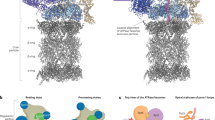
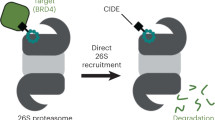
Abstract
Classical proteolysis targeting chimeras (Protacs) bind specific targets and E3 ubiquitin-ligases, promoting ubiquitination and degradation of targets by the proteasome. Multiple chimeras that degrade proteins relevant in several diseases have been developed, and the number is quickly increasing, indicating their therapeutic projection. Given the specificities of proteolytic pathways and limitations in E3-based Protacs, alternative strategies in targeted protein degradation are pursued. Herein, using two targets relevant in oncology as models (IMPDH2 and CERT1), we provide proof of concept for 26S-oriented compounds based on small-molecule ligands of USP14, a 26S-associated deubiquitinase involved in substrate processing and allosteric regulation of 26S activity. Our findings will expand the potential of targeted protein degradation.
Similar content being viewed by others
Data availability
Supporting computational and chemical methods (including compound synthesis and NMR spectra) are described in the supplementary file, which also includes Supplementary molecular modeling results, supplementary tables, figures, movies, PDB-formatted files. The raw data generated in this study are provided in the Supplementary Source Data file. The computational source data can be accessed at https://doi.org/10.5281/zenodo.18313893. Source data are provided with this paper.
References
Pettersson, M. & Crews, C. M. PROteolysis TArgeting Chimeras (PROTACs) — Past, present and future. Drug Discov. Today Technol 31, 15–27 (2019).
Verma, R., Mohl, D. & Deshaies, R. J. Harnessing the Power of Proteolysis for Targeted Protein Inactivation. Mol. Cell 77, 446–460 (2020).
Lee, J. et al. Discovery of E3 Ligase Ligands for Target Protein Degradation. Molecules 27, 6515 (2022).
Cai, H., Zhang, T. & Hu, Y. Global landscape of PROTAC: Perspectives from patents, drug pipelines, clinical trials, and licensing transactions. Eur. J. Med. Chem. 299, 118055 (2025).
Petrilli, W. L. et al. From Screening to Targeted Degradation: Strategies for the Discovery and Optimization of Small Molecule Ligands for PCSK9. Cell Chem. Biol. 27, 32–40.e3 (2020).
Lai, A. C. et al. Modular PROTAC Design for the Degradation of Oncogenic BCR-ABL. Angewandte Chemie International Edition 55, 807–810 (2016).
Ottis, P. & Crews, C. M. Proteolysis-Targeting Chimeras: Induced Protein Degradation as a Therapeutic Strategy. https://doi.org/10.1021/acschembio.6b01068 (2017)
Gustafson, J. L. et al. Small-Molecule-Mediated Degradation of the Androgen Receptor through Hydrophobic Tagging. Angewandte Chemie - International Edition 54, 9659–9662 (2015).
Dong, Y. et al. Cryo-EM structures and dynamics of substrate-engaged human 26S proteasome. Nature 565, 49–55 (2019).
Lander, G. C. et al. Complete subunit architecture of the proteasome regulatory particle. Nature 482, 186–191 (2012).
Glickman, M. H. et al. A Subcomplex of the Proteasome Regulatory Particle Required for Ubiquitin-Conjugate Degradation and Related to the COP9-Signalosome and eIF3. Cell 94, 615–623 (1998).
Groll, M. et al. Structure of 20S proteasome from yeast at 2.4 A resolution. Nature 386, 463–471 (1997).
Verma, R. et al. Role of Rpn11 metalloprotease in deubiquitination and degradation by the 26S proteasome. Science 298, 611–615 (2002).
Lee, B.-H. et al. USP14 deubiquitinates proteasome-bound substrates that are ubiquitinated at multiple sites. Nature 532, 398–401 (2016).
Lee, B.-H. et al. Enhancement of proteasome activity by a small-molecule inhibitor of USP14. Nature 467, 179–184 (2010).
Yao, T. et al. Proteasome recruitment and activation of the Uch37 deubiquitinating enzyme by Adrm1. Nat. Cell Biol. 8, 994–1002 (2006).
Zhang, S. et al. USP14-regulated allostery of the human proteasome by time-resolved cryo-EM. Nature 605, 567–574 (2022).
