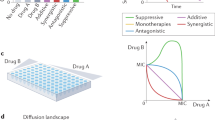
Abstract
Antibiotic combination in time and space is a key strategy to combat antimicrobial resistance. The success of such treatment designs requires their robust efficacy across treatment conditions and a pathogen’s genomic diversity. This study found that an initial treatment with a β-lactam antibiotic causes robust cellular sensitization towards an aminoglycoside antibiotic across the high-risk human pathogen Pseudomonas aeruginosa, including resistant strains. This phenomenon of cellular sensitization, termed negative hysteresis, is modulated by the Cpx envelope stress response system and linked to membrane stress during growth. The increase in efficacy is achieved through a β-lactam induced elevated cellular uptake of the subsequently administered aminoglycoside. Negative hysteresis and the Cpx system are linked in several cases to the expression of synergistic drug interactions, thus enhancing efficacy of antibiotic combinations. Overall, our study identifies the phenomenon of negative hysteresis as a robustly inducible phenotype and thus a unique focus for optimizing antimicrobial therapy.
Similar content being viewed by others
Data availability
The source data of this study is provided in a supplementary Source Data file. The statistical data generated, as well as Key Resources used, are provided in the Supplementary Data files and as Supplementary Tables. The transcriptomic data generated in this study have been deposited in the NCBI’s Gene Expression Omnibus under GEO Series accession number GSE290299. Source data are provided with this paper.
References
De Oliveira, D. M. P. et al. Antimicrobial Resistance in ESKAPE Pathogens. Clin. Microbiol. Rev. 33, e00181–19 (2020). /cmr/33/3/CMR.00181-19.atom.
Murray, C.J. et al. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. https://doi.org/10.1016/S0140-6736(21)02724-0. (2022).
Naghavi, M. et al. Global burden of bacterial antimicrobial resistance 1990–2021: a systematic analysis with forecasts to 2050. Lancet 0, 1199–1226 (2024).
Pang, Z., Raudonis, R., Glick, B. R., Lin, T.-J. & Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 37, 177–192 (2019).
Tueffers, L. et al. Pseudomonas aeruginosa populations in the cystic fibrosis lung lose susceptibility to newly applied β-lactams within 3 days. J. Antimicrob. Chemother. 74, 2916–2925 (2019).
Reynolds, D. & Kollef, M. The Epidemiology and Pathogenesis and Treatment of Pseudomonas aeruginosa Infections: An Update. Drugs 81, 2117–2131 (2021).
WHO Bacterial Priority Pathogens List 2024: Bacterial Pathogens of Public Health Importance, to Guide Research, Development, and Strategies to Prevent and Control Antimicrobial Resistance, 1st ed (World Health Organization, 2024).
Roemhild, R. & Schulenburg, H. Evolutionary ecology meets the antibiotic crisis: Can we control pathogen adaptation through sequential therapy?. Evol. Med. Public Health 2019, 37–45 (2019).
Baym, M., Stone, L. K. & Kishony, R. Multidrug evolutionary strategies to reverse antibiotic resistance. Science 351, aad3292 (2016).
Merker, M. et al. Evolutionary Approaches to Combat Antibiotic Resistance: Opportunities and Challenges for Precision Medicine. Front. Immunol. 11, 1938 (2020).
Andersson, D. I. et al. Antibiotic resistance: turning evolutionary principles into clinical reality. FEMS Microbiol. Rev. 44, 171–188 (2020).
Roemhild, R. et al. Cellular hysteresis as a principle to maximize the efficacy of antibiotic therapy. Proc. Natl. Acad. Sci. Usa. 115, 9767–9772 (2018).
Batra, A. et al. High potency of sequential therapy with only β-lactam antibiotics. eLife 10, e68876 (2021).
Roemhild, R., Barbosa, C., Beardmore, R. E., Jansen, G. & Schulenburg, H. Temporal variation in antibiotic environments slows down resistance evolution in pathogenic Pseudomonas aeruginosa. Evol. Appl. 8, 945–955 (2015).
