Abstract
Nitrogen (N) availability regulates primary productivity and hence directly affects global oceanic carbon sequestration. Global fjords account for up to 11% of marine carbon burial. However, N loss via sediment burial remains largely unquantified. Here, we show that global fjords are hotspots of N burial, accounting for up to 18% of oceanic N burial despite only covering 0.1% of the ocean area. Burial is the dominant N loss mechanism, exceeding microbial N loss via denitrification and anammox, which are generally considered the major N loss mechanisms. Microbial N loss dominates in anoxic fjords and appears to be a function of temperature and nutrient availability. Overall, fjords efficiently sequester excess N in sediments over long time scales. Accelerated warming will promote both N burial from increased primary production and microbial N loss from warmer temperatures, affecting N budgets in fjords and in the ocean in general.
Similar content being viewed by others
Introduction
Nitrogen (N) availability is intimately linked to carbon held within marine organic matter1,2. In addition to typical blue carbon habitats such as mangroves, seagrass meadows, and salt marshes, fjords are also key coastal marine ecosystems, burying ~11% of the global marine organic carbon3,4. Since N availability is a key driver of marine primary productivity, N uptake and loss are intimately linked to the efficiency of carbon sequestration5. Growing anthropogenic N inputs to coastal oceans have promoted eutrophication and subsequent oxygen (O2) depletion6. Additionally, warming-induced stratification reduces O2 supply to fjord deep waters, thereby favouring deoxygenation. Coastal N enrichments not only modify carbon sequestration efficiency7,8, but also enhance greenhouse gas emissions, which offset the climate-mitigating capacity of coastal systems9,10. Quantification of N loss in mid- to high-latitude blue carbon ecosystems is hence critical to understanding their role in mitigating N enrichment from both direct (agricultural runoff, aquaculture, and wastewater) and indirect (glacial meltwater and soil erosion) sources.
Fjords are mid- to high-latitude coastal ecosystems with steep catchments and restricted deep water movement. Such features result in high sedimentation and organic matter burial11. Despite numerous studies on carbon cycling in global fjords3,12, less is known about N cycling processes, including burial rates of blue nitrogen–N sequestered in marine sediments. Excessive N in fjords can be removed via microbial N2 production as well as burial of particulate organic N and mineral-bound inorganic N. Denitrification, the microbial reduction of nitrate to N2, is often considered the main N removal mechanism in the ocean13,14. As a consequence, earlier fjord studies have mainly focused on N loss via denitrification15,16. The contribution of global fjords to marine N burial17 remains largely unknown. In addition, the overall importance of anammox–the second most important microbial N2 production pathway18–remains unclear in fjord systems.
Fjord N loss can be directly and indirectly related to anthropogenic stressors. Weak mixing limits O2 replenishment in fjord deep waters, which may lead to hypoxic or even anoxic conditions19. Deoxygenation has been spreading in fjords worldwide, stimulating microbial N loss20,21,22,23. As a consequence of increased anthropogenic N loads to the ocean, N inventories in global fjords have increased over the past decades24. Anthropogenic activities also indirectly impact N cycles in remote, polar fjords25. Arctic atlantification, i.e., the extension of warm, nutrient-rich Atlantic waters into the Arctic26, amplifies warming in the Arctic Ocean27, favouring deoxygenation28 and thereby altering microbial N transformation29. Accelerating glacier melting influences fjord biogeochemistry via the introduction of N-rich meltwater30,31. It is thus essential to resolve key N loss mechanisms to understand N cycling in global fjords under increasing anthropogenic pressures.
Here, we show that N burial is the main N loss pathway in fjords worldwide by combining original measurements from five fjords in Sweden and Iceland with a global compilation of data from 74 fjords. N burial rates were derived using a compiled database of 121 sites worldwide, each with measured mass accumulation rates and sediment N content. The contribution of N burial to total N loss was then contrasted to empirical rates of microbial N2 production via denitrification and anammox. We used a weighted bootstrap analysis that corrects for sampling biases to upscale global N burial and N2 production rates32. Finally, we show that N2 production is primarily stimulated by O2-depletion, increasing water temperature, and inorganic N contents. Ongoing anthropogenic impacts will thus progressively promote the dominance of microbial N loss in fjords globally.
Results and discussion
Large nitrogen burial rates in global fjords
Nitrogen burial, represented as nitrogen accumulation rates (NAR), ranged from 0.10 to 16 g N m−2 yr−1 in global fjords (Fig. 1a). Our spatial weighted bootstrap analysis yielded a global NAR median (interquartile range, IQR) of 3.9 (3.7 – 4.1) g N m−2 yr−1 (Fig. 2a). This areal rate exceeds most marine systems and even global lakes (2.5 g N m−2 yr−1)33, implying that fjords are hotspots for N burial. Arctic, North Atlantic, and North Pacific fjords exhibited N burial rates 14 to 19% higher than the global median, whereas those in the South Pacific (Chile and New Zealand) and Antarctica have rates 45 to 85% lower than the median (Fig. 2a). Fjord N burial rates significantly increased with latitude (Fig. S1). Large N burial rates were mostly observed in high-latitude fjords ( ≥ 60°) in Greenland, Canadian Arctic, and Svalbard (Fig. S2). Scaling to the updated global fjord area (259,899 km2; ref. 34), we estimated a median (IQR) global N burial of 1.0 (1.0 – 1.1) Tg N yr-1 (Table S1), representing 2–10% of N burial in global ocean sediments (10–58 Tg N yr−1; ref. 17. and references therein). Using the larger, commonly referred fjord area (455,535 km2; ref. 35) increases estimate to median of 1.8 (1.7–1.8) Tg N yr-1, or 3–18% of global marine N burial17. Despite only representing 0.1% of the global ocean area34,36, such disproportionately high N burial rates reveal that fjords are hotspots for N burial in the ocean.
