Abstract
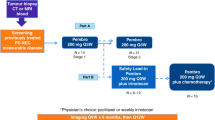
Immune checkpoint inhibitors (ICI) synergize preclinically with antibody drug conjugates (ADC), harboring anti-tubulin maytansinoid payloads. We conducted an investigator-initiated, single-arm, phase 2 trial of mirvetuximab soravtansine (MIRV), a folate receptor alpha (FOLR1/FRα)-targeting ADC with the maytansinoid payload, DM4, combined with pembrolizumab in female patients with recurrent FOLR1-expressing serous endometrial cancer (EC, NCT03835819). Co-primary objectives include objective response rate (ORR) and rate of progression-free survival at 6 months (PFS6); secondary objectives include PFS, overall survival, duration of response and safety. Exploratory objectives include correlation of tumor genomics and immunoprofiling with clinical activity. Eighteen patients initiated protocol therapy [MIRV 6 mg/kg adjusted ideal body weight IV and pembrolizumab 200 mg IV every 3 weeks]. Confirmed ORR is 28% (1 complete and 4 partial responses, 95% CI:10-53%), Kaplan Meier estimate of PFS6 is 24.4% (95% CI:7.7-46.1%) with 4 patients progression free at 6 months; trial was closed early for feasibility (planned sample size of 35 patients not reached) and hence these results are considered preliminary. G3 treatment-related adverse effects were rare with no grade ≥4 toxicities. We report a population of high FOLR1-expressing tumor-associated macrophages (CD163 + FOLR1 + ), suggesting potential on-target, off-tumor immune editing by MIRV. A composite biomarker score derived in this cohort correlates with objective response to MIRV and pembrolizumab.
Similar content being viewed by others
Data availability
The study protocol and the statistical analysis plan are available in the Supplementary Information file. Additional individual de-identified participant data can be shared upon request. The remaining data are available within the Article or Source Data file. Source data are provided with this paper.
References
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA: A Cancer J. Clin. 74, 12–49 (2024).
Siegel, R. L., Miller, K. D., Wagle, N. S. & Jemal, A. Cancer statistics, 2023. CA: A Cancer J. Clin. 73, 17–48 (2023).
O’Malley, D. M. et al. Pembrolizumab in patients with microsatellite instability-high advanced endometrial cancer: results from the KEYNOTE-158 study. J. Clin. Oncol. 40, 752–761 (2022).
Oaknin, A. et al. Dostarlimab in advanced/recurrent (AR) mismatch repair deficient/microsatellite instability–high or proficient/stable (dMMR/MSI-H or MMRp/MSS) endometrial cancer (EC): the GARNET study. J. Clin. Oncol. 40, 5509–5509 (2022).
Antill, Y. et al. Clinical activity of durvalumab for patients with advanced mismatch repair-deficient and repair-proficient endometrial cancer. A nonrandomized phase 2 clinical trial. J. Immunother. Cancer 9, e002255 (2021).
Konstantinopoulos, P. A. et al. Phase II study of avelumab in patients with mismatch repair deficient and mismatch repair proficient recurrent/persistent endometrial cancer. J. Clin. Oncol. 37, 2786–2794 (2019).
Eskander, R. N. et al. Pembrolizumab plus chemotherapy in advanced endometrial cancer. N. Engl. J. Med. 388, 2159–2170 (2023).
Mirza, M. R. et al. Dostarlimab for primary advanced or recurrent endometrial cancer. N. Engl. J. Med. 388, 2145–2158 (2023).
Colombo, N. et al. Atezolizumab and chemotherapy for advanced or recurrent endometrial cancer (AtTEnd): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 25, 1135–1146 (2024).
Westin, S. N. et al. Durvalumab plus carboplatin/paclitaxel followed by maintenance durvalumab with or without olaparib as first-line treatment for advanced endometrial cancer: the phase III DUO-E trial. J. Clin. Oncol. 42, 283–299 (2024).
Colombo, N. et al. LBA40 phase III double-blind randomized placebo controlled trial of atezolizumab in combination with carboplatin and paclitaxel in women with advanced/recurrent endometrial carcinoma. Ann. Oncol. 34, S1281–S1282 (2023).
