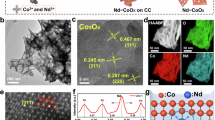
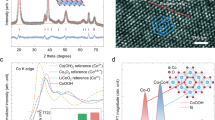
Abstract
Developing noble-metal-free electrocatalyst for oxygen evolution reaction in a proton exchange membrane water electrolyzer is a key to sustainable and economical hydrogen production. Herein, we rationally design and develop a chromium and neodymium co-doped cobalt oxide (CrNd-Co3O4) electrocatalyst that exhibits high activity and durability in the acidic oxygen evolution reaction condition. Furthermore, an in-situ acid circulation strategy is proposed to tackle the ubiquitous issue of membrane poisoning by leached cations in proton exchange membrane water electrolyzers. Consequently, the proton exchange membrane water electrolyzer with CrNd-Co3O4 anode achieves a stable operating current density of 2 amperes per square centimeter at 2.27 volts and 4 amperes per square centimeter at 2.54 volts for 1,000 hours.
Similar content being viewed by others
Data availability
Source data are provided with this paper.
References
Wang, Q. et al. Long-term stability challenges and opportunities in acidic oxygen evolution electrocatalysis. Angew. Chem. Int. Ed. 135, e202216645 (2022).
Tao, H. B. et al. The gap between academic research on proton exchange membrane water electrolysers and industrial demands. Nat. Nanotechnol. 19, 1074–1076 (2024).
An, L. et al. Recent development of oxygen evolution electrocatalysts in acidic environment. Adv. Mater. 33, 2006328 (2021).
Chen, F.-Y., Wu, Z.-Y., Adler, Z. & Wang, H. Stability challenges of electrocatalytic oxygen evolution reaction: from mechanistic understanding to reactor design. Joule 5, 1704–1731 (2021).
Seitz, L. C. et al. A highly active and stable IrOx/SrIrO3 catalyst for the oxygen evolution reaction. Science 353, 1011–1014 (2016).
Shi, W. et al. Ultrastable supported oxygen evolution electrocatalyst formed by ripening-induced embedding. Science 387, 791–796 (2025).
Liang, C. et al. Unravelling the effects of active site density and energetics on the water oxidation activity of iridium oxides. Nat. Catal. 7, 763–775 (2024).
Wang, N. et al. Doping shortens the metal/metal distance and promotes OH coverage in non-noble acidic oxygen evolution reaction catalysts. J. Am. Chem. Soc. 145, 7829–7836 (2023).
Li, A. et al. Enhancing the stability of cobalt spinel oxide towards sustainable oxygen evolution in acid. Nat. Catal. 5, 109–118 (2022).
Chong, L. et al. La- and Mn-doped cobalt spinel oxygen evolution catalyst for proton exchange membrane electrolysis. Science 380, 609–616 (2023).
Ram, R. et al. Water-hydroxide trapping in cobalt tungstate for proton exchange membrane water electrolysis. Science 384, 1373–1380 (2024).
Rong, C., Dastafkan, K., Wang, Y. & Zhao, C. Breaking the activity and stability bottlenecks of electrocatalysts for oxygen evolution reactions in acids. Adv. Mater. 35, 2211884 (2023).
Wu, Z.-Y. et al. Non-iridium-based electrocatalyst for durable acidic oxygen evolution reaction in proton exchange membrane water electrolysis. Nat. Mater. 22, 100–108 (2022).
Priamushko, T. et al. Be aware of transient dissolution processes in Co3O4 acidic oxygen evolution reaction electrocatalysts. J. Am. Chem. Soc. 147, 3517–3528 (2025).
Yoshimura, R., Wai, S., Ota, Y., Nishioka, K. & Suzuki, Y. Effects of artificial river water on PEM water electrolysis performance. Catalysts 12, 934 (2022).
Mo, J. K. et al. Electrochemical investigation of stainless steel corrosion in a proton exchange membrane electrolyzer cell. Int. J. Hydrog. Energy 40, 12506–12511 (2015).
Liu, Z. et al. Promotion of overall water splitting activity over a wide pH range by interfacial electrical effects of metallic NiCo-nitrides nanoparticle/NiCo2O4 nanoflake/graphite fibers. Adv. Sci. 6, 1801829 (2019).
Kong, S. et al. Acid-stable manganese oxides for proton exchange membrane water electrolysis. Nat. Catal. 7, 252–261 (2024).
Zhang, J. et al. Advances in thermodynamic-kinetic model for analyzing the oxygen evolution reaction. ACS Catal. 10, 8597–8610 (2020).
Tao, H. B. et al. Revealing energetics of surface oxygen redox from kinetic fingerprint in oxygen electrocatalysis. J. Am. Chem. Soc. 141, 13803–13811 (2019).
Tao, H. B. et al. A general method to probe oxygen evolution intermediates at operating conditions. Joule 3, 1498–1509 (2019).
Hao, S. et al. Dopants fixation of ruthenium for boosting acidic oxygen evolution stability and activity. Nat. Commun. 11, 5368 (2020).
Rong, X., Parolin, J. & Kolpak, A. M. A fundamental relationship between reaction mechanism and stability in metal oxide catalysts for oxygen evolution. ACS Catal. 6, 1153–1158 (2016).
Zeng, F., Mebrahtu, C., Liao, L., Beine, A. K. & Palkovits, R. Stability and deactivation of OER electrocatalysts: a review. J. Energy Chem. 69, 301–329 (2022).
Hameed, A. et al. A detailed investigation of rare earth lanthanum substitution effects on the structural, morphological, vibrational, optical, dielectric and magnetic properties of Co-Zn spinel ferrites. Front. Chem. 12, 1433004 (2024).