Global distribution of fjord (a) sediment nitrogen burial (n = 79) and (b) sediment or water column N2 production rates (n = 20). Bolded, colour-coded coastlines indicate fjord regions. Bars indicate rates at the corresponding latitude. c Contribution of sediment burial (orange) and N2 production (teal) to total nitrogen loss (sediment burial + N2 production) in fjords where both processes were measured, and * and ** denote hypoxic and anoxic fjords, respectively. d Rates of sediment burial (NAR) and N2 production (N2) in fjords globally.
The range and variability of sediment (a) nitrogen accumulation rates (NAR), (b) mass accumulation rates (MAR), and (c) sediment carbon to nitrogen molar ratio (C:N) across ocean basins (colour-coded). Dashed horizontal line indicates bootstrapped global median value. Crosses indicate bootstrapped medians of the corresponding region. Differences among ocean basins were assessed using permutation-based one-way tests, with global p values reported. Effect sizes were quantified using epsilon-squared (ε2) derived from the Kruskal–Wallis statistic.
Since N burial rates are derived from mass accumulation rates (MAR), the high N burial capacity is necessarily linked to MAR (Fig. 3a). Similar to C burial, N burial rates are predominantly associated with MAR3,37,38. N and C burial were highly correlated across global fjords (Fig. S1). High MAR was often observed in polar and subpolar regions (Fig. 2b). The global pattern of MAR generally followed that of N burial across ocean basins, except for the two glacier-influenced Antarctic fjords, where MAR was comparable to the global median yet with lower NAR (Fig. 2a, b). Higher NAR were observed at the head of polar fjords (e.g., Hornsund and Kongsfjorden) (Fig. S2) with marine-terminating glaciers acting as large allochthonous sediment sources5,37,39. In polar fjords, large sediment inputs from glacier meltwater and erosion11 can act as key sources of fjord inorganic N40, which could consequently sustain high N burial rates found in Arctic fjords and Greenland fjords in the North Atlantic ( > 60°N).
a Observed versus predicted fjord nitrogen accumulation rates (NAR) derived from a generalized linear model including sediment mass accumulation rates (MAR) and sediment C:N ratio (C:N). b Observed versus predicted fjord N2 production rates derived from a generalized linear model including water temperature (temp), nitrate and nitrite concentrations (NOx–), and oxygen concentrations (O2) fitted using restricted maximum likelihood, with effective degrees of freedom for the fit (edffO2) reported. Data points are colour-coded on the basis of fjord regions and the dashed line indicates the 1:1 relationship. Model coefficients and goodness-of-fit (R2) of the corresponding model are shown.
Sediment stoichiometry revealed latitudinal variations in the quality of buried organic matter, as indicated by the C:N ratio, and N burial (Fig. S1). The negative correlation between this ratio and N burial implies higher N burial rates in fjords with high labile organic matter. A clear shift from preponderantly terrestrial (refractory) to marine (labile) organic matter in sediments occurred from temperate towards polar fjords (Fig. S1). Labile organic matter from marine primary production has a lower C:N ratio ( < 10) than terrestrial organic matter ( > 15)41,42. This aligns with the low sediment C:N ratios in polar fjords, where organic matter is predominantly derived from marine primary production5. Notably, high productivity can be sustained by nutrient inputs from nutrient-rich deep water upwelling, subglacial discharge, and/or N-rich meltwater from retreating glaciers43,44,45. High sedimentation rates shorten O2 exposure for organic matter degradation, allowing efficient preservation of labile organic matter46 and associated N burial in Arctic fjords47. Combined with high sedimentation rates from retreating calving glaciers48, an even higher N burial is currently occurring in high-latitude fjords. With increasing sedimentation and decreasing organic matter C:N ratio (Fig. 3a), N burial in fjords is also expected to increase. In contrast, more refractory organic matter in temperate, terrestrial-dominated fjords (Fig. S1) reflects the underlying oligotrophic conditions with low primary production2, and associated lower MAR and N burial, particularly in the Southern Hemisphere (Fig. 2a, b).
