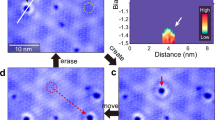
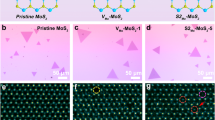
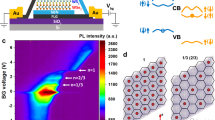
Abstract
Rapid recombination of photogenerated carriers and weak driving forces to inject hot electrons are critical bottlenecks in solar-driven ammonia borane hydrolysis. Herein, aided by machine learning, plasmon polarization-induced multi-field coupling is developed to enhance ammonia borane hydrolytic activity. The reconstructed surface unsaturated Moδ+ active sites exhibit well activity and high stability over 100 hours in AB hydrolysis, which deliver a turnover frequency up to 5806 min-1, representing competitiveness compared to non-noble and noble-metal based catalysts ever reported. It is verified that the polarized electric field facilitates carrier separation through incorporating polarization components (Ov and -OH), thereby promoting electron accumulation around Moδ+ active sites. Meanwhile, the local electric field enables highly delocalized hot electrons through plasmon oscillation, thus lowering the reaction barrier between Moδ+ and AB. In this work, the hot electrons are efficiently channeled via an enhanced feedback pathway, facilitating their transfer into B-H antibonding orbitals toward boosted AB hydrolysis.
Similar content being viewed by others
Data availability
All data generated in this study are provided in the Supplementary Information/Source Data file. The data that support the findings of this study are available from the corresponding author upon request. Source data are provided with this paper.
References
Fu, H. et al. A scalable solar-driven photocatalytic system for separated H2 and O2 production from water. Nat. Commun. 16, 990 (2025).
Guo, S. et al. Boosting photocatalytic hydrogen production from water by photothermally induced biphase systems. Nat. Commun. 12, 1343 (2021).
Jin, S. et al. 2D MoB MBene: an efficient co-catalyst for photocatalytic hydrogen production under visible light. ACS Nano 18, 12524–12536 (2024).
Liu, Y. et al. Cu2O/2D COFs core/shell nanocubes with antiphotocorrosion ability for efficient photocatalytic hydrogen evolution. ACS Nano 17, 5994–6001 (2023).
Xu, W. et al. Metal/metal-organic framework interfacial ensemble-induced dual site catalysis towards hydrogen generation. App. Catal. B Environ. 286, 119946 (2021).
Khanam, S. & Rout, S. K. Plasmonic metal/semiconductor heterostructure for visible light-enhanced H2 production. ACS Omega 7, 25466–25475 (2022).
Rej, S. et al. Facet-dependent and light-assisted efficient hydrogen evolution from ammonia borane using gold-palladium core-shell nanocatalysts. Angew. Chem. Int. Ed. 55, 7222–7226 (2016).
Shao, W. et al. Symmetry breaking in monometallic nanocrystals toward broadband and direct electron transfer enhanced plasmonic photocatalysis. Adv. Funct. Mater. 31, 2006738 (2020).
Song, J. et al. Remarkably boosting catalytic H2 evolution from ammonia borane through the visible-light-driven synergistic electron effect of non-plasmonic noble-metal-free nanoparticles and photoactive metal-organic frameworks. App. Catal. B Environ. 225, 424–432 (2018).
Zhang, S. et al. Visible-light-driven multichannel regulation of local electron density to accelerate activation of O-H and B–H bonds for ammonia borane hydrolysis. ACS Catal. 10, 14903–14915 (2020).
Chen, W. et al. Restructuring the interfacial active sites to generalize the volcano curves for platinum-cobalt synergistic catalysis. Nat. Commun. 15, 8995 (2024).
Chen, Y. et al. Practical H2 supply from ammonia borane enabled by amorphous iron domain. Nat. Commun. 15, 9113 (2024).
Wan, C. et al. Silica confinement for stable and magnetic Co-Cu alloy nanoparticles in nitrogen-doped carbon for enhanced hydrogen evolution. Angew. Chem. Int. Ed. 63, e202404505 (2024).