Fernández-Billón, M. et al. Collateral susceptibility-guided alternation of ceftolozane/tazobactam with imipenem prevents resistance development in XDR Pseudomonas aeruginosa biofilms. Biofilm 8, 100231 (2024).
Hernando-Amado, S. et al. Rapid Phenotypic Convergence towards Collateral Sensitivity in Clinical Isolates of Pseudomonas aeruginosa Presenting Different Genomic Backgrounds. Microbiol. Spectr. 11, e02276–22 (2022).
Hernando-Amado, S., Laborda, P., Valverde, J. R. & Martínez, J. L. Mutational background influences P. aeruginosa ciprofloxacin resistance evolution but preserves collateral sensitivity robustness. Proc. Natl. Acad. Sci. 119, e2109370119 (2022).
Hernando-Amado, S., Sanz-García, F. & Martínez, J. L. Rapid and robust evolution of collateral sensitivity in Pseudomonas aeruginosa antibiotic-resistant mutants. Sci. Adv. 6, eaba5493 (2020).
Roemhild, R., Bollenbach, T. & Andersson, D. I. The physiology and genetics of bacterial responses to antibiotic combinations. Nat. Rev. Microbiol. 20, 478–490 (2022).
Plotz, P. H. & Davis, B. D. Synergism between Streptomycin and Penicillin: A Proposed Mechanism. Science 135, 1067–1068 (1962).
Sullivan, G. J., Delgado, N. N., Maharjan, R. & Cain, A. K. How antibiotics work together: molecular mechanisms behind combination therapy. Curr. Opin. Microbiol. 57, 31–40 (2020).
Gil-Gil, T. & Berryhill, B.A. Antibiotic killing of drug-induced bacteriostatic cells. Antimicrob. Agents Chemother. 69, e00156–25 (2025).
Lew, C. et al. Proteomic Correlates of Enhanced Daptomycin Activity following β-Lactam Preconditioning in Daptomycin-Resistant, Methicillin-Resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 66, e0201721 (2022).
Chen, Z. et al. Enhanced efficacy of sequential administration of fosfomycin and linezolid against methicillin-resistant Staphylococcus aureus. Front. Microbiol. 16, 1511707 (2025).
Botelho, J. et al. Phylogroup-specific variation shapes the clustering of antimicrobial resistance genes and defence systems across regions of genome plasticity in Pseudomonas aeruginosa. eBioMedicine 90, 104532 (2023).
Hilker, R. et al. Interclonal gradient of virulence in the Pseudomonas aeruginosa pangenome from disease and environment. Environ. Microbiol. 17, 29–46 (2015).
Tueffers, L. et al. Variation in the response to antibiotics and life-history across the major Pseudomonas aeruginosa clone type (mPact) panel. Microbiol. Spectr. 0, e00143–24 (2024).
Barbosa, C. et al. Alternative Evolutionary Paths to Bacterial Antibiotic Resistance Cause Distinct Collateral Effects. Mol. Biol. Evol. 34, 2229–2244 (2017).
MacKenzie, F. M. & Gould, I. M. The post-antibiotic effect. J. Antimicrob. Chemother. 32, 519–537 (1993).
Rahme, L. G., Stevens, E. J., Wolfort, S. F. & Shao, J. Common Virulence Factors for Bacterial Pathogenicity in Plants and Animals. 268, 5 (1995).
Holloway, B. W. Genetic Recombination in Pseudomonas aeruginosa. Microbiology 13, 572–581 (1955).
Römling, U. et al. Epidemiology Of Chronic Pseudomonas aeruginosa Infections In Cystic Fibrosis. J. Infect. Dis. 170, 1616–1621 (1994).
Parkins, M.D., Somayaji, R., & Waters, V. J. Epidemiology, Biology, and Impact of Clonal Pseudomonas aeruginosa Infections in Cystic Fibrosis. Clin. Microbiol. Rev. 31, https://doi.org/10.1128/cmr.00019-18 (2018).
Mitchell, A. M. & Silhavy, T. J. Envelope stress responses: balancing damage repair and toxicity. Nat. Rev. Microbiol. 17, 417–428 (2019).