Fjord nitrogen burial surpasses microbial nitrogen loss by denitrification and anammox
Denitrification (nitrate → N2) and anammox (ammonium + nitrite → N2) are often considered the dominant N loss pathways in marine ecosystems49. Nitrogen loss by these two N2 production processes ranged from 0.10 to 21 g N m−2 yr−1 across 20 fjords globally, including our observations from five fjords (Fig. 1b). Spatially weighted bootstrap analysis yielded a global median (IQR) of 2.4 (2.0–2.8) g N m−2 yr−1. Fjord N2 production decreased at high latitudes and increased in temperate and subpolar fjords ( < 60°) in the North Atlantic (Fig. 1a). The lower regional median ( ~ 0.9 g N m−2 yr−1) in Greenland and Svalbard likely reflects lower temperatures and organic content availability for N2 production processes compared to temperate fjords, given that both temperature and organic carbon were positively correlated with sediment N2 production (Fig. S1). Overall, water temperature, nitrate + nitrite concentrations, and oxygen concentrations are significant factors driving fjord N2 production (Fig. 3b). While based on measurements from only two anoxic fjords, N2 production was notably higher under anoxic conditions (Fig. 4a). Median N2 production rates in anoxic ( < 1 µM O2) fjords is 9 times higher than in oxic fjords (Fig. 4). Hence, anoxic fjords, in particular, are hotspots of N2 production at rates up to 6 times higher than the global fjord median.
a N2 production and (b) nitrogen accumulation rates (NAR) in anoxic, hypoxic, and oxic fjords. c Contribution of N burial to total fjord N loss (sum of N2 production and N burial) under each redox condition. Global differences among redox conditions were assessed using permutation-based one-way tests, with p values reported. Effect sizes were quantified using epsilon-squared (ε²).
Denitrification and anammox primarily occur in anoxic sediment layers, where fixed N derives mostly from nitrification or diffusion from the overlying water. Benthic denitrification increases with decreasing O2, particularly below the typical threshold of hypoxia ( < 63 µM; ref. 50.). For instance, greater sediment denitrification and anammox rates were measured when bottom water O2 concentrations decreased from 260 to 61 µM in Gullmar Fjord, Sweden (Fig. S3). Similarly, greater N2 production was observed in the hypoxic Loch Etive51 compared to oxic fjords (Fig. S4). When anoxia develops in fjord basin waters, benthic N2 production will cease due to restricted nitrification and limited nitrate concentration in deep water52. The active nitrate reduction zone then rises from the sediment to the water column53. Typically, this layer’s thickness increases orders of magnitude from millimetres-centimetres in sediments54 to decimetres-meters scale in the water column oxic-anoxic interface21,22. Such substantial increases in the volume of the nitrate reduction zone enhance overall N2 production in anoxic fjords.
Including our measurements from five fjords, paired quantifications of N burial and N2 production become available for a total of 16 fjords worldwide (Fig. 1c). N burial exceeded microbial N2 production as the major N loss mechanism in 75% of these fjords. The median N burial contribution to total N loss (N burial + N2 production) was 65% across fjords (Fig. S4). High-latitude fjords with large sedimentation rates and low N2 production rates in Greenland and Svalbard exhibit remarkably high N burial contributions reaching 90% (e.g., Kobbefjord). In contrast, microbial N2 production remarkably exceeded burial in three anoxic/hypoxic fjords (Saanich Inlet, By Fjord, and Loch Etive) (Fig. S4). However, even including exceptionally high N2 production hotspots in anoxic/hypoxic fjords, these global estimates show that N burial exceeds N2 production (Fig. 4c), challenging general paradigms for N removal in the marine environment55,56,57. Our analysis clearly indicates the importance of N burial as a long-term N storage mechanism in fjords globally.
Fate of nitrogen in deoxygenated, eutrophic and warmer fjords
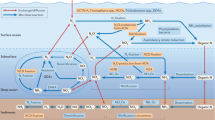
Up to nine-fold greater N2 production in anoxic than in oxic fjords (Fig. 4a) implies that complete deoxygenation shifts the dominant N removal pathway from sedimentary burial toward microbially mediated N loss. Fjord N2 production increased gradually from oxic to hypoxic conditions with a first threshold at 99 µM O2, followed by a sharp increase as O2 declined toward anoxia down to low micromolar range ( < 20 µM) (Fig. S5; Table S2). Remarkably, N2 production rates in anoxic fjords were comparable to or even higher than those in the anoxic, highly eutrophic Baltic Sea (0.30 to 11 g N m−2 yr−1)58. The substantial increase in average N2 production in anoxic fjords is primarily due to the regime shift and expansion of the nitrate reduction zone, i.e., the anoxic but nitrate containing zone58 (Fig. 5). Indeed, anoxia can inhibit nitrification and subsequentially limit N2 production, resulting in a transient phase of elevated N2 production between oxic and anoxic conditions. Since no significant differences in N burial were found between oxic and anoxic fjords (Fig. 4b) we propose that N2 production will surpass N burial and dominate N loss with developing anoxia.
Nitrogen (N) sources in both particulate and dissolved form are introduced to the fjord via terrestrial runoff, glacial melting, and oceanic inflow. Dissolved N is assimilated by primary production (or binds with minerals) and is subsequently deposited onto sediments as particulate nitrogen (PN) via sedimentation. Fractions of the deposited PN are then remineralised, nitrified and utilised for microbial N2 production, while a conspicuous remaining fraction is buried in the deep layer. Under oxic conditions ( > 100 µM O2), N burial surpasses benthic N2 production, and water column N2 production is not active. We hypothesize that N loss via sediment burial remains consistent despite the bottom water may experience occasional deoxygenation. Benthic N2 production peaks at typical hypoxic thresholds (63 µM O2) as low O2 levels stimulate microbial N2 production, yet it diminishes when approaching anoxia. Water column N2 production dominates fjord N loss when anoxia (defined as <1 µM O2) develops in bottom water.
