 (2016).
Lai, S. Z. et al. How does intensity-modulated radiotherapy versus conventional two-dimensional radiotherapy influence the treatment results in nasopharyngeal carcinoma patients? Int. J. Radiat. Oncol. Biol. Phys. 80, 661–668 (2011).
Zhang, Q., Huang, S. & Lin, S. Delineation of clinical target volume in nasopharyngeal carcinoma. Holist. Integr. Oncol. 2, 22 (2023).
Rubinstein, L. V. et al. Design issues of randomized phase II trials and a proposal for phase II screening trials. J. Clin. Oncol. 23, 7199–7206 (2005).
Schoenfeld, D. Partial residuals for the proportional hazards regression model. Biometrika 69, 239–241 (1982).
Fine J. P. & Gray R. J. A proportional hazards model for the subdistribution of a competing risk. J. Am. Stat. Assoc. 94, 496–509 (1999).
Rytgaard, H. C. W., Eriksson, F. & van der Laan, M. J. Estimation of time-specific intervention effects on continuously distributed time-to-event outcomes by targeted maximum likelihood estimation. Biometrics https://doi.org/10.1111/biom.13856 (2023).
Fisher, D. J., Copas, A. J., Tierney, J. F. & Parmar, M. K. A critical review of methods for the assessment of patient-level interactions in individual participant data meta-analysis of randomized trials, and guidance for practitioners. J. Clin. Epidemiol. 64, 949–967 (2011).
Acknowledgements
The Elsevier Language Editing Service was used to assist with manuscript editing during drafting. This study was funded by grants from the National Key Research and Development Programme of China (2022YFC2505800, and 2022YFC2705005), National Natural Science Foundation of China (82173287, 82372980, 82203776, 82203125, 82222050, 82272739, 82272882, 82073003, 82003267, 82002852, 82373258, 82361168664, 8247101588, 82473038, and 32200651), Guangdong Basic and Applied Basic Research Foundation (2021B1515230002, and 2023B1515120092), Science and Technology Programme of Guangzhou (202201011561, 2023A04J2127, and 2024B03J1248), Sun Yat-sen University Clinical Research 5010 Programme (2024003, 2015021, 2019023, and 2017010), Postdoctoral Innovative Talent Support Programme (BX20220361), Planned Science and Technology Project of Guangdong Province (2019B020230002), Key Youth Teacher Cultivating Programme of Sun Yat-sen University (20ykzd24), and Fundamental Research Funds for the Central Universities.
Author information
Authors and Affiliations
Contributions
H.-Q.M. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. L.-T.L., X.-S.S., T.-T.Q., X.-Y.L., L.G., and H.-Y.M. contributed equally to this work. H.-Q.M., L.-Q.T., and Q.-Y.C. jointly supervised this work. Concept and design: L.-T.L., H.-Q.M., L.-Q.T., Q.-Y.C. Acquisition, analysis, or interpretation of data: L.-T.L., X.-S.S., T.-T.Q., X.-Y.L., L.G., H.-Y.M. Drafting of the manuscript: L.-T.L., X.-S.S., S.-S.G., S.-L.L., Y.H., D.-H.L., R.S. Critical review of the manuscript for important intellectual content: L.-T.L., X.-S.S., G.-D.J., J.-B.L., Q.L., P.W., Y.-J.L., J.C., Y.-F.L. Statistical analysis: L.-T.L., H.C., W.-X.X. Administrative, technical, or material support: F.Q., J.-H.Y. Trial management and toxicity review: Q.Y, D.-X.W., J.-J.Y., C.Z. Obtained funding: H.-Q.M. Supervision: Q.-Y.C., L.-Q.T., H.-Q.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Cristina Gurizzan and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, LT., Sun, XS., Quan, TT. et al. Concurrent chemoradiotherapy plus nimotuzumab versus chemoradiotherapy alone for locoregionally advanced nasopharyngeal carcinoma with a suboptimal response to induction chemotherapy: a randomized phase 2 trial. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71019-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71019-5