Pignata, S. et al. Carboplatin and paclitaxel plus avelumab compared with carboplatin and paclitaxel in advanced or recurrent endometrial cancer (MITO END-3): a multicentre, open-label, randomised, controlled, phase 2 trial. Lancet Oncol. 24, 286–296 (2023).
Ott, P. A. et al. Safety and antitumor activity of pembrolizumab in advanced programmed death ligand 1-positive endometrial cancer: results from the KEYNOTE-028 study. J. Clin. Oncol. 35, 2535–2541 (2017).
Fucà, G., Sabatucci, I., Paderno, M. & Lorusso, D. The clinical landscape of antibody-drug conjugates in endometrial cancer. Int J. Gynecol. Cancer 34, 1795–1804 (2024).
Ab, O. et al. IMGN853, a folate receptor-α (FRα)–targeting antibody–drug conjugate, exhibits potent targeted antitumor activity against FRα-expressing tumors. Mol. Cancer Ther. 14, 1605–1613 (2015).
U.S. Food & Drug Administration. FDA Approval of Elahere (Mirvetuximab Soravtansine-Gynx) for FRα Positive, Platinum-Resistant Epithelial Ovarian, Fallopian Tube, or Peritoneal Cancer (FDA, 2022). Accessed 17 February 2023.
Matulonis, U. A. et al. Efficacy and safety of mirvetuximab soravtansine in patients with platinum-resistant ovarian cancer with high folate receptor alpha expression: results from the SORAYA study. J. Clin. Oncol. 41, 2436–2445 (2023).
Moore, K. N. et al. Mirvetuximab soravtansine in FRα-positive, platinum-resistant ovarian cancer. N. Engl. J. Med. 389, 2162–2174 (2023).
FDA gives nod to mirvetuximab soravtansine. Cancer Discov. 13, 8 (2023).
Dainty, L. A. et al. Overexpression of folate binding protein and mesothelin are associated with uterine serous carcinoma. Gynecol. Oncol. 105, 563–570 (2007).
Senol, S. et al. Folate receptor α expression and significance in endometrioid endometrium carcinoma and endometrial hyperplasia. Int. J. Clin. Exp. Pathol. 8, 5633–5641 (2015).
Martin, L. P. et al. Characterization of folate receptor alpha (FRα) expression in archival tumor and biopsy samples from relapsed epithelial ovarian cancer patients: a phase I expansion study of the FRα-targeting antibody-drug conjugate mirvetuximab soravtansine. Gynecol. Oncol. 147, 402–407 (2017).
Gerber, H. P., Sapra, P., Loganzo, F. & May, C. Combining antibody-drug conjugates and immune-mediated cancer therapy: what to expect?. Biochem. Pharm. 102, 1–6 (2016).
Kepp, O., Zitvogel, L. & Kroemer, G. Clinical evidence that immunogenic cell death sensitizes to PD-1/PD-L1 blockade. Oncoimmunology 10, e1637188 (2019).
Nicolò, E. et al. Combining antibody-drug conjugates with immunotherapy in solid tumors: current landscape and future perspectives. Cancer Treat. Rev. 106, 102395 (2022).
Tsao, L.-C. et al. Effective extracellular payload release and immunomodulatory interactions govern the therapeutic effect of trastuzumab deruxtecan (T-DXd). Nat. Commun. 16, 3167 (2025).
Olson, D. et al. 1187 Enfortumab vedotin induces immunogenic cell death, elicits antitumor immune memory, and shows enhanced preclinical activity in combination with immune checkpoint inhibitors. J. Immunother. Cancer 10, A1231 (2022).
Fuentes-Antrás, J., Genta, S., Vijenthira, A. & Siu, L. L. Antibody–drug conjugates: in search of partners of choice. Trends Cancer 9, 339–354 (2023).
Müller, P. et al. Trastuzumab emtansine (T-DM1) renders HER2+ breast cancer highly susceptible to CTLA-4/PD-1 blockade. Sci. Transl. Med. 7, 315ra188 (2015).
O’Donnell, P. H. et al. Enfortumab vedotin with or without pembrolizumab in cisplatin-ineligible patients with previously untreated locally advanced or metastatic urothelial cancer. J. Clin. Oncol. 41, 4107–4117 (2023).
Powles, T. et al. Enfortumab vedotin and pembrolizumab in untreated advanced urothelial cancer. N. Engl. J. Med. 390, 875–888 (2024).
