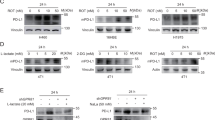
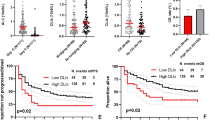
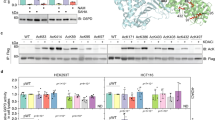
Abstract
p53 is crucial for cellular functions and disease mechanisms, yet effective clinical strategies targeting it remain challenging. Lactylation has emerged as a key factor in understanding disease pathology and offering therapeutic options. Herein, we identify lactylated KAT8 at lysine 145 (K145) as a modulator of p53 activity. GCN5 and SIRT6 function as the acyltransferase and delactylase for KAT8, respectively. K145 lactylation fosters the formation of KAT8-TIP60 complex, which couples with p53 to facilitate its acetylation at lysine 120 (K120). The KAT8-TIP60 complex promotes K120-acetylated p53 binding to the BAX and PUMA promoters, activating their transcription. Furthermore, we link KAT8 lactylation to doxorubicin-induced cardiotoxicity (DIC), showing that doxorubicin increases K145 lactylation, amplifying p53’s pro-apoptotic function and triggering cardiomyocyte apoptosis. Glimepiride, a therapeutic agent for type 2 diabetes, could target KAT8, disrupt its interaction with GCN5, inhibit KAT8 K145 lactylation, and mitigate DIC. These findings provide insight into how KAT8 K145 lactylation modulates p53 activity and contributes to DIC.
Similar content being viewed by others
Data availability
The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium (https://proteomecentral.proteomexchange.org) via the iProX partner repository67,68 with the dataset identifier PXD075302. The AlphaFold models corresponding to Figs. 2I, 4B, and 7E have been deposited in ModelArchive (www.modelarchive.org) and are available under the accession codes ma-amz8q, ma-9k082, and ma-tp0qr, respectively. Other data generated in this study are provided in the Article, its Supplementary Information and Source Data file. Source data are provided with this paper.
References
Liu, J., Qian, C. & Cao, X. Post-translational modification control of innate immunity. Immunity 45, 15–30 (2016).
Gomez-Pastor, R., Burchfiel, E. T. & Thiele, D. J. Regulation of heat shock transcription factors and their roles in physiology and disease. Nat. Rev. Mol. Cell Biol. 19, 4–19 (2018).
Zhang, D. et al. Metabolic regulation of gene expression by histone lactylation. Nature 574, 575–580 (2019).
Li, W. et al. Tumor-derived lactate promotes resistance to bevacizumab treatment by facilitating autophagy enhancer protein RUBCNL expression through histone H3 lysine 18 lactylation (H3K18la) in colorectal cancer. Autophagy 20, 114–130 (2024).
Pan, R.-Y. et al. Positive feedback regulation of microglial glucose metabolism by histone H4 lysine 12 lactylation in Alzheimer’s disease. Cell Metab. 34, 634–648.e6 (2022).
Zhou, C. et al. Mutant KRAS-activated circATXN7 fosters tumor immunoescape by sensitizing tumor-specific T cells to activation-induced cell death. Nat. Commun. 15, 499 (2024).
Chen, Y. et al. Metabolic regulation of homologous recombination repair by MRE11 lactylation. Cell 187, 294–311.e21 (2024).
Chen, H. et al. NBS1 lactylation is required for efficient DNA repair and chemotherapy resistance. Nature 631, 663–669 (2024).
Zong, Z. et al. Alanyl-tRNA synthetase, AARS1, is a lactate sensor and lactyltransferase that lactylates p53 and contributes to tumorigenesis. Cell 187, 2375–2392.e33 (2024).
Dang, W. et al. Histone H4 lysine 16 acetylation regulates cellular lifespan. Nature 459, 802–807 (2009).
Füllgrabe, J. et al. The histone H4 lysine 16 acetyltransferase hMOF regulates the outcome of autophagy. Nature 500, 468–471 (2013).