Hanna, J. et al. Deubiquitinating enzyme Ubp6 functions noncatalytically to delay proteasomal degradation. Cell 127, 99–111 (2006).
Boselli, M. et al. An inhibitor of the proteasomal deubiquitinating enzyme USP14 induces tau elimination in cultured neurons. Journal of Biological Chemistry 292, 19209–19225 (2017).
Wang, Y. et al. Small molecule inhibitors reveal allosteric regulation of USP14 via steric blockade. Cell Res 28, 1186–1194 (2018).
Bard, J. A. M., Bashore, C., Dong, K. C. & Martin, A. The 26S Proteasome Utilizes a Kinetic Gateway to Prioritize Substrate Degradation. Cell 177, e15 (2019).
de la Peña, A. H., Goodall, E. A., Gates, S. N., Lander, G. C. & Martin, A. Substrate-engaged 26S proteasome structures reveal mechanisms for ATP-hydrolysis-driven translocation. Science 362, (2018).
Chu, B. W. et al. The E3 ubiquitin ligase UBE3C enhances proteasome processivity by ubiquitinating partially proteolyzed substrates. J. Biol. Chem. 288, 34575–34587 (2013).
Gottlieb, C. D., Thompson, A. C. S., Ordureau, A., Harper, J. W. & Kopito, R. R. Acute unfolding of a single protein immediately stimulates recruitment of ubiquitin protein ligase E3C (UBE3C) to 26S proteasomes. J. Biol. Chem. 294, 16511–16524 (2019).
Ding, Z. et al. Structural Snapshots of 26S Proteasome Reveal Tetraubiquitin-Induced Conformations. Mol. Cell 73, e6 (2019).
Prakash, S., Tian, L., Ratliff, K. S., Lehotzky, R. E. & Matouschek, A. An unstructured initiation site is required for efficient proteasome-mediated degradation. Nat. Struct. Mol. Biol. 11, 830–837 (2004).
Erales, J. & Coffino, P. Ubiquitin-independent proteasomal degradation. Biochim. Biophys. Acta 1843, 216–221 (2014).
Bashore, C. et al. Ubp6 deubiquitinase controls conformational dynamics and substrate degradation of the 26S proteasome. Nat. Struct. Mol. Biol. 22, 712–719 (2015).
Janse, D. M., Crosas, B., Finley, D. & Church, G. M. Localization to the proteasome is sufficient for degradation. J. Biol. Chem. 279, 21415–21420 (2004).
Balzarini, M. et al. Chemically Induced Degradation of Native Proteins by Direct Recruitment to the 26S Proteasome. bioRxiv https://doi.org/10.1101/2023.07.19.549534 (2023). 2023.07.19.549534.
Ma, Y., Xu, J., Huang, P., Bai, X. & Gao, H. Ubiquitin-independent, Proteasome-mediated targeted degradation of KRAS in pancreatic adenocarcinoma cells using an engineered ornithine decarboxylase/antizyme system. IUBMB Life 71, 57–65 (2019).
Husnjak, K. et al. Proteasome subunit Rpn13 is a novel ubiquitin receptor. Nature 453, 481–488 (2008).
Testa, A., Hughes, S., Butcher, S. P. & Ciulli, Alessio. Preparation of bifunctional molecules for targeting Usp14. PCT Int. Appl. 279pp. (2019).
Bashore, C. et al. Targeted degradation via direct 26S proteasome recruitment. Nat. Chem. Biol. 19, 55–63 (2023).
Liu, X. et al. IMPDH inhibition activates TLR-VCAM1 pathway and suppresses the development of MLL-fusion leukemia. EMBO Mol. Med. 15, e15631 (2023).
Tzoneva, G. et al. Clonal evolution mechanisms in NT5C2 mutant-relapsed acute lymphoblastic leukaemia. Nature 553, 511–514 (2018).
Li, H.-X. et al. IMPDH2 mediate radioresistance and chemoresistance in osteosarcoma cells. Eur. Rev. Med. Pharmacol. Sci. 18, 3038–3044 (2014).
He, Y. et al. Over-expression of IMPDH2 is associated with tumor progression and poor prognosis in hepatocellular carcinoma. Am. J. Cancer Res. 8, 1604–1614 (2018).
Tian, Y., Zhang, J., Chen, L. & Zhang, X. The expression and prognostic role of IMPDH2 in ovarian cancer. Ann. Diagn. Pathol. 46, 151511 (2020).