Combining the two mechanisms, median (IQR) total N loss was 5.3 (3.5–6.1) g N m−2 yr−1 in oxic fjords and increased to 6.2 – 14.0 and to 3.6 – 25.0 g N m−2 yr−1 in hypoxic and anoxic fjords, respectively. The extent of future changes in fjord N loss pathways are intrinsically sensitive to perturbations of O2 levels, temperature and nutrient availability (Fig. 3b), which are all intimately linked to a warming climate59. In particular, intensified warming in the Arctic, or Arctic amplification, will stimulate deoxygenation and will likely create hypoxic zones in narrow and inner coastal basins28. Hence, we expect a stronger contribution of N2 production to N loss in fjords as currently observed in hypoxic and anoxic fjords worldwide (Fig. 4c). Ocean warming also promotes the northward expansion of warmer, nutrient-rich waters from the Atlantic into the Arctic60,61,62. Warmer fjord waters favour sediment N2 production (Fig. S1)63. Higher inorganic N availability (for example from increased Atlantic inflow64) can also stimulate sediment N2 production (Fig. 3b), particularly via anammox65. In addition to the direct stimulatory effects of warming and nutrient enrichment onto N2 production (Fig. 3b), the increased availability of labile organic matter due to atlantification-driven primary production may increase sediment N2 production in Arctic fjords45,63,66. Climate change stimulates primary production in high-latitude fjords and subsequent deposition of labile organic matter on sediments45, favouring both N burial and benthic N2 production (Fig. S1). Combined with potentially higher N burial due to warming-mediated glacial retreat and increased sedimentation rates48, our results suggest an increased N loss in global fjords along with a warmer and less oxygenated ocean.
Our global analysis demonstrates that sediment burial is the most effective N sink mechanism in fjord systems under current conditions. Despite covering <0.1% of the ocean, fjords contribute up to 18% of global N burial. This makes them hotspots of blue nitrogen, i.e., nitrogen sequestered in marine sediments. On average across fjords globally, sediment burial accounts for 60.6 ± 5.0% of total N loss, exceeding denitrification (34.8 ± 4.5%) and anammox (4.6 ± 2.0%). Thus, burial plays a surprisingly significant role in the long-term N removal of fjords globally, making it a crucial mechanism for mitigating coastal N enrichment at medium and high latitudes. Additionally, N burial is an effective removal pathway with minimum climatic impact, compared to microbial nitrification and denitrification, which produce the greenhouse gas nitrous oxide (N2O; equivalent to ~1% of N2 production in an anoxic fjord; ref. 21,67.). Burial in fjords is driven by sedimentation rates and the higher lability of deposited organic particles (Fig. 3a), which can be favoured by increasing rates of primary production, particle deposition, and glacial retreat68,69. However, natural hazards such as landslides and floods can alter sediment delivery and hence N burial efficiency70,71.
The control of environmental factors on fjord microbial N loss via denitrification and anammox is intricate72. At the regional scale, warming and increasing N concentrations will enhance microbial N loss following expanding deoxygenated areas24,28. Given the hydrogeomorphic characteristics of fjords and the warming climate, O2 deficiency is expected to develop more frequently, including high-latitude fjords impacted by Arctic amplification and atlantification26,59. Indeed, we show that microbial N loss dominates over geological burial loss due to the shift in N2 production regimes following the development of anoxia (Fig. 5). Rates of microbial N loss are governed by redox conditions, substrate availability, and the extent of the active nitrate reduction zone. Despite large rate variability, our observations establish a robust link between fjord N loss and deoxygenation. Additional work should focus on the climatic effect of fjord N loss via N2O production, and on refining the role of fjords in the global N cycle. Whether N burial can counter coastal nutrient overenrichment (i.e., eutrophication) and curb greenhouse gas emissions largely depends on coastal nutrient management. The future partitioning of N loss mechanisms in fjords is predominantly linked to their deoxygenation level, eutrophic status, and hydrological response to global warming.
Method
Global fjord database collection and upscaling
Our global fjord database combining sediment mass accumulation rates and N content, consists of 158 observations across 79 fjords. Major fjord regions are covered11, including North-western Europe (Scotland, Sweden, Norway, Iceland, and Faroe Islands), Greenland, Svalbard, Western Canada, Eastern Canada, the Canadian Arctic archipelagos, Alaska, Patagonia, New Zealand, and Antarctica. We excluded shallow fjards (which are not true fjords but rather drowned estuaries) with average water depth <10 meters, such as those in Denmark, due to the hydrodynamical and physicochemical differences. We assumed that sediment particle size was relatively homogenous across sites, given a mean porosity of 0.78 ± 0.06. Global rates were determined by upscaling to the global fjord area34,35 (Table S1).
Nitrogen burial rates, expressed as NAR, were further compared to microbial N loss rates (i.e., N2 production) that were measured in the corresponding fjords. Our global compilation of fjord N2 production measurements consists of 47 observations across 20 fjords. Reported areal rates were measured by either intact core incubations, water column incubations, or in situ chamber incubations using isotope pairing techniques73,74. Areal N2 production rates obtained in the water column were obtained in the case of anoxic fjords. Measurements of both N2 production and N burial are available for a total of 16 fjords located mainly in NW Europe, Svalbard, Greenland, and Eastern Canada.
