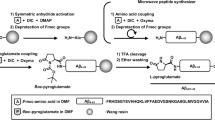
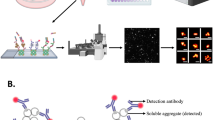
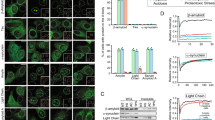
Abstract
In early drug discovery, in vitro screening is frequently used, but selected candidates often fail in vivo. Induced pluripotent stem cell (iPSC)–based disease models offer improved physiological relevance; however, the high costs of media and differentiation procedures limit large-scale testing. Here, we develop a high-throughput conditioned-media-based screening system—the High-throughput screening technology for Aggregation Inhibitors of Diseased cell-derived Aggregative Proteins (HaiDap) system—to identify inhibitors of aggregation induced by iPSC-secreted amyloid β (Aβ). Using conditioned media derived from differentiated iPSCs of a male Alzheimer’s disease patient, we screen extracts from 22 edible plants. Whereas PBS-based assays showed 40.9% (9/22) apparent selectivity, the HaiDap system demonstrates higher specificity (13.6%; 3/22). All three identified extracts (O. aristatus, S. aromaticum, and G. yesoense) significantly delay Aβ aggregation on neuronal surfaces in an iPSC-based assay. These findings suggest that the HaiDap system enables efficient, accurate, and low-cost screening of amyloid aggregation inhibitors.
Similar content being viewed by others
Data availability
All data supporting the findings of this study are provided in the manuscript and Supplementary Information file. Source data are provided with this paper as a Source Data file.
Code availability
Data analysis for Fig. 4b, Supplementary Fig. 6b and Supplementary Fig. 7b was performed as follows: The software application to perform logistic regression and visualization was written in Python. Nonlinear regression was implemented using the least square fitting implementation provided by the Scipy library. The plots were created using matplotlib. The web app, as well as its source code, is available at https://zenodo.org/account/settings/github/repository/StefanBaar/LGR_drug_screening. All libraries used for producing the figure are open-source and are referenced at the provided URL.
References
Van Drie, J. H. Hit diffusion: limitations to drug discovery and structure-based design. J. Comput. Aided Mol. Des. 36, 373–379 (2022).
Yamanaka, S. Induced pluripotent stem cells: past, present, and future. Cell Stem Cell 10, 678–684 (2012).
Inoue, H., Nagata, N., Kurokawa, H. & Yamanaka, S. iPS cells: a game changer for future medicine. EMBO J. 33, 409–417 (2014).
Wu, X., Chen, S. & Lu, C. Amyloid precursor protein promotes the migration and invasion of breast cancer cells by regulating the MAPK signaling pathway. Int. J. Mol. Med. 45, 162–174 (2019).
Israel, M. A. et al. Probing sporadic and familial Alzheimer’s disease using induced pluripotent stem cells. Nature 482, 216–220 (2012).
Kondo, T. et al. Modeling Alzheimer’s disease with iPSCs reveals stress phenotypes associated with intracellular Aβ and differential drug responsiveness. Cell Stem Cell 12, 487–496 (2013).
Pepys, M. B. Amyloidosis. Annu. Rev. Med. 57, 223–241 (2006).
Ikejima, C. et al. Multicenter population-based study on the prevalence of early onset dementia in Japan: Vascular dementia as its prominent cause. Psychiatry Clin. Neurosci. 68, 216–224 (2014).
Plassman, B. L. et al. Prevalence of dementia in the United States: the aging, demographics, and memory study. Neuroepidemiology 29, 125–132 (2007).
Ikejima, C. et al. Multicentre population-based dementia prevalence survey in Japan: a preliminary report. Psychogeriatrics 12, 120–123 (2012).
Hardy, J. & Selkoe, D. J. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297, 353–356 (2002).
Blennow, K., de Leon, M. J. & Zetterberg, H. Alzheimer’s disease. Lancet 368, 387–403 (2006).
Jack, C. R. et al. Hypothetical model of dynamic biomarkers of the Alzheimer’s pathological cascade. Lancet Neurol. 9, 119–128 (2010).
Jack, C. R. et al. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 12, 207–216 (2013).
Tokuraku, K., Marquardt, M. & Ikezu, T. Real-time imaging and quantification of amyloid-β peptide aggregates by novel quantum-dot nanoprobes. PLoS ONE 4, e8492 (2009).