Li, Y. et al. Ruthenium nanoparticles anchored on cobalt oxide married with nitrogen and phosphorus Co-doped carbon for hydrolytic dehydrogenation of ammonia borane. ACS Catal. 15, 9158–9170 (2025).
Fu, F. et al. Highly selective and sharp volcano-type synergistic Ni2Pt@ZIF-8-catalyzed hydrogen evolution from ammonia borane hydrolysis. J. Am. Chem. Soc. 140, 10034–10042 (2018).
Guan, S. et al. Efficient hydrogen generation from ammonia borane hydrolysis on a tandem ruthenium-platinum-titanium catalyst. Angew. Chem. Int. Ed. 63, e202408193 (2024).
Xie, S. et al. Synergizing Mg single atoms and Ru nanoclusters for boosting the ammonia borane hydrolysis to produce hydrogen. Angew. Chem. Int. Ed. 64, e202424316 (2025).
Zhu, A. et al. Geminal synergy in Pt-Co dual-atom catalysts: from synthesis to photocatalytic hydrogen production. J.Am.Chem.Soc. 146, 33002–33011 (2024).
Li, J. et al. Sub-2 nm ternary metallic alloy encapsulated within montmorillonite interlayers for efficient hydrogen generation from ammonia borane hydrolysis. ACS Catal. 14, 14665–14677 (2024).
Shen, J. et al. Efficient generation of negative hydrogen with bimetallic-ternary-structured catalysts for nitrobenzene hydrogenation. Angew. Chem. Int. Ed. 64, e202423626 (2025).
Wen, M. et al. Non-noble-metal nanoparticle supported on metal-organic framework as an efficient and durable catalyst for promoting H2 production from ammonia borane under visible light irradiation. ACS Appl. Mater. Interfaces. 8, 21278–21284 (2016).
Yang, X. J. et al. Non-noble metallic nanoparticles supported on titania spheres as catalysts for hydrogen generation from hydrolysis of ammonia borane under ultraviolet light irradiation. Int. J. Hydrog. Energy. 43, 16556–16565 (2018).
Guo, J. et al. Graphene supported Pt-Ni bimetallic nanoparticles for efficient hydrogen generation from KBH4/NH3BH3 hydrolysis. Int. J. Hydrog. Energy. 47, 11601–11610 (2022).
Liu, K. et al. Enhanced catalytic behavior of h-BN supported CuNi bimetallic catalysts in hydrolytic dehydrogenation of NH3BH3. Int. J. Hydrogen Energy. 47, 33741–33753 (2022).
Wang, Y. et al. Co-Mo-B nanoparticles supported on carbon cloth as effective catalysts for the hydrolysis of ammonia borane. Int. J. Hydrogen Energy. 45, 14418–14427 (2020).
Yang, H. et al. One-pot MOFs-encapsulation derived in-doped ZnO@In2O3 hybrid photocatalyst for enhanced visible-light-driven photocatalytic hydrogen evolution. Adv. Sustain. Syst. 7, 2200443–2200454 (2022).
Liu, X. et al. Full-spectrum plasmonic semiconductors for photocatalysis. Mater. Horiz. 11, 5470–5498 (2024).
Guo, J. et al. Boosting hot electrons in hetero-superstructures for plasmon-enhanced catalysis. J. Am. Chem. Soc. 139, 17964–17972 (2017).
Pan, L. et al. Boosting the performance of Cu2O photocathodes for unassisted solar water splitting devices. Nat. Catal. 1, 412–420 (2018).
Hertel, T., Knoesel, E., Wolf, M. & Ertl, G. Ultrafast electron dynamics at Cu(111): response of an electron gas to optical excitation. Phys. Rev. Lett. 76, 535–538 (1996).
Hodak, J. H., Martini, I. & Hartland, G. V. Spectroscopy and dynamics of nanometer-sized noble metal particles. J. Phys. Chem. B. 102, 6958–6967 (1998).
Chu, K. et al. Unveiling the synergy of O-vacancy and heterostructure over MoO3-x/MXene for N2 electroreduction to NH3. Adv. Energy Mater. 12, 2103022 (2021).