Hoimes, C. J. et al. Enfortumab vedotin plus pembrolizumab in previously untreated advanced urothelial cancer. J. Clin. Oncol. 41, 22–31 (2023).
Villacampa, G. et al. Safety and efficacy of antibody-drug conjugates plus immunotherapy in solid tumours: a systematic review and meta-analysis. Cancer Treat. Rev. 131, 102847 (2024).
Skaletskaya, A., Ponte, J., Chittenden, T. & Setiady, Y. Treatment of tumor cells with mirvetuximab soravtansine, a FRα-targeting antibody-drug conjugate (ADC), activates monocytes through Fc-FcγR interaction and immunogenic cell death. Society for Immunotherapy in Cancer Annual Meeting, Abstract 316P (2016).
Martin, K. et al. The microtubule-depolymerizing agent ansamitocin P3 programs dendritic cells toward enhanced anti-tumor immunity. Cancer Immunol., Immunother. 63, 925–938 (2014).
Müller, P. et al. Microtubule-depolymerizing agents used in antibody-drug conjugates induce antitumor immunity by stimulation of dendritic cells. Cancer Immunol. Res. 2, 741–755 (2014).
Elahere (mirvetuximab soravtansine) Prescribing Information, AbbVie Inc. NothChicago, IL 60064, USA. https://www.rxabbvie.com/pdf/elahere_pi.pdf (2025).
Weng, C. C., Wu, C. C. & Lin, P. Y. Corneal melting in a case undergoing treatment with pembrolizumab. Clin. Exp. Optom. 103, 379–381 (2020).
Ramaekers, A. et al. Bilateral corneal perforation in a patient under anti-PD1 therapy. Cornea 40, 245–247 (2021).
Schmid, P. et al. 379MO Datopotamab deruxtecan (Dato-DXd) + durvalumab (D) as first-line (1L) treatment for unresectable locally advanced/metastatic triple-negative breast cancer (a/mTNBC): updated results from BEGONIA, a phase Ib/II study. Ann. Oncol. 34, S337 (2023).
Tolaney, S. M. et al. Sacituzumab govitecan (SG) + pembrolizumab (pembro) vs chemotherapy (chemo) + pembro in previously untreated PD-L1–positive advanced triple-negative breast cancer (TNBC): primary results from the randomized phase 3 ASCENT-04/KEYNOTE-D19 study. J. Clin. Oncol. 43, LBA109-LBA109 (2025).
Makker, V. et al. Lenvatinib plus pembrolizumab in previously treated advanced endometrial cancer: updated efficacy and safety from the randomized phase III study 309/KEYNOTE-775. J. Clin. Oncol. 41, 2904–2910 (2023).
Makker, V. et al. Evaluation of potential biomarkers for lenvatinib plus pembrolizumab among patients with advanced endometrial cancer: results from Study 111/KEYNOTE-146. J. Immunother. Cancer. https://doi.org/10.1136/jitc-2023-007929 (2024).
Lin, D. I. et al. Molecular analysis of endometrial serous carcinoma reveals distinct clinicopathologic and genomic subgroups. Gynecol. Oncol. 164, 558–565 (2022).
Gorski, J. W., Ueland, F. R. & Kolesar, J. M. CCNE1 amplification as a predictive biomarker of chemotherapy resistance in epithelial ovarian cancer. Diagnostics 10, 279 (2020).
Nakayama, N. et al. Gene amplification CCNE1 is related to poor survival and potential therapeutic target in ovarian cancer. Cancer 116, 2621–2634 (2010).
van Wagensveld, L. et al. Homologous recombination deficiency and cyclin E1 amplification are correlated with immune cell infiltration and survival in high-grade serous ovarian cancer. Cancers. https://doi.org/10.3390/cancers14235965 (2022).
Yu, S. et al. Cyclin E1 overexpression triggers interferon signaling and is associated with antitumor immunity in breast cancer. J. Immunother. Cancer. https://doi.org/10.1136/jitc-2024-009239 (2025).
Moy, R. H. et al. Molecular landscape and clinical implication of CCNE1-amplified esophagogastric cancer. J. Clin. Oncol. 41, 440–440 (2023).
Puig-Kröger, A. et al. Folate receptor beta is expressed by tumor-associated macrophages and constitutes a marker for M2 anti-inflammatory/regulatory macrophages. Cancer Res. 69, 9395–9403 (2009).