Raivio, T. L. & Silhavy, T. J. Transduction of envelope stress in Escherichia coli by the Cpx two-component system. J. Bacteriol. 179, 7724–7733 (1997).
Raivio, T. L. Everything old is new again: An update on current research on the Cpx envelope stress response. Biochim. Biophys. Acta BBA - Mol. Cell Res. 1843, 1529–1541 (2014).
Allen, R.C., Pfrunder-Cardozo, K. R. & Hall, A.R. Collateral Sensitivity Interactions between Antibiotics Depend on Local Abiotic Conditions. 6, 15 (2021).
Lázár, V. et al. Bacterial evolution of antibiotic hypersensitivity. Mol. Syst. Biol. 9, 700 (2013).
Mahoney, T. F. & Silhavy, T. J. The Cpx Stress Response Confers Resistance to Some, but Not All, Bactericidal Antibiotics. J. Bacteriol. 195, 1869–1874 (2013).
Hews, C.L., Cho, T., Rowley, G. & Raivio, T. L. Maintaining Integrity Under Stress: Envelope Stress Response Regulation of Pathogenesis in Gram-Negative Bacteria. Front. Cell. Infect. Microbiol. 9, 313 (2019).
Chan, D. C. K. et al. Nutrient Limitation Sensitizes Pseudomonas aeruginosa to Vancomycin. ACS Infect. Dis. 9, 1408–1423 (2023).
Galdino, A. C. M. et al. Siderophores promote cooperative interspecies and intraspecies cross-protection against antibiotics in vitro. Nat. Microbiol. 9, 631–646 (2024).
Tian, Z.-X. & Wang, Y.-P. Identification of cpxS mutational resistome in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 0, e00921–e00923 (2023).
O’Malley, M. R. et al. The Pseudomonas aeruginosa Cpx system provides a cyclic-di-GMP independent link between cell envelope stress and surface sensing. mBio 0, e02726–25 (2025).
Emami, K. et al. Regulation of the Xylan-degrading Apparatus of Cellvibrio japonicus by a Novel Two-component System. J. Biol. Chem. 284, 1086–1096 (2009).
Tian, Z.-X., Yi, X.-X., Cho, A., O’Gara, F. & Wang, Y.-P. CpxR Activates MexAB-OprM Efflux Pump Expression and Enhances Antibiotic Resistance in Both Laboratory and Clinical nalB-Type Isolates of Pseudomonas aeruginosa. PLOS Pathog. 12, e1005932 (2016).
DiGiuseppe, P. A. & Silhavy, T. J. Signal Detection and Target Gene Induction by the CpxRA Two-Component System. J. Bacteriol. 185, 2432–2440 (2003).
Price, N. L. & Raivio, T. L. Characterization of the Cpx Regulon in Escherichia coli Strain MC4100. J. Bacteriol. 191, 1798–1815 (2009).
Shigapova, N. et al. Membrane fluidization triggers membrane remodeling which affects the thermotolerance in Escherichia coli. Biochem. Biophys. Res. Commun. 328, 1216–1223 (2005).
Parasassi, T., De Stasio, G., d’Ubaldo, A. & Gratton, E. Phase fluctuation in phospholipid membranes revealed by Laurdan fluorescence. Biophys. J. 57, 1179–1186 (1990).
K. Scheinpflug, O. Krylova, H. Strahl, “Measurement of Cell Membrane Fluidity by Laurdan GP: Fluorescence Spectroscopy and Microscopy” in Antibiotics: Methods and Protocols, P. Sass, Ed. (Springer, 2017), pp. 159–174.
Krause, K. M., Serio, A. W., Kane, T. R. & Connolly, L. E. Aminoglycosides: An Overview. Cold Spring Harb. Perspect. Med. 6, a027029 (2016).
Lang, M., Carvalho, A., Baharoglu, Z. & Mazel, D. Aminoglycoside uptake, stress, and potentiation in Gram-negative bacteria: new therapies with old molecules. Microbiol. Mol. Biol. Rev. 87, e00036–22 (2023).