Sohbati, H., Amini, M. & Balalaie, S. Synthesis and Biological Evaluation of Novel Anti-leukemia Proteolysis-Targeting Chimeras in Degradating Inosine Monophosphate Dehydrogenase. Iran. J. Pharm. Res. 21, e129251 (2022).
Chung, L. H., Liu, D., Liu, X. T. & Qi, Y. Ceramide Transfer Protein (CERT): An Overlooked Molecular Player in Cancer. Int. J. Mol. Sci. 22, (2021).
Swanton, C. et al. Regulators of mitotic arrest and ceramide metabolism are determinants of sensitivity to paclitaxel and other chemotherapeutic drugs. Cancer Cell 11, 498–512 (2007).
Sun, T. et al. Targeted Degradation of Histone Deacetylases via Bypassing E3 Ligase Targeting Chimeras (BYETACs). ACS Med. Chem. Lett. 16, 1155–1162 (2025).
Sintchak, M. D. & Nimmesgern, E. The structure of inosine 5’-monophosphate dehydrogenase and the design of novel inhibitors. Immunopharmacology 47, 163–184 (2000).
Kudo, N. et al. Crystal structures of the CERT START domain with inhibitors provide insights into the mechanism of ceramide transfer. J. Mol. Biol. 396, 245–251 (2010).
Weng, G., Li, D., Kang, Y. & Hou, T. Integrative Modeling of PROTAC-Mediated Ternary Complexes. J. Med. Chem. 64, 16271–16281 (2021).
Huang, X., Luan, B., Wu, J. & Shi, Y. An atomic structure of the human 26S proteasome. Nat. Struct. Mol. Biol. 23, 778–785 (2016).
Chen, K. et al. Differential Sensitivities of Fast- and Slow-Cycling Cancer Cells to Inosine Monophosphate Dehydrogenase 2 Inhibition by Mycophenolic Acid. Molecular Medicine 21, 792–802 (2015).
Chiarelli, L. R. et al. Inosine monophosphate dehydrogenase variability in renal transplant patients on long-term mycophenolate mofetil therapy. Br. J. Clin. Pharmacol. 69, 38–50 (2010).
Jain, J. et al. Regulation of inosine monophosphate dehydrogenase type I and type II isoforms in human lymphocytes. Biochem. Pharmacol. 67, 767–776 (2004).
Sanquer, S. et al. Induction of inosine monophosphate dehydrogenase activity after long-term treatment with mycophenolate mofetil. Clin. Pharmacol. Ther. 65, 640–648 (1999).
Zhou, W. et al. Purine metabolism regulates DNA repair and therapy resistance in glioblastoma. Nat. Commun. 11, 3811 (2020).
Liu, C.-J. et al. The Role of Purine Metabolism-Related Genes PPAT and IMPDH1 in the Carcinogenesis of Intrahepatic Cholangiocarcinoma Based on Metabonomic and Bioinformatic Analyses. J. Oncol. 2023, 5141836 (2023).
Huang, Y. et al. Wnt/β-catenin signalling activates IMPDH2-mediated purine metabolism to facilitate oxaliplatin resistance by inhibiting caspase-dependent apoptosis in colorectal cancer. J. Transl. Med. 22, 133 (2024).
Mamrosh, J. L. et al. Quantitative measurement of the requirement of diverse protein degradation pathways in MHC class I peptide presentation. Sci. Adv. 9, eade7890 (2024).
Borodovsky, A. et al. A novel active site-directed probe specific for deubiquitylating enzymes reveals proteasome association of USP14. EMBO J 20, 5187–5196 (2001).
Koulich, E., Li, X. & DeMartino, G. N. Relative Structural and Functional Roles of Multiple Deubiquitylating Proteins Associated with Mammalian 26S Proteasome. Mol. Biol. Cell 19, 1072–1082 (2007).
Hu, M. et al. Structure and mechanisms of the proteasome-associated deubiquitinating enzyme USP14. EMBO J 24, 3747–3756 (2005).
Leggett, D. S. et al. Multiple associated proteins regulate proteasome structure and function. Mol. Cell 10, 495–507 (2002).
Crimmins, S. et al. Transgenic rescue of ataxia mice with neuronal-specific expression of ubiquitin-specific protease 14. J. Neurosci. 26, 11423–11431 (2006).
