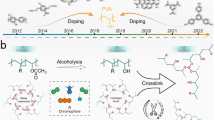
Abstract
Ultralong organic phosphorescence (UOP) and especially high-temperature phosphorescence (HTP) are difficult to be achieved in pure organic nontraditional luminogens (NTLs). Herein, we develop a strategy to prepare UOP materials by solvent exchange of an NTLs-based hydrogel with a deep eutectic solvent (DES), followed by wet annealing. The obtained polyacrylamide-DES complexes exhibit afterglows up to 9.5 s, room-temperature phosphorescence (RTP) lifetimes up to 622.5 ms, and high phosphorescence quantum yields of 17.6%. Excitingly, they also exhibit HTP with a lifetime of 366.2 ms even at 120°C and exhibit highly stable UOP in organic solvents. DESs form strong hydrogen bonding with the polymer matrix, the heavy atom effect of bromine in the DESs promotes the occurrence of RTP, and the rigidified and stable structures enable the HTP of the complexes. This work provides a universal strategy for preparing non-aromatic UOP materials for wide applications.
Similar content being viewed by others
Data availability
The data generated in this study are provided in the main text, Supplementary Information and Source Data file. Atomic coordinates of the optimized computational models of DES (TD), PAM-PAM and PAM-TD are available in Supplementary Data. Data is available from the corresponding author on request. Source data are provided with this paper.
References
Zhao, W. J., He, Z. K. & Tang, B. Z. Room-temperature phosphorescence from organic aggregates. Nat. Rev. Mater. 5, 869–885 (2020).
Chen, C. J. & Liu, B. Enhancing the performance of pure organic room-temperature phosphorescent luminophores. Nat. Commun. 10, 2111 (2019).
Ye, W. P. et al. Confining isolated chromophores for highly efficient blue phosphorescence. Nat. Mater. 20, 1539–1544 (2021).
Kwon, M. S. et al. Suppressing molecular motions for enhanced room-temperature phosphorescence of metal-free organic materials. Nat. Commun. 6, 8947 (2015).
Gong, Y. Y. et al. Achieving persistent room temperature phosphorescence and remarkable mechanochromism from pure organic luminogens. Adv. Mater. 27, 6195–6201 (2015).
Dou, X. Y. et al. Advances in polymer-based organic room-temperature phosphorescence materials. Adv. Funct. Mater. 34, 2314069 (2024).
An, Z. F. et al. Stabilizing triplet excited states for ultralong organic phosphorescence. Nat. Mater. 14, 685–690 (2015).
Liang, Y. H. et al. Enabling highly robust full-color ultralong room-temperature phosphorescence and stable white organic afterglow from polycyclic aromatic hydrocarbons. Angew. Chem. Int. Ed. 63, e202318516 (2024).
Cai, S. Z. et al. Ultralong organic phosphorescent foams with high mechanical strength. J. Am. Chem. Soc. 143, 16256–16263 (2021).
Chen, C. J. et al. Carbazole isomers induce ultralong organic phosphorescence. Nat. Mater. 20, 175–180 (2021).
Singh, M. et al. Recent advances of cocrystals with room temperature phosphorescence. Adv. Opt. Mater. 9, 2002197 (2021).
Kongasseri, A. A., Ansari, S. N., Garain, S., Wagalgave, S. M. & George, S. J. Revisiting organic charge-transfer cocrystals for wide-range tunable, ambient phosphorescence. Chem. Sci. 14, 12548–12553 (2023).
Wang, Y. S. et al. High performance of simple organic phosphorescence host-guest materials and their application in time-resolved bioimaging. Adv. Mater. 33, 2007811 (2021).
Xia, Y. et al. Host-guest doping in flexible organic crystals for room-temperature phosphorescence. Angew. Chem. Int. Ed. 62, e202217547 (2023).
Cai, S. Z. et al. Enabling long-lived organic room temperature phosphorescence in polymers by subunit interlocking. Nat. Commun. 10, 4247 (2019).
Cai, S. Z. et al. Visible-light-excited ultralong organic phosphorescence by manipulating intermolecular interactions. Adv. Mater. 29, 1701244 (2017).
Lucenti, E. et al. H-aggregates granting crystallization-induced emissive behavior and ultralong phosphorescence from a pure organic Molecule. J. Phys. Chem. Lett. 8, 1894–1898 (2017).
Zheng, H., Zhang, Z. Y., Cai, S. Z., An, Z. F. & Huang, W. Enhancing purely organic room temperature phosphorescence via supramolecular self-assembly. Adv. Mater. 36, 2311922 (2024).