Original observations in Swedish and Icelandic fjords
Data on both N burial and N2 production were collected in five temperate (57–62˚N) and subpolar (65°N) fjords along the west coast of Sweden and the east coast of Iceland, respectively (Fig. S6). These fjords have distinct watershed characteristics, anthropogenic influences, and redox gradients, including oxic, seasonally hypoxic, and long-term anoxic conditions (Table S3). Samples were collected at three sampling stations at the head, centre, and mouth of each fjord aboard the R/V Skagerrak. For the Swedish fjords, samples for mass accumulation rates and sediment stoichiometric analysis were collected in September-October 202175 and N2 production rates in sediments and water columns were determined by 15N-labelling incubation experiments in 2023 and 2024. For Icelandic fjords, samples for all analyses and incubations were collected in June 2023. Mass accumulation rates and sediment stoichiometry from Icelandic fjords were reported in a previous publication76, which were determined based on particle-reactive radiotracer-derived age models and sediment stoichiometry. Detailed descriptions of sampling procedures of each campaign are given in the Supplementary Materials.
Determination of nitrogen accumulation rates and N2 production
Total N burial rate, defined as N accumulation rate (NAR) in the sediment, was calculated from mass accumulation rates, porosity, bulk density, and sediment N content in collected sediment cores and in the literature (Supplementary Materials and references therein). Mass accumulation rates (MAR) were determined based on particle-reactive radiotracer-derived age models in the sediment cores and were obtained from published data75. Age models were created for each core using 210Pb,137Cs and/or 241Am profiles. Dry bulk density and porosity were determined for each sediment slice by measured wet and dry weights. Radionuclides 210Pb, 137Cs, and 241Am were analysed using an ORTEC HPGe GWL-series well-type coaxial low-background germanium detector. Linear sedimentation rates were estimated by the constant flux–constant sedimentation (CF-CS) model, ensuring a logarithmic regression fit with R² > 0.75, and were further constrained by 137Cs and 241Am peaks. Mass sediment accumulation rates were calculated using the constant rate of supply model and validated against 137Cs peaks. N accumulation rates were derived from multiplying MAR by the weight fraction of total nitrogen (TN%). When only organic carbon accumulation rates and sediment C:N ratio were reported in the literature, NAR were calculated considering the sediment stoichiometric ratio.
Sediment N2 production rates were determined through 15N-labelling incubation experiments by the revised-isotope pairing technique74. Collected intact cores were spiked with 15NO3– (11, 24, 56, and 120 µM mean final concentration) to determine rates of N2 production (denitrification and anammox) and N recycling (dissimilatory nitrate reduction to ammonium rates, DNRA). At the centre station of By Fjord, total N2 production rates were determined in the water column as N2 production is restricted in the sediment due to depletion of O2, NOx– and any other N oxide species23. 15N-labelling incubation experiments were conducted with samples collected 21 m below the surface, which covered the NOx– containing oxic-anoxic water layer. Additional incubation procedures and rate calculations are given in the Supplementary Materials.
Spatial weighted bootstrap analysis
We performed spatial weighted bootstrap analysis to minimize the impact of geographical sampling bias on our estimates of the global mean of N loss processes. Such an analysis helps to minimise uncertainties derived from small datasets with geographical sampling biases. We assigned sample weights based on their spatial proximity to other samples using the inverse weighting algorithm32,77 with a spatial scale of ~55 km (0.5 degree). Using the resulting weights (Fig. S7), we performed a bootstrap analysis to generate distributions of 1000 weighted bootstrapped means for both NAR and N2 production rates. The observed raw data distribution and bootstrapped data used in the analyses were illustrated in histograms (Fig. S8). Both spatial weighing and bootstrapping analysis were performed in R version 4.3.2.
Statistical analysis
The relationship between environmental variability and N removal rates was estimated from the correlation of N burial vs. total N2 production rates with individual environmental factors. Spearman’s rank correlation coefficients were calculated to minimize impacts of collinearity. A one-way permutation test was used to reveal the difference of NAR and N2 production rates across oxic and deoxygenated fjords, as well as the difference in mean N2 production processes between oxic and hypoxic sediment in Gullmar Fjord. To examine potential change points in the relationship between N2 production and O2 concentration across redox regimes, we fitted a Bayesian segmented regression model with three linear segments using the mcp package (Table S2). Correlations and differences across conditions were considered statistically significant with p value < 0.05. All statistical analyses were performed in R version 4.3.2.
Data availability
The compiled dataset of measurements from sampling campaigns and referenced observations of sediment burial and microbial N2 production rates used in this study is available in the Zenodo repository with accession code 18875989.
Code availability
The R code used to run the spatial bootstrap analyses is in R Markdown format and is available in the Zenodo repository with accession code 18875989.
References
Sanders, C. J. et al. Elevated rates of organic carbon, nitrogen, and phosphorus accumulation in a highly impacted mangrove wetland. Geophys. Res. Lett. 41, 2475–2480 (2014).