Li, P. et al. Stabilization of MOF (KAT8) by USP10 promotes esophageal squamous cell carcinoma proliferation and metastasis through epigenetic activation of ANXA2/wnt signaling. Oncogene 43, 899–917 (2024).
Gupta, A. et al. MOF phosphorylation by ATM regulates 53BP1-mediated double-strand break repair pathway choice. Cell Rep. 8, 177–189 (2014).
Yuan, H. et al. MYST protein acetyltransferase activity requires active site lysine autoacetylation. EMBO J. 31, 58–70 (2012).
Qiu, B. et al. KAT8 acetylation-controlled lipolysis affects the invasive and migratory potential of colorectal cancer cells. Cell Death Dis. 14, 164 (2023).
Liu, Y., Su, Z., Tavana, O. & Gu, W. Understanding the complexity of p53 in a new era of tumor suppression. Cancer Cell 42, 946–967 (2024).
Toledo, F. & Wahl, G. M. Regulating the p53 pathway: In vitro hypotheses, in vivo veritas. Nat. Rev. Cancer 6, 909–923 (2006).
Levine, A. J. p53, the cellular gatekeeper for growth and division. Cell 88, 323–331 (1997).
Gu, W. & Roeder, R. G. Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell 90, 595–606 (1997).
Liu, Y., Tavana, O. & Gu, W. p53 modifications: exquisite decorations of the powerful guardian. J. Mol. Cell Biol. 11, 564–577 (2019).
Caspa Gokulan, R. et al. Protein adduction causes non-mutational inhibition of p53 tumor suppressor. Cell Rep. 42, 112024 (2023).
Zhang, X. et al. TRAF6 restricts p53 mitochondrial translocation, apoptosis, and tumor suppression. Mol. Cell 64, 803–814 (2016).
Huai, W. et al. KAT8 selectively inhibits antiviral immunity by acetylating IRF3. J. Exp. Med. 216, 772–785 (2019).
De Leo, A. et al. Glucose-driven histone lactylation promotes the immunosuppressive activity of monocyte-derived macrophages in glioblastoma. Immunity 57, 1105–1123.e8 (2024).
Wang, F. et al. Glycolytic stimulation is not a requirement for M2 macrophage differentiation. Cell Metab. 28, 463–475.e4 (2018).
Zhang, D. et al. Lysine L-lactylation is the dominant lactylation isomer induced by glycolysis. Nat. Chem. Biol. 21, 91–99 (2025).
Zhang, N. et al. α-myosin heavy chain lactylation maintains sarcomeric structure and function and alleviates the development of heart failure. Cell Res. 33, 679–698 (2023).
Zhou, M.-M. & Cole, P. A. Targeting lysine acetylation readers and writers. Nat. Rev. Drug Discov. 24, 112–133 (2025).
Bockwoldt, M. et al. Identification of evolutionary and kinetic drivers of NAD-dependent signaling. Proc. Natl. Acad. Sci. USA. 116, 15957–15966 (2019).
Li, H., Sun, L., Gao, P. & Hu, H. Lactylation in cancer: current understanding and challenges. Cancer Cell 42, 1803–1807 (2024).
Fan, H. et al. Lactylation: novel epigenetic regulatory and therapeutic opportunities. Am. J. Physiol. Endocrinol. Metab. 324, E330–E338 (2023).
Burrell, J. A. & Stephens, J. M. KAT8, lysine acetyltransferase 8, is required for adipocyte differentiation in vitro. Biochim. Biophys. Acta Mol. Basis Dis. 1867, 166103 (2021).
Liu, T.-W., Zhao, Y.-M., Jin, K.-Y., Wang, J.-X. & Zhao, X.-F. KAT8 is upregulated and recruited to the promoter of Atg8 by FOXO to induce H4 acetylation for autophagy under 20-hydroxyecdysone regulation. J. Biol. Chem. 300, 105704 (2024).