Zhu, Q. et al. Hydrogen-doping-induced metal-like ultrahigh free-carrier concentration in metal-oxide material for giant and tunable plasmon resonance. Adv. Mater. 32, 2004059 (2020).
Yu, Y. et al. Surface engineering for extremely enhanced charge separation and photocatalytic hydrogen evolution on g-C3N4. Adv. Mater. 30, 1705060 (2018).
Dou, M. et al. Qualitative and quantitative analysis of electrons donated by pollutants in electron transfer-based oxidation system: Electrochemical measurement and theoretical calculations. J. Hazard. Mater. 473, 134720–134732 (2024).
Qu, Z. et al. Designing C-Fe-O bonded MIL-88B(Fe)/jasmine petal-derived-carbon composite biosensor for the simultaneous detection of dopamine and uric acid. Chem. Eng. J. 404, 126570–126630 (2021).
Shiroudi, A. et al. Computational analysis of substituent effects on proton affinity and gas-phase basicity of TEMPO derivatives and their hydrogen bonding interactions with water molecules. Sci. Rep. 14, 1–20 (2024).
Dai, T. Y. et al. Recent progress on computation-guided catalyst design for highly efficient nitrogen reduction reaction. Adv. Funct. Mater. 34, 2400773–2400793 (2024).
Zhang, S. et al. Photocatalytic H2 evolution from ammonia borane: improvement of charge separation and directional charge transmission. ChemSusChem 13, 5264–5272 (2020).
Li, Z. et al. Covalent triazine framework supported non-noble metal nanoparticles with superior activity for catalytic hydrolysis of ammonia borane: from mechanistic study to catalyst design. Chem. Sci. 8, 781–788 (2017).
elville, H. W. M. et al. The sorption of hydrogen and deuterium by copper and palladium I-the behaviour of copper and copper oxides. Proc. R. Soc. A. 151, 77–88 (1935).
Zhang, C. et al. Two-dimensional molybdenum boride coordinating with ruthenium nanoparticles to boost hydrogen generation from hydrolytic dehydrogenation of ammonia borane. J. Colloid Interface Sci. 669, 794–803 (2024).
Zheng, J.-H. et al. General strategy for evaluating the d-band center shift and ethanol oxidation reaction pathway towards Pt-based electrocatalysts. Sci. China Chem. 66, 279–288 (2022).
He, Y. et al. Double-plasmonic-coupled heterojunction photocatalysts for highly-efficient full-spectrum-light-driven H2 evolution from ammonia borane. Chem. Eng. J. 481, 148299–148311 (2024).
Xie, Z. et al. Rationally-designed core-shell structure with double-plasmon effect for efficient and tunable upconversion luminescence emission. Appl. Surf. Sci. 643, 158726–158736 (2024).
Sun, G. et al. Structural design and electronic regulation of atomically dispersed Mo-S4 sites by second shell N species reconfiguration for intensifying hydrogen spillover. Chem. Eng. J. 500, 156679–156687 (2024).
Su, D. et al. Efficient amine-assisted CO2 hydrogenation to methanol co-catalyzed by metallic and oxidized sites within ruthenium clusters. Nat. Commun. 16, 590–600 (2025).
Rohrbach, A. & Hafner, J. Molecular adsorption of NO on NiO(100). Phys. Rev. B 71, 1–7 (2005).
Acknowledgements
The authors acknowledge the financial support from the National Natural Science Foundation of China (NSFC, Grant Nos. 62205165, 52102362, 51672143, 51808303).
Author information
Authors and Affiliations
Contributions
Y.Y., D.Y., and W.Y. conceived the idea and directed the project. P. Li performed the synthesis of the samples, material characterizations, experimental measurement and data analysis. N. Tu conducted the theoretical calculations and data analysis. J. Li gave assistance with data collection. H. Hou analyzed and discussed the results. Y.Y., D.Y., and W.Y. carried out the data analyses and co-wrote the manuscript. All authors discussed the data and contributed to the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yu-Jia Zeng and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, P., Tu, N., Yang, Y. et al. Multi-field coupling enhanced plasmonic Moδ+ active site to efficiently hydrolyze ammonia borane. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71055-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71055-1