Nalio Ramos, R. et al. Tissue-resident FOLR2(+) macrophages associate with CD8(+) T cell infiltration in human breast cancer. Cell 185, 1189–1207.e25 (2022).
Ledermann, J. A., Canevari, S. & Thigpen, T. Targeting the folate receptor: diagnostic and therapeutic approaches to personalize cancer treatments. Ann. Oncol. 26, 2034–2043 (2015).
Kalli, K. R. et al. Folate receptor alpha as a tumor target in epithelial ovarian cancer. Gynecol. Oncol. 108, 619–626 (2008).
Markert, S. et al. Alpha-folate receptor expression in epithelial ovarian carcinoma and non-neoplastic ovarian tissue. Anticancer Res. 28, 3567–3572 (2008).
O’Shannessy, D. J., Somers, E. B., Smale, R. & Fu, Y. S. Expression of folate receptor-α (FRA) in gynecologic malignancies and its relationship to the tumor type. Int. J. Gynecol. Pathol. 32, 258–268 (2013).
Bax, H. J. et al. Folate receptor alpha in ovarian cancer tissue and patient serum is associated with disease burden and treatment outcomes. Br. J. Cancer 128, 342–353 (2023).
Risinger, J. I. et al. Microarray analysis reveals distinct gene expression profiles among different histologic types of endometrial cancer. Cancer Res. 63, 6–11 (2003).
Wu, M., Gunning, W. & Ratnam, M. Expression of folate receptor type alpha in relation to cell type, malignancy, and differentiation in ovary, uterus, and cervix. Cancer Epidemiol. Biomark. Prev. 8, 775–782 (1999).
Boerman, O. C. et al. Comparative immunohistochemical study of four monoclonal antibodies directed against ovarian carcinoma-associated antigens. Int. J. Gynecol. Pathol. 10, 15–25 (1991).
Garin-Chesa, P., Campbell, I., Saigo, P. E., Lewis, J. L. Jr, Old, L. J. & Rettig, W. J. Trophoblast and ovarian cancer antigen LK26. Sensitivity and specificity in immunopathology and molecular identification as a folate-binding protein. Am. J. Pathol. 142, 557–567 (1993).
Kim, D. K. et al. Folate binding protein peptide 191-199 presented on dendritic cells can stimulate CTL from ovarian and breast cancer patients. Anticancer Res. 19, 2907–2916 (1999).
Xiao, Y. et al. FRα expression in advanced endometrial carcinoma: clinicopathological, molecular, and prognosis correlates. Gynecol. Oncol. 197, 146–154 (2025).
Bogani, G. et al. Uterine serous carcinoma. Gynecol. Oncol. 162, 226–234 (2021).
Matulonis, U. A. et al. Safety and efficacy of mirvetuximab soravtansine, a folate receptor alpha (FRα)-targeting antibody-drug conjugate (ADC), in combination with pembrolizumab in patients with platinum-resistant ovarian cancer. Gynecol Oncol. 200, 96–104 (2025).
Makker, V. et al. Lenvatinib plus pembrolizumab for advanced endometrial cancer. N. Engl. J. Med. 386, 437–448 (2022).
Lheureux, S. et al. Translational randomized phase II trial of cabozantinib in combination with nivolumab in advanced, recurrent, or metastatic endometrial cancer. J. Immunother. Cancer. https://doi.org/10.1136/jitc-2021-004233 (2022).
Lee, E. K. et al. Phase 2, two-stage study of avelumab and axitinib in patients with mismatch repair proficient recurrent or persistent endometrial cancer. Gynecol. Oncol. 198, 1–8 (2025).
Matulonis, U. A. et al. Safety and efficacy of mirvetuximab soravtansine, a folate receptor alpha (FRα)-targeting antibody-drug conjugate (ADC), in combination with pembrolizumab in patients with platinum-resistant ovarian cancer. Gynecol. Oncol. 200, 96–104 (2025).
Sholl, L. M. et al. Institutional implementation of clinical tumor profiling on an unselected cancer population. JCI Insight 1, e87062 (2016).
Garcia, E. P. et al. Validation of OncoPanel: a targeted next-generation sequencing assay for the detection of somatic variants in cancer. Arch. Pathol. Lab. Med. 141, 751–758 (2017).