Bian, L. F. et al. Simultaneously enhancing efficiency and lifetime of ultralong organic phosphorescence materials by molecular self-assembly. J. Am. Chem. Soc. 140, 10734–10739 (2018).
Feng, W. H. et al. Supramolecular entanglement driven emissive aggregate densification enabling room-temperature phosphorescence hydrogels with ultrastretchability and crack-tolerance. Angew. Chem. Int. Ed. 64, e202505192 (2025).
Shi, H. F. et al. Ultralong organic phosphorescence: From material design to applications. Acc. Chem. Res. 55, 3445–3459 (2022).
Wang, H. et al. Abnormal thermally-stimulated dynamic organic phosphorescence. Nat. Commun. 15, 2134 (2024).
Singh, M. et al. Achieving high-temperature phosphorescence by organic cocrystal engineering. Angew. Chem. Int. Ed. 63, e202319694 (2024).
Chen, Y., Xu, Z. Z., Yu, M. & Fu, H. B. High-temperature dynamic organic phosphorescence based on cyclodextrins supramolecular assemblies. ACS Appl. Mater. Interfaces 17, 24351–24358 (2025).
Luo, W. Z. et al. Leveraging multivalent assembly towards high-temperature liquid-phase phosphorescence. Angew. Chem. Int. Ed. 64, e202423650 (2025).
Guan, L. H. et al. Ultra-stable and colorful persistent organic phosphorescence of host-guest composites under solution, UV irradiation, and high temperature. Microchem. J. 212, 113505 (2025).
Chen, K. J. et al. Twofold rigidity activates ultralong organic high-temperature phosphorescence. Nat. Commun. 15, 1269 (2024).
Tang, S. M. et al. Cycloolefin copolymers with a multiply rigid structure for protecting triplet exciton from thermo- and moisture-quenching. Adv. Mater. 37, 2416397 (2025).
Zhang, M. et al. Dynamic organic phosphorescence glass by rigid-soft coupling. Angew. Chem. Int. Ed. 64, e202415250 (2025).
Liao, P. L., Huang, J. B., Yan, Y. & Tang, B. Z. Clusterization-triggered emission (CTE): one for all, all for one. Mater. Chem. Front. 5, 6693–6717 (2021).
Tang, S. X. et al. Nonconventional luminophores: Characteristics, advancements and perspectives. Chem. Soc. Rev. 50, 12616–12655 (2021).
Wu, J. Y. et al. Multiple hydrogen-bonding induced nonconventional red fluorescence emission in hydrogels. Nat. Commun. 15, 3482 (2024).
Zhang, X. M., Bai, Y. H., Deng, J. W., Zhuang, P. F. & Wang, H. L. Effects of nonaromatic through-bond conjugation and through-space conjugation on the photoluminescence of nontraditional luminogens. Aggregate 5, e517 (2024).
Zhou, Q. et al. Emission mechanism understanding and tunable persistent room temperature phosphorescence of amorphous nonaromatic polymers. Mater. Chem. Front. 3, 257–264 (2019).
Wang, Y. Z. et al. Nonconventional luminophores with unprecedented efficiencies and color-tunable afterglows. Mater. Horiz. 7, 2105–2112 (2020).
Deng, J. W. et al. Yellow and orange-red room-temperature phosphorescence from amorphous nonaromatic polymers. Adv. Opt. Mater. 11, 2300715 (2023).
Deng, J. W. et al. Persistent room-temperature phosphorescent organohydrogels based on nonaromatic luminogens crosslinked by hydrogen bonds. Adv. Funct. Mater. 34, 2408821 (2024).
Deng, J. W. et al. Room-temperature phosphorescent tough hydrogels based on ionically crosslinked nonaromatic polymers. Adv. Funct. Mater. 34, 2308420 (2024).
Xiao, J. S. et al. Stable persistent room-temperature phosphorescent hydrogels based on ionically crosslinked nonaromatic carboxylate polymers. Small 20, 2405615 (2024).
Xie, W. D. et al. Nonaromatic persistent room-temperature phosphorescent hydrogels with shape memory behavior and ultrahigh elastic moduli based on partially hydrolyzed polyacrylonitrile. Adv. Funct. Mater. 35, 2504411 (2025).
Ju, H. Q. et al. Polymerization-induced crystallization of dopant molecules: an efficient strategy for room-temperature phosphorescence of hydrogels. J. Am. Chem. Soc. 145, 3763–3773 (2023).
Fan, X. Z. et al. Intrinsic room-temperature phosphorescent hydrogel driven by phase separation and glass transition. Adv. Opt. Mater. 13, 2402627 (2025).