Catic, A. et al. Screen for ISG15-crossreactive deubiquitinases. PLoS One 2, e679 (2007).
Ali, E. M. H., Loy, C. A. & Trader, D. J. ByeTAC: Bypassing E-Ligase-Targeting Chimeras for Direct Proteasome Degradation. J. Med. Chem. 68, 9694–9705 (2025).
Pack, C.-G. et al. Quantitative live-cell imaging reveals spatio-temporal dynamics and cytoplasmic assembly of the 26S proteasome. Nat. Commun. 5, 3396 (2014).
Kim, E. et al. Dual Function of USP14 Deubiquitinase in Cellular Proteasomal Activity and Autophagic Flux. Cell Rep 24, 732–743 (2018).
Xu, D. et al. USP14 regulates autophagy by suppressing K63 ubiquitination of Beclin 1. Genes Dev 30, 1718–1730 (2016).
Hajj, C., Becker, A. & Haimovitz-Friedman, A. Novel mechanisms of action of classical chemotherapeutic agents on sphingolipid pathways. Biol. Chem. 396, 669–679 (2015).
Lee, A. J. X. et al. CERT depletion predicts chemotherapy benefit and mediates cytotoxic and polyploid-specific cancer cell death through autophagy induction. Journal of Pathology 226, 482–494 (2012).
Juul, N. et al. Assessment of an RNA interference screen-derived mitotic and ceramide pathway metagene as a predictor of response to neoadjuvant paclitaxel for primary triple-negative breast cancer: A retrospective analysis of five clinical trials. Lancet Oncol 11, 358–365 (2010).
Schrödinger. Release 2022-4. Schrödinger, LLC. New York (2022).
Schrödinger Release 2022-4. Maestro, version 13.4. Schrödinger, LLC. New York (2022).
Schrödinger Release 2022-4. MacroModel, version 13.8. Schrödinger, LLC. New York (2022).
Lu, C. et al. OPLS4: Improving force field accuracy on challenging regimes of chemical space. J. Chem. Theory Comput. 17, 4291–4300 (2021).
Still, W. C., Tempczyk, A., Hawley, R. C. & Hendrickson, T. Semianalytical treatment of solvation for molecular mechanics and dynamics. J. Am. Chem. Soc. 112, 6127–6129 (1990).
Schrödinger Release 2022-4. Protein Preparation Wizard 2022-4. Schrödinger, LLC. New York (2022).
Schrödinger Release 2022-4. Glide, version 9.7. Schrödinger, LLC. New York (2022).
Friesner, R. A. et al. Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem 47, 1739–1749 (2004).
Halgren, T. A. et al. Glide: a new approach for rapid, accurate docking and scoring. 2. Enrichment factors in database screening. J Med Chem 47, 1750–1759 (2004).
Schrödinger. Release 2022-4: Induced Fit Docking Protocol. Schrödinger, LLC. New York (2022).
Sherman, W., Day, T., Jacobson, M. P., Friesner, R. A. & Farid, R. Novel procedure for modeling ligand/receptor induced fit effects. J Med Chem 49, 534–553 (2006).
Johnson, M. C. & Kollman, J. M. Cryo-EM structures demonstrate human IMPDH2 filament assembly tunes allosteric regulation. Elife 9, (2020).
Bowers, K. J. et al. Scalable Algorithms for Molecular Dynamics Simulations on Commodity Clusters. in Proceedings of the ACM/IEEE Conference on Supercomputing (SC06) (Tampa, Florida, 2006).
Schrödinger. Release 2022-4: Desmond Molecular Dynamics System. D. E. Shaw Research. New York (2022).
Schrödinger. Release 2022-4 : Maestro-Desmond Interoperability Tools. D. E. Shaw Research. Schrodinger, New York (2021).
Martyna, G. J., Klein, M. L. & Tuckerman, M. Nosé–Hoover chains: The canonical ensemble via continuous dynamics. J. Chem. Phys. 97, 2635–2643 (1992).
Martyna, G. J., Tobias, D. J. & Klein, M. L. Constant pressure molecular dynamics algorithms. J. Chem. Phys. 101, 4177–4189 (1994).
Tuckerman, M., Berne, B. J. & Martyna, G. J. Reversible multiple time scale molecular dynamics. J. Chem. Phys. 97, 1990–2001 (1992).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: An N⋅log(N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089–10092 (1993).