Lin, X. et al. Real-time 3D imaging and inhibition analysis of various amyloid aggregations using quantum dots. Int. J. Mol. Sci. 21, 1978 (2020).
Lin, X. et al. Dietary intake of rosmarinic acid increases serum inhibitory activity in amyloid A aggregation and suppresses deposition in the organs of mice. Int. J. Mol. Sci. 21, 6031 (2020).
Ishigaki, Y. et al. A microliter-scale high-throughput screening system with quantum-dot nanoprobes for amyloid-β aggregation inhibitors. PLoS One 8, e72992 (2013).
Cai, W., Li, L., Sang, S., Pan, X. & Zhong, C. Physiological roles of β-amyloid in regulating synaptic function: implications for AD pathophysiology. Neurosci. Bull. 39, 1289–1308 (2022).
Brewer, G. J., Torricelli, J. R., Evege, E. K. & Price, P. J. Optimized survival of hippocampal neurons in B27-supplemented neurobasal? A new serum-free medium combination. J. Neurosci. Res. 35, 567–576 (1993).
Kuo, Y.-M. et al. Amyloid-β peptides interact with plasma proteins and erythrocytes: implications for their quantitation in plasma. Biochem. Biophys. Res. Commun. 268, 750–756 (2000).
Biere, A. L. et al. Amyloid β-peptide is transported on lipoproteins and albumin in human plasma. J. Biol. Chem. 271, 32916–32922 (1996).
Bode, D. C. et al. Serum albumin’s protective inhibition of amyloid-β fiber formation is suppressed by cholesterol, fatty acids and warfarin. J. Mol. Biol. 430, 919–934 (2018).
Nishishita, N., Kobayashi, A., Tokuraku, K., Uwai, K. & Kuragano, M. Method for measuring aggregating proteins, method for visualizing aggregation, and kit used therefor. US Patent App. 18/285,090 (2024).
Sasaki, R. et al. An automated microliter-scale high-throughput screening system (MSHTS) for real-time monitoring of protein aggregation using quantum-dot nanoprobes. Sci. Rep. 9, 2587 (2019).
Zhu, H.-L. et al. Quantitative characterization of heparin binding to tau protein. J. Biol. Chem. 285, 3592–3599 (2010).
Bassil, F. et al. Amyloid-beta (Aβ) plaques promote seeding and spreading of alpha-synuclein and tau in a mouse model of Lewy body disorders with Aβ pathology. Neuron 105, 260–275.e6 (2020).
Ono, K., Hasegawa, K., Naiki, H. & Yamada, M. Curcumin has potent anti-amyloidogenic effects for Alzheimer’s β-amyloid fibrils in vitro. J. Neurosci. Res. 75, 742–750 (2004).
Hamaguchi, T., Ono, K., Murase, A. & Yamada, M. Phenolic compounds prevent Alzheimer’s pathology through different effects on the amyloid-β aggregation pathway. Am. J. Pathol. 175, 2557–2565 (2009).
Hase, T. et al. Rosmarinic acid suppresses Alzheimer’s disease development by reducing amyloid β aggregation by increasing monoamine secretion. Sci. Rep. 9, 8711 (2019).
Kuragano, M., Yamashita, R., Chikai, Y., Kitamura, R. & Tokuraku, K. Three-dimensional real time imaging of amyloid β aggregation on living cells. Sci. Rep. 10, 9742 (2020).
Levites, Y., Amit, T., Mandel, S. & Youdim, M. B. H. Neuroprotection and neurorescue against Aβ toxicity and PKC-dependent release of non-amyloidogenic soluble precursor protein by green tea polyphenol (-)-epigallocatechin-3-gallate. FASEB J. 17, 1–23 (2003).
Shibata, S. et al. Green tea consumption and cerebral white matter lesions in community-dwelling older adults without dementia. npj Sci. Food 9, 2 (2025).
Picón-Pagès, P. et al. Human albumin impairs amyloid β-peptide fibrillation through its C-terminus: from docking modeling to protection against neurotoxicity in Alzheimer’s disease. Comput. Struct. Biotechnol. J. 17, 963–971 (2019).
Lu, N., Yang, Q., Li, J., Tian, R. & Peng, Y. Y. Inhibitory effect of human serum albumin on Cu-induced Aβ40 aggregation and toxicity. Eur. J. Pharmacol. 767, 160–164 (2015).
Milojevic, Raditsis, J. & Melacini, A. G. Human serum albumin inhibits Aβ fibrillization through a ‘monomer-competitor’ mechanism. Biophys. J. 97, 2585–2594 (2009).