Lin, H.-P. et al. Destabilization of fatty acid synthase by acetylation inhibits de novo lipogenesis and tumor cell growth. Cancer Res. 76, 6924–6936 (2016).
Sykes, S. M. et al. Acetylation of the p53 DNA-binding domain regulates apoptosis induction. Mol. Cell 24, 841–851 (2006).
Liu, Y. et al. WSTF acetylation by MOF promotes WSTF activities and oncogenic functions. Oncogene 39, 5056–5067 (2020).
Wu, Y. et al. Disrupting the phase separation of KAT8-IRF1 diminishes PD-L1 expression and promotes antitumor immunity. Nat. Cancer 4, 382–400 (2023).
Liu, N. et al. ING5 is a Tip60 cofactor that acetylates p53 in response to DNA damage. Cancer Res. 73, 3749–3760 (2013).
Tang, Y.-J. et al. Irisin attenuates type 1 diabetic cardiomyopathy by anti-ferroptosis via SIRT1-mediated deacetylation of p53. Cardiovasc. Diabetol. 23, 116 (2024).
Luo, J. et al. Acetylation of p53 augments its site-specific DNA binding both in vitro and in vivo. Proc. Natl. Acad. Sci. USA. 101, 2259–2264 (2004).
Ikura, T. et al. Involvement of the TIP60 histone acetylase complex in DNA repair and apoptosis. Cell 102, 463–473 (2000).
Fuchs, M. et al. The p400 complex is an essential E1A transformation target. Cell 106, 297–307 (2001).
Brooks, C. L. & Gu, W. The impact of acetylation and deacetylation on the p53 pathway. Protein Cell 2, 456–462 (2011).
Tang, Y., Luo, J., Zhang, W. & Gu, W. Tip60-dependent acetylation of p53 modulates the decision between cell-cycle arrest and apoptosis. Mol. Cell 24, 827–839 (2006).
Christidi, E. & Brunham, L. R. Regulated cell death pathways in doxorubicin-induced cardiotoxicity. Cell Death Dis. 12, 339 (2021).
Li, M. The role of P53 up-regulated modulator of apoptosis (PUMA) in ovarian development, cardiovascular and neurodegenerative diseases. Apoptosis 26, 235–247 (2021).
Luo, W. et al. Critical role of the cGAS-STING pathway in doxorubicin-induced cardiotoxicity. Circ. Res. 132, e223–e242 (2023).
Lee, J. M., Hammarén, H. M., Savitski, M. M. & Baek, S. H. Control of protein stability by post-translational modifications. Nat. Commun. 14, 201 (2023).
Zhang, X.-L. et al. K235 acetylation couples with PSPC1 to regulate the m6A demethylation activity of ALKBH5 and tumorigenesis. Nat. Commun. 14, 3815 (2023).
Wang, M. et al. Competitive antagonism of KAT7 crotonylation against acetylation affects procentriole formation and colorectal tumorigenesis. Nat. Commun. 16, 2379 (2025).
Yu, H. et al. Neddylation inhibitor MLN4924 enhances H3K18 lactylation via binding to LDH and downregulates ITGB4 to block metastasis. J. Biol. Chem. 301, 110575 (2025).
Yu, J. et al. Histone lactylation drives oncogenesis by facilitating m6A reader protein YTHDF2 expression in ocular melanoma. Genome Biol. 22, 85 (2021).
Li, L. et al. Lysine acetyltransferase 8 is involved in cerebral development and syndromic intellectual disability. J. Clin. Investig. 130, 1431–1445 (2020).
O’Brien, M. E. R. et al. Reduced cardiotoxicity and comparable efficacy in a phase III trial of pegylated liposomal doxorubicin HCl (CAELYX/doxil) versus conventional doxorubicin for first-line treatment of metastatic breast cancer. Ann. Oncol. 15, 440–449 (2004).