Hinojosa, J. L., Moy, C. M., Stirling, C. H., Wilson, G. S. & Eglinton, T. I. Carbon cycling and burial in New Zealand’s fjords. Geochem. Geophys. Geosyst. 15, 4047–4063 (2014).
Smith, R. W., Bianchi, T. S., Allison, M., Savage, C. & Galy, V. High rates of organic carbon burial in fjord sediments globally. Nat. Geosci. 8, 450–453 (2015).
James, K., Macreadie, P. I., Burdett, H. L., Davies, I. & Kamenos, N. A. It’s time to broaden what we consider a ‘blue carbon ecosystem. Glob. Change Biol. 30, e17261 (2024).
Włodarska-Kowalczuk, M. et al. Organic carbon origin, benthic faunal consumption, and burial in sediments of Northern Atlantic and Arctic Fjords (60–81°N). J. Geophys. Res.: Biogeosci. 124, 3737–3751 (2019).
Breitburg, D. et al. Declining oxygen in the global ocean and coastal waters. Science 359, eaam7240 (2018).
Macreadie, P. I. et al. Can we manage coastal ecosystems to sequester more blue carbon?. Front. Ecol. Environ. 15, 206–213 (2017).
Macreadie, P. I. et al. The future of blue carbon science. Nat. Commun. 10, 3998 (2019).
Bonaglia, S. et al. High methane emissions from an anoxic fjord driven by mixing and oxygenation. Limnol. Oceanogr. Lett. 7, 392–400 (2022).
Politi, T. et al. Global Fjords as minor sources of nitrous oxide to the atmosphere. Geophys. Res. Lett. 52, e2024GL111624 (2025).
Bianchi, T. S. et al. Fjords as Aquatic Critical Zones (ACZs). Earth-Sci. Rev. 203, 103145 (2020).
Cui, X. et al. Global fjords as transitory reservoirs of labile organic carbon modulated by organo-mineral interactions. Sci. Adv. 8, eadd0610 (2022).
Hulth, S. et al. Nitrogen removal in marine environments: recent findings and future research challenges. Mar. Chem. 94, 125–145 (2005).
Seitzinger, S. P. Denitrification in freshwater and coastal marine ecosystems: ecological and geochemical significance. Limnol. Oceanogr. 33, 702–724 (1988).
Glud, R. N., Risgaard-Petersen, N., Thamdrup, B., Fossing, H. & Rysgaard, S. Benthic carbon mineralization in a high-Arctic sound (Young Sound, NE Greenland). Mar. Ecol. Prog. Ser. 206, 59–71 (2000).
Glud, R. N., Holby, O., Hoffmann, F. & Canfield, D. E. Benthic mineralization and exchange in Arctic sediments (Svalbard, Norway). Mar. Ecol. Prog. Ser. 173, 237–251 (1998).
Zhang, X., Ward, B. B. & Sigman, D. M. Global nitrogen cycle: critical enzymes, organisms, and processes for nitrogen budgets and dynamics. Chem. Rev. 120, 5308–5351 (2020).
Dalsgaard, T., Thamdrup, B. & Canfield, D. E. Anaerobic ammonium oxidation (anammox) in the marine environment. Res Microbiol 156, 457–464 (2005).
Friedrich, J. et al. Investigating hypoxia in aquatic environments: diverse approaches to addressing a complex phenomenon. Biogeosciences 11, 1215–1259 (2014).
Pakhomova, S., Braaten, H. F. V., Yakushev, E. & Skei, J. Biogeochemical consequences of an oxygenated intrusion into an anoxic fjord. Geochem. Trans. 15, 5 (2014).
Michiels, C. C. et al. Rates and pathways of N2 production in a persistently anoxic Fjord: Saanich Inlet, British Columbia. Front. Mar. Sci. 6, 27 (2019).
Kristiansen, S. & Thorne Schaanning, M. Denitrification in the water column of an intermittently anoxic fjord. Hydrobiologia 469, 77–86 (2002).
De Brabandere, L. et al. Oxygenation of an anoxic fjord basin strongly stimulates benthic denitrification and DNRA. Biogeochemistry 126, 131–152 (2015).
Liu, X. et al. Simulated global coastal ecosystem responses to a half-century increase in river nitrogen loads. Geophys. Res. Lett. 48, e2021GL094367 (2021).
Duarte, P. et al. Ecosystem metabolism and nitrogen budget of a glacial Fjord in the Arctic. Sci. Rep. 15, 22946 (2025).
Polyakov, I. V. et al. Atlantification advances into the Amerasian Basin of the Arctic Ocean. Sci. Adv. 11, eadq7580 (2025).
Polyakov, I. V. et al. Fluctuating Atlantic inflows modulate Arctic atlantification. Science 381, 972–979 (2023).
Whitney, M. M. Observed and projected global warming pressure on coastal hypoxia. Biogeosciences 19, 4479–4497 (2022).
Yamamoto-Kawai, M., Carmack, E. & McLaughlin, F. Nitrogen balance and Arctic throughflow. Nature 443, 43–43 (2006).
Sharp, M. & Tranter, M. Glacier biogeochemistry. Geochem. Perspect. 6, 173–339 (2017).
Wadham, J. L. et al. Ice sheets matter for the global carbon cycle. Nat. Commun. 10, 3567 (2019).