Yao, P. Q. et al. Environmentally stable, robust, adhesive, and conductive supramolecular deep eutectic gels as ultrasensitive flexible temperature sensor. Adv. Mater. 35, 2300114 (2023).
Xu, C. G. et al. Ultrastrong eutectogels engineered via integrated mechanical training in molecular and structural engineering. Nat. Commun. 16, 2589 (2025).
Tang, N. et al. Evolutionary reinforcement of polymer networks: A stepwise-enhanced strategy for ultrarobust eutectogels. Adv. Mater. 36, 2309576 (2024).
Du, S. et al. Ultratough and highly conductive supramolecular Poly(Vinyl Alcohol) eutectogels via a sequentially enhanced strategy. Adv. Funct. Mater. 35, 2409726 (2025).
Criado-Gonzalez, M. et al. Mixed conductive, injectable, and fluorescent supramolecular eutectogel composites. Angew. Chem. Int. Ed. 62, e202301489 (2023).
Li, C. C. et al. Photoswitchable and reversible fluorescent eutectogels for conformal information encryption. Angew. Chem. Int. Ed. 62, e202313971 (2023).
Jiang, X. et al. Solvation chemistry enables eutectogels with ultrahigh autofluorescence, toughness, and adhesion under extreme environments. Adv. Funct. Mater. 35, 2412313 (2025).
Bo, C. C. et al. Ultralong room temperature phosphorescence emission from gels induced by multiple confinement effects. Laser Photon. Rev. 19, 2400997 (2025).
Xiao, J. S. et al. Hydrothermal processing as an effective strategy for producing coordinated non-aromatic hydrogels with persistent and reversible UV-activated room-temperature phosphorescence. Angew. Chem. Int. Ed. 64, e202513371 (2025).
Wu, J., Wu, B. H., Xiong, J. Q., Sun, S. T. & Wu, P. Y. Entropy-mediated polymer-cluster interactions enable dramatic thermal stiffening hydrogels for mechanoadaptive smart fabrics. Angew. Chem. Int. Ed. 61, e202204960 (2022).
Shokri, S., Ebrahimi, N. & Sadeghi, R. Combined experimental and computational investigation of tetrabutylammonium bromide-carboxylic acid-based deep eutectic solvents. J. Mol. Graph. 131, 108805 (2024).
Chen, L. et al. Rapid, accurate, and simultaneous measurement of water and oil contents in the fried starchy system using low-field NMR. Food Chem. 233, 525–529 (2017).
Xu, Z. R. et al. Full-color programmable high temperature afterglow polymers based on single-molecule emitters. Nat. Commun. 16, 9668 (2025).
Li, Z. et al. Sensitive mechanical compression-patterned phosphorescence in the rubbery state of cross-linked polymer network. Nat. Commun. 16, 10163 (2025).
Bauernschmitt, R. & Ahlrichs, R. Treatment of electronic excitations within the adiabatic approximation of time dependent density functional theory. Chem. Phys. Lett. 256, 454–464 (1996).
Adamo, C. & Barone, V. Toward reliable density functional methods without adjustable parameters: The PBE0 model. J. Chem. Phys. 110, 6158–6170 (1999).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Neese, F. The ORCA program system. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2, 73–78 (2012).
Lu, T. & Chen, Q. X. Interaction region indicator: A simple real space function clearly revealing both chemical bonds and weak interactions. Chem. Method. 1, 231–239 (2021).
Humphrey, W., Dalke, A. & Schulten, K. VMD: Visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Lu, T. & Chen, Q. X. Shermo: A general code for calculating molecular thermochemistry properties,. Comput. Theor. Chem. 1200, 113249 (2021).
Acknowledgements
We are grateful for financial support from the National Natural Science Foundation of China (Grant No. 22472010, H.W.) and the Program for Changjiang Scholars and Innovative Research Team (PCSIRT) at the University.
Author information
Authors and Affiliations
Contributions
X.Z. conceived the study, performed the experiments, conducted data analysis and wrote the manuscript. Y.B. carried out theoretical calculations. G.Q. participated in the experiments. Y.S. participated in the conception of the study. W.X. analyzed the data. J.X. and J.D. participated in the revision of the manuscript. H.D. provided related drawing supports. Y.Z corrected the manuscript. H.W. conceived, supervised the study and revised the manuscript. All authors have agreed with the manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Haoke Zhang, who co-reviewed with Xiong Liu; and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhong, X., Bai, Y., Qiao, G. et al. Nonaromatic polymer-deep eutectic solvent complexes with ultralong room-temperature and high-temperature phosphorescence. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71041-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71041-7