Kasianowicz, J., Benz, R. & McLaughlin, S. The kinetic mechanism by which CCCP (carbonyl cyanidem-Chlorophenylhydrazone) transports protons across membranes. J. Membr. Biol. 82, 179–190 (1984).
Bush, K. & Bradford, P. A. β-Lactams and β-Lactamase Inhibitors: An Overview. Cold Spring Harb. Perspect. Med. 6, a025247 (2016).
Govers, S. K., Mortier, J., Adam, A. & Aertsen, A. Protein aggregates encode epigenetic memory of stressful encounters in individual Escherichia coli cells. PLOS Biol. 16, e2003853 (2018).
Lambert, G. & Kussell, E. Memory and fitness optimization of bacteria under fluctuating environments. PLoS Genet 10, e1004556 (2014).
Cho, S.-H., Dekoninck, K. & Collet, J.-F. Envelope-Stress Sensing Mechanism of Rcs and Cpx Signaling Pathways in Gram-Negative Bacteria. J. Microbiol. 61, 317–329 (2023).
Evans, K. L., Kannan, S., Li, G., de Pedro, M. A. & Young, K. D. Eliminating a Set of Four Penicillin Binding Proteins Triggers the Rcs Phosphorelay and Cpx Stress Responses in Escherichia coli. J. Bacteriol. 195, 4415–4424 (2013).
Masi, M., Pinet, E. & Pagès, J.-M. Complex Response of the CpxAR Two-Component System to β-Lactams on Antibiotic Resistance and Envelope Homeostasis in Enterobacteriaceae. Antimicrob. Agents Chemother. 64, e00291–20 (2020).
Guest, R. L. & Raivio, T. L. Role of the Gram-Negative Envelope Stress Response in the Presence of Antimicrobial Agents. Trends Microbiol 24, 377–390 (2016).
Mechaly, A. E., Sassoon, N., Betton, J.-M. & Alzari, P. M. Segmental Helical Motions and Dynamical Asymmetry Modulate Histidine Kinase Autophosphorylation. PLOS Biol. 12, e1001776 (2014).
Lv, B. et al. Heat shock potentiates aminoglycosides against gram-negative bacteria by enhancing antibiotic uptake, protein aggregation, and ROS. Proc. Natl. Acad. Sci. 120, e2217254120 (2023).
Yen, P. & Papin, J. A. History of antibiotic adaptation influences microbial evolutionary dynamics during subsequent treatment. PLOS Biol. 15, e2001586 (2017).
Jorth, P. et al. Evolved Aztreonam Resistance Is Multifactorial and Can Produce Hypervirulence in Pseudomonas aeruginosa. mBio 8, e00517-17 (2017).
Chowdhury, P. R. oy et al. Genomic islands 1 and 2 play key roles in the evolution of extensively drug-resistant ST235 isolates of Pseudomonas aeruginosa. Open Biol. 6, 150175 (2016).
Barbosa, C. et al. The Genomic Basis of Rapid Adaptation to Antibiotic Combination Therapy in Pseudomonas aeruginosa. Mol. Biol. Evol. 38, 449–464 (2021).
Gomis-Font, M. A., Sastre-Femenia, M. À, Taltavull, B., Cabot, G. & Oliver, A. In vitro dynamics and mechanisms of cefiderocol resistance development in wild-type, mutator and XDR Pseudomonas aeruginosa. J. Antimicrob. Chemother. 78, 1785–1794 (2023).
Hernando-Amado, S., Gomis-Font, M. A., Valverde, J. R., Oliver, A. & Martínez, J. L. Ceftazidime-avibactam use selects multidrug-resistance and prevents designing collateral sensitivity-based therapies against Pseudomonas aeruginosa. Nat. Commun. 16, 3323 (2025).
Beckmann, C. et al. Use of Phage Display To Identify Potential Pseudomonas aeruginosa Gene Products Relevant to Early Cystic Fibrosis Airway Infections. Infect. Immun. 73, 444–452 (2005).