Mehra, A. et al. Curation and analysis of global sedimentary geochemical data to inform earth history. GSA Today 31, 4–10 (2021).
Wang, M. et al. Human-caused increases in reactive nitrogen burial in sediment of global lakes. Innovation 2, 100158 (2021).
Laruelle, G. G., Rosentreter, J. A. & Regnier, P. Extrapolation-based regionalized re-evaluation of the global estuarine surface area. Estuaries Coasts 48, 34 (2024).
Dürr, H. H. et al. Worldwide typology of nearshore coastal systems: defining the estuarine filter of river inputs to the Oceans. Estuaries Coasts 34, 441–458 (2011).
Costello, M. J., Cheung, A. & De Hauwere, N. Surface area and the seabed area, volume, depth, slope, and topographic variation for the world’s seas, oceans, and countries. Environ. Sci. Technol. 44, 8821–8828 (2010).
Cui, X., Bianchi, T. S., Savage, C. & Smith, R. W. Organic carbon burial in fjords: terrestrial versus marine inputs. Earth Planet. Sci. Lett. 451, 41–50 (2016).
Wang, Y. et al. High rates of marine organic carbon burial on the southwest Greenland margin induced by Neoglacial advances. Commun. Earth Environ. 5, 335 (2024).
Howe, J. et al. Fjord systems and archives: A review. Geol. Soc. Lond. Spec. Publ. 344, 5–15 (2010).
Winkelmann, D. & Knies, J. Recent distribution and accumulation of organic carbon on the continental margin west off Spitsbergen. Geochem. Geophys. Geosyst. 6, Q09012 (2005).
Stein R., & Macdonald R. W. Organic Carbon Budget: Arctic Ocean vs. Global Ocean. In: The Organic Carbon Cycle in the Arctic Ocean (eds Stein R., MacDonald R. W.). Springer Berlin Heidelberg (2004).
Rullkötter J. et al. Organic Matter: The Driving Force for Early Diagenesis. In: Marine Geochemistry (eds Schulz H. D., Zabel M.). Springer Berlin Heidelberg (2006).
Hoshiba, Y., Matsumura, Y., Kanna, N., Ohashi, Y. & Sugiyama, S. Impacts of glacial discharge on the primary production in a Greenlandic fjord. Sci. Rep. 14, 15530 (2024).
Hopwood, M. J. et al. Non-linear response of summertime marine productivity to increased meltwater discharge around Greenland. Nat. Commun. 9, 3256 (2018).
Sørensen, H. L. et al. Seasonal carbon cycling in a Greenlandic fjord: an integrated pelagic and benthic study. Mar. Ecol. Prog. Ser. 539, 1–17 (2015).
Hartnett, H. E., Keil, R. G., Hedges, J. I. & Devol, A. H. Influence of oxygen exposure time on organic carbon preservation in continental margin sediments. Nature 391, 572–575 (1998).
Herbert, L. C. et al. Tight benthic-pelagic coupling drives seasonal and interannual changes in iron‑sulfur cycling in Arctic fjord sediments (Kongsfjorden, Svalbard). J. Mar. Syst. 225, 103645 (2022).
Boldt, K. V. et al. Modern rates of glacial sediment accumulation along a 15° S-N transect in fjords from the Antarctic Peninsula to southern Chile. J. Geophys. Res.: Earth Surf. 118, 2072–2088 (2013).
Devol, A. H. Denitrification, anammox, and N2 production in marine sediments. Annu. Rev. Mar. Sci. 7, 403–423 (2015).
Diaz, R. J. & Rosenberg, R. Spreading dead zones and consequences for marine ecosystems. Science 321, 926–929 (2008).
Glud, R. N. et al. Benthic carbon mineralization and nutrient turnover in a Scottish sea loch: an integrative in situ study. Aquat. Geochem. 22, 443–467 (2016).
Song, G. et al. Response of benthic nitrogen cycling to estuarine hypoxia. Limnol. Oceanogr. 66, 652–666 (2021).
Jäntti, H. & Hietanen, S. The effects of hypoxia on sediment nitrogen cycling in the Baltic Sea. Ambio 41, 161–169 (2012).
Choquel, C. et al. Denitrification by benthic foraminifera and their contribution to N-loss from a fjord environment. Biogeosciences 18, 327–341 (2021).
Codispoti, L. A. et al. The Oceanic fixed nitrogen and nitrous oxide budgets: moving targets as we enter the Anthropocene? Sci. Mar. 65, 85–105 (2000).
Gruber, N. et al. The vulnerability of the carbon cycle in the 21st century: an assessment of carbon-climate-human interactions. In The global carbon cycle:integrating humans, climate and the natural world. 45–76 (Island Press, Washington, 2004).
Galloway, J. N. et al. Nitrogen cycles: past, present, and future. Biogeochemistry 70, 153–226 (2004).
Dalsgaard, T., De Brabandere, L. & Hall, P. O. J. Denitrification in the water column of the central Baltic Sea. Geochim. Cosmochim. Acta 106, 247–260 (2013).
He, Y. et al. Arctic Amplification of marine heatwaves under global warming. Nat. Commun. 15, 8265 (2024).
Wang, Q., Shu, Q. & Wang, F. Recent emergence of Arctic atlantification dominated by climate warming. Sci. Adv. 10, eadq5235 (2024).