Matar, S. M. et al. Chemical synthesis of NdxCo1−xFe2O4 hybrid nanoparticles for permanent magnet applications: structural, magnetic and electrical properties. Nanoscale Adv. 7, 2725–2741 (2025).
Zhou, Y. et al. Enlarged Co-O covalency in octahedral sites leading to highly efficient spinel oxides for oxygen evolution reaction. Adv. Mater. 30, 1802912 (2018).
Zhao, Y. et al. Oxygen evolution/reduction reaction catalysts: from in situ monitoring and reaction mechanisms to rational design. Chem. Rev. 123, 6257–6358 (2023).
Tan, A. et al. Poisoning effects of Na-ions on membrane electrode assemblies in proton exchange membrane water electrolysis and strategies for recovery. Chem. Eng. Sci. 293, 120083 (2024).
Padgett, E. et al. Performance losses and current-driven recovery from cation contaminants in PEM water electrolysis. J. Electrochem. Soc. 171, 064510 (2024).
Wang, X. et al. The influence of Ferric ion contamination on the solid polymer electrolyte water electrolysis performance. Electrochim. Acta 158, 253–257 (2015).
Tan, A. et al. The poisoning effects of Ti-ion from porous transport layers on the membrane electrode assembly of proton exchange membrane water electrolyzers. Chem. Eng. J. 471, 144624 (2023).
Sin, P. S., Wai, S., Ota, Y., Nishioka, K. & Suzuki, Y. Performance recovery of proton exchange membrane electrolyzer degraded by metal cations contamination. Int. J. Hydrog. Energy 53, 86–92 (2024).
Lu, Y., Zhao, C.-Z., Huang, J.-Q. & Zhang, Q. The timescale identification decoupling complicated kinetic processes in lithium batteries. Joule 6, 1172–1198 (2022).
Li, Y. et al. Application of distribution of relaxation times method in polymer electrolyte membrane water electrolyzer. Chem. Eng. J. 451, 138327 (2023).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Hammer, B., Hansen, L. B. & Nørskov, J. K. Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals. Phys. Rev. B 59, 7413 (1999).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Singh, A. et al. Successive spin reorientations and rare earth ordering in Nd0.5Dy0.5FeO3: Experimental and ab initio investigations. Phys. Rev. B 102, 144432 (2020).
Peng, Y., Hajiyani, H. & Pentcheva, R. Influence of Fe and Ni doping on the OER performance at the Co3O4 (001) surface: insights from DFT+ U calculations. ACS Catal. 11, 5601–5613 (2021).
Lawes, G. et al. Dielectric anomalies and spiral magnetic order in CoCr2O4. Phys. Rev. B—Condens. Matter Mater. Phys. 74, 024413 (2006).
Liu, Y. et al. Surface oxygen vacancies on reduced Co3O4 (100): superoxide formation and ultra-low-temperature CO oxidation. Angew. Chem. Int. Ed. 60, 16514–16520 (2021).
Montoya, A. & Haynes, B. S. Periodic density functional study of Co3O4 surfaces. Chem. Phys. Lett. 502, 63–68 (2011).
Huang, J. et al. Surface oxidation/spin state determines oxygen evolution reaction activity of cobalt-based catalysts in acidic environment. Nat. Commun. 15, 3067 (2024).
Nørskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Gao, G., O’Mullane, A. P. & Du, A. 2D MXenes: a new family of promising catalysts for the hydrogen evolution reaction. Acs Catal. 7, 494–500 (2017).
McCrory, C. C. L., Jung, S., Peters, J. C. & Jaramillo, T. F. Benchmarking heterogeneous electrocatalysts for the oxygen evolution reaction. J. Am. Chem. Soc. 135, 16977–16987 (2013).
McCrory, C. C. L. et al. Benchmarking hydrogen evolving reaction and oxygen evolving reaction electrocatalysts for solar water splitting devices. J. Am. Chem. Soc. 137, 4347–4357 (2015).
Acknowledgements
This work was financially supported by a grant (CHI-P2022-04) from Centre for Hydrogen Innovations at National University of Singapore. B.L. acknowledges support from the City University of Hong Kong startup fund (9020003), ITF–RTH - Global STEM Professorship (9446006), and JC STEM Lab of Advanced CO2 Upcycling (9228005). H.T. thanks National Key R&D Program of China (2023YFB4004600), Science and Technology Projects (RD2021010401) of the Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), the Science and Technology Project (2022L3077) of Fujian Province, and the New Cornerstone Science Foundation. F.L. thanks the Fundamental Research Funds for the Central Universities, Southwest Jiaotong University (2682025CX062). S.-F.H. acknowledges financial support from the National Science and Technology Council, Taiwan (NSTC 114-2628-M-A49-005) and support from the Yushan Young Scholar Program and the Center for Emergent Functional Matter Science (Ministry of Education, Taiwan).
Author information
Authors and Affiliations
Contributions
B.L., P.C., H.T. and T.G. conceived and designed the project. T.G. performed the catalyst synthesis, most of the structural characterizations, and electrochemical tests. F.L. carried out the theoretical calculations. S.-F.H. and K.-S.P performed the XAS measurements. T.G. and H.T. conducted MEA preparation and PEMWE measurements. H.Y., Y.S., Q.S. and Q.M. assisted with data analysis and paper revision. T.G., P.C. and B.L. wrote the paper. All the authors reviewed and contributed to this paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Valentín Briega-Martos for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gao, T., Li, F., Hung, SF. et al. Cr and Nd co-doped cobalt oxide for stable proton exchange membrane water electrolysis. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71066-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71066-y