Essmann, U. et al. A smooth particle mesh Ewald method. J. Chem. Phys. 103, 8577–8593 (1995).
Kräutler, V., van Gunsteren, W. F. & Hünenberger, P. H. A fast SHAKE algorithm to solve distance constraint equations for small molecules in molecular dynamics simulations. J. Comput. Chem. 22, 501–508 (2001).
Ramírez-Aportela, E., López-Blanco, J. R. & Chacón, P. FRODOCK 2.0: Fast protein-protein docking server. Bioinformatics 32, 2386–2388 (2016).
Garzon, J. I. et al. FRODOCK: A new approach for fast rotational protein-protein docking. Bioinformatics 25, 2544–2551 (2009).
Gray, J. J. et al. Protein-protein docking with simultaneous optimization of rigid-body displacement and side-chain conformations. J. Mol. Biol. 331, 281–299 (2003).
RDKit: Open-source cheminformatics. https://www.rdkit.org
O’Boyle, N. M. et al. Open Babel: An open chemical toolbox. J. Cheminform. 3, (2011).
Trott, O. & Olson, A. J. AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31, 455–461 (2010).
Olechnovič, K. & Venclovas, Č VoroMQA: Assessment of protein structure quality using interatomic contact areas. Proteins: Structure, Function and Bioinformatics 85, 1131–1145 (2017).
Olechnovič, K. & Venclovas, C. D. S. VoroMQA web server for assessing three-dimensional structures of proteins and protein complexes. Nucleic Acids Res 47, W437–W442 (2019).
Rodrigues, J. P. G. L. M. et al. Clustering biomolecular complexes by residue contacts similarity. Proteins: Structure, Function and Bioinformatics 80, 1810–1817 (2012).
Vangone, A., Spinelli, R., Scarano, V., Cavallo, L. & Oliva, R. COCOMAPS: A web application to analyze and visualize contacts at the interface of biomolecular complexes. Bioinformatics 27, 2915–2916 (2011).
Sintchak, M. D. et al. Structure and mechanism of inosine monophosphate dehydrogenase in complex with the immunosuppressant mycophenolic acid. Cell 85, 921–930 (1996).
Acknowledgements
We thank Dr. Daniel Reynes (Emory University School of Medicine) for IMPDH2 cDNA, and Dr. Scott Wilson (U. Alabama) and Dr. Min Jae Lee for Usp14‾/‾ MEFs cells, Dr. Marta Taulés (Molecular interaction analysis Technology, CCiTUB) for SPR analysis, Carles Bonet (Genomics and Transcriptomics core service, BMB) for qPCR analysis, Dr. Lluís Ribas (IRB-Barcelona) for lentiviral vectors, and Dr. Daniel Finley, Dr. Cristina Mayor-Ruiz and Dr. Jordi Casanova for their help with reagents. The authors are thankful to Dr. Adolfo Ferrando and Dr. Clara Reglero (Columbia U.) for insightful ideas, and to Dr. Gemma Triola and Dr. Ignacio Alfonso for their critical reading of the manuscript. The authors thankfully acknowledge the use of the computational resources of the Consorci de Serveis Universitaris de Catalunya (CSUC) and of the Centro de Supercomputación de Galicia (CESGA). We thank Albaha University for their funding to A.A. This work was supported with grants by Spanish Ministry of Science and Innovation (PID2020-113813RB-I00 and PID2023-146670OB-C21) and the European Commission – NextGenerationEU (Regulation EU 2020/2094), through CSIC's Global Health Platform (PTI Salud Global) and Generalitat de Catalunya (2021-SGR-00504).
Author information
Authors and Affiliations
Contributions
MC, TR, AZ, NGS, AGA and BCM performed the biochemical and biological experiments. HC, JLA, and AA synthesized the compounds. PFN generated and provided cell lines. JB performed the computational analysis. JC, AD, JB, GF, and BC designed the research. JB, GF and BC wrote the manuscript with contributions of all authors.
Corresponding authors
Ethics declarations
Competing interests
JC, JB, GF, BC, JLA, and AD are named as inventors on a patent application (PCT/EP2025/056573) claiming the use of the described 26S-UIDs for use for the treatment of cancer related to CERT1, filed by the Spanish National Research Council. The other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Casasampere, M., Carneros, H., Roda, T. et al. Expanding the targeted protein degradation approach with small molecule chimeras directed to the 26S proteasome. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71132-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71132-5