Wang, Q. et al. Intensification of the Atlantic water supply to the Arctic Ocean through Fram Strait induced by Arctic Sea Ice decline. Geophys. Res. Lett. 47, e2019GL086682 (2020).
Wang Q., Shu Q., Wang F. Recent emergence of Arctic atlantification dominated by climate warming. Sci. Adv. 10, eadq5235 (2024).
Canion, A. et al. Temperature response of denitrification and anaerobic ammonium oxidation rates and microbial community structure in Arctic fjord sediments. Environ. Microbiol. 16, 3331–3344 (2014).
Arrigo, K. R., Mills, M. M. & Juranek, L. W. The Arctic Ocean nitrogen cycle. J. Geophys. Res.: Biogeosci. 129, e2024JG008088 (2024).
Rysgaard, S., Glud, R. N., Risgaard-Petersen, N. & Dalsgaard, T. Denitrification and anammox activity in Arctic marine sediments. Limnol. Oceanogr. 49, 1493–1502 (2004).
Gihring, T. M., Lavik, G., Kuypers, M. M. M. & Kostka, J. E. Direct determination of nitrogen cycling rates and pathways in Arctic fjord sediments (Svalbard, Norway). Limnol. Oceanogr. 55, 740–752 (2010).
Capelle, D. W., Hawley, A. K., Hallam, S. J. & Tortell, P. D. A. multi-year time-series of N2O dynamics in a seasonally anoxic fjord: Saanich Inlet, British Columbia. Limnol. Oceanogr. 63, 524–539 (2018).
Brinkerhoff, D., Truffer, M. & Aschwanden, A. Sediment transport drives tidewater glacier periodicity. Nat. Commun. 8, 90 (2017).
Meire, L. et al. Glacier retreat alters downstream Fjord ecosystem structure and function in Greenland. Nat. Geosci. 16, 671–674 (2023).
Moreau, A.-L., Locat, J., Hill, P., Long, B. & Ouellet, Y. Resuspension potential of surficial sediments in Saguenay Fjord (Québec, Canada). Mar. Geol. 225, 85–101 (2006).
Syvitski, J. & Normandeau, A. Sediment redistribution processes in Baffin Island fjords. Mar. Geol. 458, 107024 (2023).
Barnard, R., Leadley, P. W. & Hungate, B. A. Global change, nitrification, and denitrification: a review. Global Biogeochem. Cycles 19, GB1007 (2005).
Nielsen, L. P. Denitrification in sediment determined from nitrogen isotope pairing. FEMS Microbiol. Lett. 86, 357–362 (1992).
Risgaard-Petersen, N., Nielsen, L. P., Rysgaard, S., Dalsgaard, T. & Meyer, R. L. Application of the isotope pairing technique in sediments where anammox and denitrification coexist. Limnol. Oceanogr.: Methods 1, 63–73 (2003).
Watts, E. G. et al. Burial of organic carbon in Swedish Fjord sediments: highlighting the importance of sediment accumulation rate in relation to Fjord redox conditions. J. Geophys. Res. Biogeosci. 129, e2023JG007978 (2024).
Watts, E. G. et al. Efficient burial of labile organic carbon in sediments of oxygenated Icelandic fjords. Estuaries Coasts 48, 167 (2025).
Stockey, R. G. et al. Sustained increases in atmospheric oxygen and marine productivity in the Neoproterozoic and Palaeozoic eras. Nat. Geosci. 17, 667–674 (2024).
Acknowledgements
We thank T. Cardis and M. Olsson for their contribution to the sediment sampling in Iceland and Sweden; L. Bristow for advice on water column incubation procedures and for helpful discussions; C.M. Hill for laboratory support; and crews on R/V Skagerak for logistical support. Research funding was provided by the Swedish Research Council VR to S.B. (grant no. 2022-04710). The Iceland and Swedish expeditions were partially funded by the Knut and Alice Wallenberg Foundation (grant no. 2022.0096) and the Swedish Research Council VR (grant no. 2020-00457) to I.R.S., and L.S.L. was supported by the Independent Research Fund Denmark to B.T. (grant no. 1127-00362B).
Funding
Open access funding provided by University of Gothenburg.
Author information
Authors and Affiliations
Contributions
H.L.S.C., I.R.S., and S.B. designed the research. H.L.S.C, L.S.L., T.P., I.R.S and S.B. conducted field sampling and collection of geochemical samples. H.L.S.C., L.S.L., and T.P. performed incubation experiments for microbial process rates. C.S. performed analyses on geochemical characteristics and accumulation rates, and provided unpublished geochemical data. H.L.S.C. and L.S.L. performed analyses on GC-IRMS. H.L.S.C. and B.T. conducted formal analysis, compiled geochemical databases, and calculated process rates. S.B. led the research and supervised the project. S.B. and I.R.S. acquired funding for the research. H.L.S.C. wrote the original draft of the manuscript with important contributions from S.B. All authors were responsible for the review and editing of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ludovic Pascal and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cheung, H.L.S., Levin, L.S., Smeaton, C. et al. Long-term nitrogen burial exceeds denitrification in global fjords. Nat Commun 17, 3148 (2026). https://doi.org/10.1038/s41467-026-71116-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-71116-5