Oliveira, G. et al. Preexisting tumor-resident T cells with cytotoxic potential associate with response to neoadjuvant anti-PD-1 in head and neck cancer. Sci. Immunol. 8, eadf4968 (2023).
Alessi, J. V. et al. Immunoprofiling at an institutional scale reveals that high numbers of intratumoral CD8(+) and PD-1(+) cells predict superior patient survival across major cancer types independent of major risk factors. JCO Precis Oncol. https://doi.org/10.1200/po-25-00240 (2025).
Sill, M. W., Rubinstein, L., Litwin, S. & Yothers, G. A method for utilizing co-primary efficacy outcome measures to screen regimens for activity in two-stage Phase II clinical trials. Clin. Trials 9, 385–395 (2012).
Acknowledgements
We thank all the patients and their families for their participation in this trial. This investigator-initiated study (IND holder P.A.K.) was funded by Merck and AbbVie, which also provided pembrolizumab and mirvetuximab soravtansine, respectively. We would also like to acknowledge support from the Friends of Dana Farber (R.L.P.), the Breast Cancer Research Foundation, and The Lewin Fund to Fight Women’s Cancers.
Author information
Authors and Affiliations
Contributions
R.L.P. and J.V. conceived the study; R.L.P. coordinated, analyzed, and interpreted all the data and wrote the manuscript. P.A.K. supervised and was the IND holder of the study. Y.Z., N.T., and N.X. performed the statistical analysis and were the lead statisticians of the study; R.L.P., E.K.L., C.K., S.C., A.A.W., J.F.L, E.H.S., S.Z., V.J., J.V., U.A.M., and P.A.K. provided clinical data and contributed to the analysis and interpretation of the data; N.E., K.L.P., and S.J.R. performed the immunoprofiling data and analysis; M.H., M.P., and H.S. were Clinical Research Project Managers for the study and coordinated the processing and distribution of the clinical trial samples. All authors contributed to the writing and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
R.L.P. declares consulting and/or advisory board participation for Gilead, unrelated to this work; Payment for educational events: OncLive/MJH Life Sciences. E.K.L. declares advisory board participation: Oncusp Therapeutics and Genmab, unrelated to this work, and institutional research support from: Repare Therapeutics, KSQ Therapeutics, Genmab, GSK, Merck, OnCusp Therapeutics, and NiKang Therapeutics. J.F.L. declares consulting and/or advisory board participation for AbbVie, AstraZeneca, Bristol-Myers Squibb, Clovis Oncology, Daiichi Sankyo, Eisai, Genentech/Roche, Genmab, GlaxoSmithKline, LoxoLilly, Merck, SystImmune, Regeneron Therapeutics, Revolution Medicine, and Zentalis Pharmaceuticals, unrelated to this work. S.J.R. declares research support from Coherus Pharmaceuticals, Delcath Pharmaceuticals, and Bristol Myers Squibb. S.R. is a member of the Scientific Advisory Board of Immunitas Therapeutics. J.V. declares current employment at GSK, unrelated to this work. U.A.M. declares participation in scientific advisory boards: NextCure, Abbvie, Immunogen, Profound Bio, Eisai, the Ovarian Cancer Research Alliance, Tango Therapeutics, Novartis, GSK, Daiichi Sankyo, DayOne Bio, and Whitehawk Therapeutics; UAM also reports participation in a data safety-monitoring board: Mural Oncology, Macrogenics, Daiichi Sankyo, Astrazseneca and Symphogen, all unrelated to this work. P.A.K. declares consulting and/or advisory board participation for AstraZeneca, Bayer, GSK, Merck, Pfizer, BMS, Repare, IMV, Artios, Kadmon, Cardiff, Immunogen, EMD Serono, Scorpion, Schrodinger, Nimbus, Mural Oncology, unrelated to this work. Y.Z., N.E., M.H., M.P., C.K., S.C., A.A.W., E.H.S., H.S., N.X., K.L.P., N.T., S.Z., V.J. declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Casey Cosgrove, Ramez Eskander, and Xianming Tan for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Porter, R.L., Zhou, Y., Eskndir, N. et al. Mirvetuximab soravtansine plus pembrolizumab in recurrent folate receptor alpha-positive uterine serous carcinoma: a phase II trial. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71102-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71102-x