A. Oliver, J. Arca-Suárez, M. A. Gomis-Font, L. González-Pinto, C. López-Causapé, Emerging resistance mechanisms to newer β-lactams in Pseudomonas aeruginosa. Clin. Microbiol. Infect. S1198743X25001314 (2025). https://doi.org/10.1016/j.cmi.2025.03.013.
N. K. Boyd, C. Teng, C. R. Frei, Brief Overview of Approaches and Challenges in New Antibiotic Development: A Focus On Drug Repurposing. Front. Cell. Infect. Microbiol. 11, 684515 (2021).
Hernando-Amado, S., Laborda, P. & Martínez, J. L. Tackling antibiotic resistance by inducing transient and robust collateral sensitivity. Nat. Commun. 14, 1723 (2023).
Lew, C., Mishra, N. N., Bayer, A. S. & Rose, W. E. β-Lactam-Induced Cell Envelope Adaptations, Not Solely Enhanced Daptomycin Binding, Underlie Daptomycin-β-Lactam Synergy in Methicillin-Resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 65, e00356-21 (2021).
Guggenbichler, J. P., Allerberger, F., Dierich, M. P., Schmitzberger, R. & Semenitz, E. SPACED ADMINISTRATION OF ANTIBIOTIC COMBINATIONS TO ELIMINATE PSEUDOMONAS FROM SPUTUM IN CYSTIC FIBROSIS. Lancet 332, 749–750 (1988).
(EUCAST) European Committee for Antimicrobial Susceptibility Testing, Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution. Clin. Microbiol. Infect. 9, ix–xv (2003).
Nelder, J. A. & Mead, R. A Simplex Method for Function Minimization. Comput. J. 7, 308–313 (1965).
Fatsis-Kavalopoulos, N., Roemhild, R., Tang, P.-C., Kreuger, J. & Andersson, D. I. CombiANT: Antibiotic interaction testing made easy. PLOS Biol. 18, e3000856 (2020).
Fatsis-Kavalopoulos, N., Roelofs, L. & Andersson, D. I. Potential risks of treating bacterial infections with a combination of β-lactam and aminoglycoside antibiotics: A systematic quantification of antibiotic interactions in E. coli blood stream infection isolates. eBioMedicine 78, 103979 (2022).
Tang, P.-C., Sánchez-Hevia, D. L., Westhoff, S., Fatsis-Kavalopoulos, N. & Andersson, D. I. Within-species variability of antibiotic interactions in Gram-negative bacteria. mBio 15, e00196–24 (2024).
N. Fatsis-Kavalopoulos, D. L. Sánchez-Hevia, D. I. Andersson, Beyond the FIC index: the extended information from fractional inhibitory concentrations (FICs). J. Antimicrob. Chemother. dkae233 (2024). https://doi.org/10.1093/jac/dkae233.
Barbosa, C., Beardmore, R., Schulenburg, H. & Jansen, G. Antibiotic combination efficacy (ACE) networks for a Pseudomonas aeruginosa model. PLOS Biol. 16, e2004356 (2018).
Hegreness, M., Shoresh, N., Damian, D., Hartl, D. & Kishony, R. Accelerated evolution of resistance in multidrug environments. Proc. Natl. Acad. Sci. Usa. 105, 13977–13981 (2008).
Geourjon, C. & Deléage, G. SOPMA: significant improvements in protein secondary structure prediction by consensus prediction from multiple alignments. Bioinformatics 11, 681–684 (1995).
Schwede, T., Kopp, J., Guex, N. & Peitsch, M. C. SWISS-MODEL: an automated protein homology-modeling server. Nucleic Acids Res 31, 3381–3385 (2003).
Söding, J., Biegert, A. & Lupas, A. N. The HHpred interactive server for protein homology detection and structure prediction. Nucleic Acids Res 33, W244–W248 (2005).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D. Biol. Crystallogr. 60, 2126–2132 (2004).
Schrödinger, L. L. C., The PyMOL Molecular Graphics System, Version 1.8. (2015).
Hmelo, L. R. et al. Precision-engineering the Pseudomonas aeruginosa genome with two-step allelic exchange. Nat. Protoc. 10, 1820–1841 (2015).
