Abstract
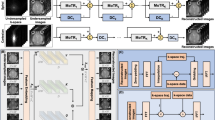
Chemical exchange saturation transfer (CEST) MRI could detect proteins/peptides, creatine, glucose, and glycogen by labeling their exchangeable amide, amine, and hydroxyl groups respectively, via frequency-selective RF pulses. Without the need for contrast agents or specialized hardware, CEST can be conveniently integrated into existing clinical MR protocols. However, its abdominal application is limited by long scan time (> 5 min) and susceptibility to respiratory motion (60–70% successful scan rate). We develop an ultra-fast 3D CEST MRI approach using spatial-spectral encoding (SSE), enabling a full spectral scan of whole-liver 3D images within a single breath-hold. SSE-CEST employs an efficient z-ω encoding pattern by applying a saturation gradient, followed by a data-driven spatial spectral reconstruction based on the low-rankness of CEST spectra. SSE-CEST is comprehensively evaluated in glycogen phantoms, ex vivo porcine liver, healthy volunteers and patients. Single breath-hold SSE-CEST largely improves successful rate, with a correlation of 0.95 between two repeated scans. SSE-CEST enables the detection of multi-metabolite changes in the liver and pancreas after an overnight fasting, and the dynamic mapping of hepatic glucose metabolism during an oral glucose test. For liver cancer patients, SSE could differentiate active lesions from post-treatment necrosis, featuring superior in-slice spatial resolution and motion-stabilized images. SSE-CEST MRI potentially could facilitate the diagnosis and patient management for liver and other abdominal diseases.
Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. The raw data and quantification results from a representative participant in the fasting experiments are available at https://doi.org/10.5281/zenodo.18795840. Source data are provided with this paper.
Code availability
The SSE-CEST reconstruction code is available at https://doi.org/10.5281/zenodo.18795840, the analysis code is available at http://github.com/easycest/SSE-CEST.
References
Devarbhavi, H. et al. Global burden of liver disease: 2023 update. J. Hepatol. 79, 516–537 (2023).
Banerjee, R. et al. Multiparametric magnetic resonance for the non-invasive diagnosis of liver disease. J. Hepatol. 60, 69–77 (2014).
Qayyum, A. MR Spectroscopy of the Liver: Principles and Clinical Applications. Radiographics 29, 1653–U1142 (2009).
Befroy, D. E. et al. Direct assessment of hepatic mitochondrial oxidative and anaplerotic fluxes in humans using dynamic 13C magnetic resonance spectroscopy. Nat. Med 20, 98 (2014).
Casey, A. et al. Effect of carbohydrate ingestion on glycogen resynthesis in human liver and skeletal muscle, measured by 13C MRS. Am. J. Physiol. -Endoc M 278, E65–E75 (2000).
Solga, S. F., Horska, A., Clark, J. M. & Diehl, A. M. Hepatic 31P magnetic resonance spectroscopy: a hepatologist’s user guide. Liver Int 25, 490–500 (2005).
van Zijl, P. C., Jones, C. K., Ren, J., Malloy, C. R. & Sherry, A. D. MRI detection of glycogen in vivo by using chemical exchange saturation transfer imaging (glycoCEST). Proc. Natl. Acad. Sci. USA 104, 4359–4364 (2007).
Zhou, Y. et al. Magnetic resonance imaging of glycogen using its magnetic coupling with water. Proc. Natl. Acad. Sci. USA 117, 3144–3149 (2020).
Zhou, J. Y., Payen, J. F., Wilson, D. A., Traystman, R. J. & van Zijl, P. C. M. Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat. Med 9, 1085–1090 (2003).
Cai, Z. et al. Non-invasive mapping of brown adipose tissue activity with magnetic resonance imaging. Nat. Metab. 6, 1367–1379 (2024).
Walker-Samuel, S. et al. Imaging of glucose uptake and metabolism in tumors. Nat. Med 19, 1067 (2013).
Zhou, J. et al. Differentiation between glioma and radiation necrosis using molecular magnetic resonance imaging of endogenous proteins and peptides. Nat. Med 17, 130–134 (2011).
Zhou, J. et al. Review and consensus recommendations on clinical APT-weighted imaging approaches at 3T: Application to brain tumors. Magn. Reson. Med. 88, 546–574 (2022).
Togao, O. et al. Amide proton transfer imaging of adult diffuse gliomas: correlation with histopathological grades. Neuro-Oncol. 16, 441–448 (2014).
Paech, D. et al. Assessing the predictability of mutation and methylation status in glioma patients using relaxation-compensated multipool CEST MRI at 7.0 T. Neuro-Oncol. 20, 1661–1671 (2018).
Sagiyama, K. et al. In vivo chemical exchange saturation transfer imaging allows early detection of a therapeutic response in glioblastoma. P Natl. Acad. Sci. USA 111, 4542–4547 (2014).
Vinogradov, E., Keupp, J., Dimitrov, I. E., Seiler, S. & Pedrosa, I. CEST-MRI for body oncologic imaging: are we there yet?. Nmr Biomed. 36, e4906 (2023).