Pujade-Lauraine, E. et al. Pegylated liposomal doxorubicin and carboplatin compared with paclitaxel and carboplatin for patients with platinum-sensitive ovarian cancer in late relapse. J. Clin. Oncol. 28, 3323–3329 (2010).
Belger, C., Abrahams, C., Imamdin, A. & Lecour, S. Doxorubicin-induced cardiotoxicity and risk factors. Int. J. Cardiol. Heart Vasc. 50, 101332 (2024).
Curigliano, G. et al. Cardiotoxicity of anticancer treatments: epidemiology, detection, and management. CA Cancer J. Clin. 66, 309–325 (2016).
Kuno, A. et al. SIRT1 in the cardiomyocyte counteracts doxorubicin-induced cardiotoxicity via regulating histone H2AX. Cardiovasc. Res. 118, 3360–3373 (2023).
Chen, S. et al. PDE10A inactivation prevents doxorubicin-induced cardiotoxicity and tumor growth. Circ. Res. 133, 138–157 (2023).
Zhang, X. et al. FNDC5 alleviates oxidative stress and cardiomyocyte apoptosis in doxorubicin-induced cardiotoxicity via activating AKT. Cell Death Differ. 27, 540–555 (2020).
Wang A et al. Cardiac SIRT1 ameliorates doxorubicin-induced cardiotoxicity by targeting sestrin 2. Redox Biol. 52, 102310 (2022).
Sheibani, M. et al. Doxorubicin-induced cardiotoxicity: an overview on pre-clinical therapeutic approaches. Cardiovasc. Toxicol. 22, 292–310 (2022).
Tebbi, C. K. et al. Dexrazoxane-associated risk for acute myeloid leukemia/myelodysplastic syndrome and other secondary malignancies in pediatric Hodgkin’s disease. J. Clin. Oncol. 25, 493–500 (2007).
Dempke, W. C. M. et al. Anthracycline-induced cardiotoxicity—are we about to clear this hurdle? Eur. J. Cancer 185, 94–104 (2023).
Rosenstock, J. et al. Effect of linagliptin vs glimepiride on major adverse cardiovascular outcomes in patients with type 2 diabetes: the CAROLINA randomized clinical trial. JAMA 322, 1155–1166 (2019).
Pogátsa, G. What kind of cardiovascular alterations could be influenced positively by oral antidiabetic agents? Diabetes Res. Clin. Pract. 31, S27–S31 (1996).
Chen, T. et al. iProX in 2021: connecting proteomics data sharing with big data. Nucleic Acids Res. 50, D1522–D1527 (2022).
Ma, J. et al. iProX: an integrated proteome resource. Nucleic Acids Res. 47, D1211–D1217 (2019).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82473451, 82503937, 82403751, 82200569, 82503937, and 82472930), Guangzhou Basic and Applied Research Project (2025A04J4446), Guangdong Basic and Applied Basic Research Foundation (2024A1515012875), Innovative Clinical Technique of Guangzhou, and National Key Clinical Discipline.
Author information
Authors and Affiliations
Contributions
H.S.L., Z.W.Z., L.K. and W.X.L. conceived the ideas and designed the experiments. H.S.L., Z.L., D.X.L., H.X., X.H.Y., C.Z., W.X.L. and Z.W.Z. performed the experiments. H.S.L., S.J.L., Z.W.Z., L.K. and W.X.L. analyzed and interpreted the data. L.K., Z.W.Z., W.X.L. and H.S.L. wrote the manuscript. L.K., Z.W.Z., W.X.L., H.S.L., Z.L., D.X.L., H.X., X.H.Y. and C.Z. revised the paper. H.S.L. and S.J.L. performed the statistical analysis. All authors read and approved the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Edward Seto, who co-reviewed with Changmin Peng; Tapas Kundu; Bingteng Xie and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, H., Li, Z., Lei, D. et al. Lactylation at lysine 145 fosters KAT8-TIP60 complex formation to promote p53 acetylation at lysine 120 and its pro-apoptotic function. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71108-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71108-5