Obeng, N. et al. Bacterial c-di-GMP has a key role in establishing host–microbe symbiosis. Nat. Microbiol. 8, 1809–1819 (2023).
Heeb, S., Blumer, C. & Haas, D. Regulatory RNA as mediator in GacA/RsmA-dependent global control of exoproduct formation in Pseudomonas fluorescens CHA0. J. Bacteriol. 184, 1046–1056 (2002).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinforma. Oxf. Engl. 30, 2114–2120 (2014).
Langmead, B., Wilks, C., Antonescu, V. & Charles, R. Scaling read aligners to hundreds of threads on general-purpose processors. Bioinforma. Oxf. Engl. 35, 421–432 (2019).
Magoc, T., Wood, D. & Salzberg, S. L. EDGE-pro: Estimated Degree of Gene Expression in Prokaryotic Genomes. Evol. Bioinforma. Online 9, 127–136 (2013).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Winsor, G. L. et al. Enhanced annotations and features for comparing thousands of Pseudomonas genomes in the Pseudomonas genome database. Nucleic Acids Res 44, D646–D653 (2016).
Acknowledgements
We are very grateful to S. Hernando-Amado (Madrid, Spain), C. Pál (Szeged, Hungary), and T. Bollenbach (Cologne, Germany) for critical comments and advice on the manuscript. We further thank D. Rogers, J. Summers (Ploen, Germany) for guidance in allelic exchange, P. Rainey (Ploen, Germany) for providing the plasmids and strains, then J. Lorenzen, K. Flinder, N. Steinbach, S. Butze (all Schulenburg lab), and L. Kirchhoff (Rupp lab) for supporting the experimental work, and also the Rupp and Schulenburg groups for general feedback. We are grateful for financial support from the German Research Foundation within the Research and Training Group 2501 (RTG 2501) on Translational Evolutionary Research (project 4.2 to H.S.), within the Excellence cluster Precision Medicine in chronic Inflammation (PMI; funding under Germany’s Excellence Strategy EXC 2167-390884018, to B.K., K.R., J.R., H.S.), within the Clinician Scientist Program in Evolutionary Medicine (CSEM) – project number 413490537 (to EEG), and as part of the individual grants SCHU 1415/12-2 (to H.S.) and BR-2915/7-1 (to M.B.). We are grateful for financial support from the Swedish Research Council, project number 2021-02091 (to D.I.A.). We are also grateful for financial support from the Max-Planck Society (Fellowship to H.S.), the Leibniz Association within the Leibniz Science-Campus Evolutionary Medicine of the Lung (EvoLUNG, to H.S.), and the project SKILLED funded by the DAMP foundation (to J.R., H.S.). This work was also supported by the ZMB Young Scientist award and the FWF grant 10.55776/ESP219 (to R.R.) and the TransEvo Innovation prize (to F.B.). The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization: F.B., L.U., L.T., E.E.G., J.R., D.I.A., H.So, M.B., R.R., H.Schu. Formal analysis: F.B., L.U., L.T., E.E.G., D.S., A.S.S., R.R.; Funding acquisition: F.B., K.F.R., J.R., R.R., H.Schu. Investigation and methodology: F.B., L.U., L.T., E.E.G., K.H., D.S., A.S.S., A.B., S.P., S.B., D.N., B.N.D., S.F., B.K., R.R.; Resources: E.E.G., B.K., K.F.R., J.R., H.Schu. Supervision: F.B., L.U., L.T., E.E.G., D.N., J.R., D.I.A., H.So, M.B., R.R., H.Schu. Writing of original draft: F.B., L.U., R.R., H.Schu. Review and editing: all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare the following competing interest: Access to the CombiANT technology was provided by Rx Dynamics AB as a product BETA test. R.R. and D.I.A. are co-founders of Rx Dynamics AB. The company had no influence on study design, investigation, data analysis, manuscript writing and decision to publish. Apart from this, the authors declare no conflict of interest.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Buchholz, F., Upterworth, L.M., Tueffers, L. et al. Robust antibiotic sensitization of pathogenic Pseudomonas aeruginosa via negative hysteresis in the cell envelope. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71178-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71178-5