Seo, N. et al. Liver MRI with amide proton transfer imaging: feasibility and accuracy for the characterization of focal liver lesions. Eur. Radio. 31, 222–231 (2021).
Qi, X. et al. Image quality assessment and feasibility of three-dimensional amide proton transfer-weighted imaging for hepatocellular carcinoma. Quant. Imaging Med Surg. 14, 1778–1790 (2024).
Wang, Y. X. J., Dou, W. Q., Shen, Z. W. & Zhang, Y. An update on liver chemical exchange saturation transfer imaging with a focus on clinical translation. Quant. Imag. Med. Surg. 13, 4057–4076 (2023).
Song, X. et al. Label-free in vivo molecular imaging of underglycosylated mucin-1 expression in tumour cells. Nat. Commun. 6, 6719 (2015).
Xu, X., Leforestier, R., Xia, D., Block, K. T. & Feng, L. MRI of GlycoNOE in the human liver using GraspNOE-Dixon. Magn. Reson Med 93, 507–518 (2025).
Chen, Z. et al. Free-breathing abdominal chemical exchange saturation transfer imaging using water presaturation and respiratory gating at 3.0. T. Nmr Biomed. 37, e5134 (2024).
Han, P. et al. Free-breathing 3D CEST MRI of human liver at 3.0 T. Magn. Reson Med 89, 738–745 (2023).
Xu, X., Lee, J. S. & Jerschow, A. Ultrafast scanning of exchangeable sites by NMR spectroscopy. Angew. Chem. Int Ed. Engl. 52, 8281–8284 (2013).
Wilson, N. E., D’Aquilla, K., Debrosse, C., Hariharan, H. & Reddy, R. Localized, Gradient-Reversed Ultrafast Z-Spectroscopy In Vivo at 7T. Magn. Reson Med 76, 1039–1046 (2016).
Bie, C. X., van Zijl, P. C. M., Mao, D. & Yadav, N. N. Ultrafast Z-spectroscopic imaging in vivo at 3T using through-slice spectral encoding (TS-UFZ). Magn. Reson Med 89, 1429–1440 (2023).
Zhao, Y. B. et al. Ultrafast J-resolved magnetic resonance spectroscopic imaging for high-resolution metabolic brain imaging. Nat. Biomed. Eng. 9, 1835–1847 (2025).
van Zijl, P. C. M., Lam, W. W., Xu, J. D., Knutsson, L. & Stanisz, G. J. Magnetization Transfer Contrast and Chemical Exchange Saturation Transfer MRI. Features and analysis of the field-dependent saturation spectrum. Neuroimage 168, 222–241 (2018).
Knutsson, L. et al. Dynamic glucose enhanced imaging using direct water saturation. Magn. Reson Med 94, 15–27 (2025).
Seidemo, A. et al. Towards robust glucose chemical exchange saturation transfer imaging in humans at 3 T: Arterial input function measurements and the effects of infusion time. NMR Biomed. 35, e4624 (2022).
Xu, X. et al. d-glucose weighted chemical exchange saturation transfer (glucoCEST)-based dynamic glucose enhanced (DGE) MRI at 3T: early experience in healthy volunteers and brain tumor patients. Magn. Reson Med 84, 247–262 (2020).
Jones, K. M. et al. Clinical Translation of Tumor Acidosis Measurements with AcidoCEST MRI. Mol. Imaging Biol. 19, 617–625 (2017).
Li, Y. F., Chen, W. X., Wang, Y. & Song, X. L. Two-point B1 correction for CEST MRI by fusing voxel-wise interpolation and T1W voxel-clustering. Magn. Reson. Med. 95, 1223–1238 (2025).
Acknowledgements
This work was supported by National Key R&D Program of China (2022YFC3602500, X.S.), National Natural Science Foundation of China (12271434, X.H.), the Natural Science Basic Research Plan in Shaanxi Province of China (2023-JC-JQ-57, X.H.), stipend to C.L. from Jingjinji National Center of Technology Innovation, and Tsinghua University Initiative Research Program to X.S.; We thank Kaixiang Li for assistance with figure preparation and data organization.
Author information
Authors and Affiliations
Contributions
C.L., N.G., and X.S. conceived the project, designed the experiments and wrote the manuscript; X.S. supervised the study. C.L. developed the acquisition and reconstruction technique. C.L. and N.G. performed all the MRI experiments. H.R., H.L., J.H., Z.L., and X.H. assisted with data analysis. B.Z., Y.Y., and Z.Z. recruited the patients and performed clinical diagnoses. All authors discussed the results and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
X.S., C.L., and N.G. have filed a patent application on the SSE-CEST method (PCT/CN2023/125056), The other authors declare no competing financial interests.
Peer review
Peer review information
Nature Communications thanks Debiao Li and the other anonymous reviewer for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, C., Gao, N., Ren, H. et al. Single-breath-hold 3D abdominal metabolic MRI enables label-free diagnosis of liver cancer. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71124-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71124-5