Kim, J. W. et al. Serum albumin and beta-amyloid deposition in the human brain. Neurology 95, e815–e826 (2020).
Magy, N., Liepnieks, J. J., Benson, M. D. & Kluve-Beckerman, B. Amyloid-enhancing factor mediates amyloid formation on fibroblasts via a nidus/template mechanism. Arthritis Rheum. 48, 1430–1437 (2003).
Chuang, E., Hori, A. M., Hesketh, C. D. & Shorter, J. Amyloid assembly and disassembly. J. Cell Sci. 131, jcs189928 (2018).
Wechalekar, A. D., Gillmore, J. D. & Hawkins, P. N. Systemic amyloidosis. Lancet 387, 2641–2654 (2016).
Forny-Germano, L. et al. Alzheimer’s disease-like pathology induced by amyloid-β oligomers in nonhuman primates. J. Neurosci. 34, 13629–13643 (2014).
Yue, F. et al. Synthetic amyloid-β oligomers drive early pathological progression of Alzheimer’s disease in nonhuman primates. iScience 24, 103207 (2021).
Lambert, M. P. et al. Diffusible, nonfibrillar ligands derived from Aβ1-42 are potent central nervous system neurotoxins. Proc. Natl. Acad. Sci. USA. 95, 6448–6453 (1998).
Okuzumi, A. et al. Propagative α-synuclein seeds as serum biomarkers for synucleinopathies. Nat. Med. 29, 1448–1455 (2023).
Chau, E. & Kim, J. R. α-Synuclein-assisted oligomerization of β-amyloid (1–42). Arch. Biochem. Biophys. 717, 109120 (2022).
Lowry, O., Rosebrough, N., Farr, A. L. & Randall, R. Protein measurement with the folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951).
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970).
Sidhu, A., Segers-Nolten, I. & Subramaniam, V. Solution conditions define morphological homogeneity of α-synuclein fibrils. Biochim. Biophys. Acta Proteins Proteom. 1844, 2127–2134 (2014).
TEI, M. et al. Variation of amino acid sequences of serum amyloid a (SAA) and immunohistochemical analysis of amyloid a (AA) in Japanese domestic cats. J. Vet. Med. Sci. 80, 164–172 (2018).
Ludwig, T. E. et al. Feeder-independent culture of human embryonic stem cells. Nat. Methods 3, 637–646 (2006).
Briscoe, J. & Ericson, J. Specification of neuronal fates in the ventral neural tube. Curr. Opin. Neurobiol. 11, 43–49 (2001).
Imaizumi, K. et al. Controlling the regional identity of hPSC-derived neurons to uncover neuronal subtype specificity of neurological disease phenotypes. Stem Cell Rep. 5, 1010–1022 (2015).
Matsumoto, T. et al. Functional neurons generated from T cell-derived induced pluripotent stem cells for neurological disease modeling. Stem Cell Rep. 6, 422–435 (2016).
Bardy, C. et al. Neuronal medium that supports basic synaptic functions and activity of human neurons in vitro. Proc. Natl. Acad. Sci. USA. 112, E2725–E2734 (2015).
Acknowledgements
This work was supported by JSPS KAKENHI Grant Number JP24K08627 (K.T.) and JST Grant Number JPMJPF2213 (to K.T.). TEM observations were conducted at the Institute for Molecular Science, supported by “Advanced Research Infrastructure for Materials and Nanotechnology in Japan (ARIM)” of the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Proposal Number JPMXP1222CT0078.
Author information
Authors and Affiliations
Contributions
M.K. performed major experiments and wrote the manuscript. N.N. and A.K. performed iPSC experiments. K.A., S.B., and S.W. performed kinetic analyses. K.U. prepared plant extracts. TQPN prepared α-synuclein. K.W. prepared SAA. N.N. and K.T. designed the project. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
N.N. and A.K. are now employees of Kaneka Corporation. This research was partially funded by Kaneka Corporation. A patent related to the HaiDap system described in this manuscript has been filed. Patent applicant: Kaneka Corporation and Muroran Institute of Technology. Inventor(s): N.N., A.K., K.T., K.U., M.K. Application number: US.202218285090.A and JP2023511699A, Status: published. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kuragano, M., Nishishita, N., Araya, K. et al. A high-throughput conditioned-media-based screening system identifies inhibitors of aggregation induced by iPSC-secreted amyloid β. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71078-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71078-8