Abstract
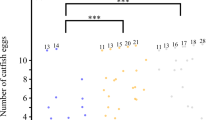
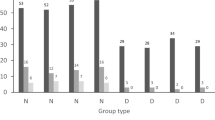
Interspecific brood parasitism has evolved repeatedly from parental care. However, many non-avian brood-parasitic lineages have ancestors lacking parental care, leaving the foundations of brood parasitism in these lineages enigmatic. We examine ecological, reproductive, and developmental data from the Lake Tanganyika radiation of Synodontis catfishes, where one species, the cuckoo catfish, exhibits brood parasitism of mouthbrooding cichlid fishes. Our comparative analyses, coupled with experimental parasitism, suggest that a combination of ancestral traits (large eggs and rapid embryo development) enabled the origin of brood parasitism. Evolutionary innovations then presumably enhanced the success of brood parasitism after it emerged. Novel traits comprise frequent production of small clutches to effectively utilize host availability, the evolution of egg mimicry to facilitate host egg adoption, and modifications to development to enhance the performance of catfish embryos in the host’s buccal cavity. Interestingly, despite the distinct ecological and life history contexts of the origin of catfish brood parasitism, its evolutionary and developmental outcomes align with those of canonical avian brood parasites. This suggests that general patterns are repeated in the evolution of brood parasitism, despite disparate starting conditions.
Similar content being viewed by others
Data availability
All trophic, reproductive, ontogenetic and experimental data generated in this study have been deposited in the Figshare database [https://doi.org/10.6084/m9.figshare.28646141] and are freely available. Source data are provided as a Source Data file. Raw ddRADseq reads have been deposited in NCBI’s Sequence Read Archive (https://www.ncbi.nlm.nih.gov/sra) (BioProject: PRJNA1132910) under accession numbers SAMN42360897–SAMN42360967. Source data are provided with this paper.
Code availability
The scripts to replicate all analyses reported in this study are available from Figshare [https://doi.org/10.6084/m9.figshare.28646141].
References
Davies, N. Cuckoos, Cowbirds and Other Cheats (AC Black, 2010).
Feeney, W. E. et al. Brood parasitism and the evolution of cooperative breeding in birds. Science 342, 1506–1508 (2013).
Feeney, W. E., Welbergen, J. A. & Langmore, N. E. Advances in the study of coevolution between avian brood parasites and their hosts. Annu. Rev. Ecol. Evol. Syst. 45, 227–246 (2014).
Kilner, R. M. & Langmore, N. E. Cuckoos versus hosts in insects and birds: adaptations, counter-adaptations and outcomes. Biol. Rev. 86, 836–852 (2011).
Medina, I. & Langmore, N. E. The evolution of acceptance and tolerance in hosts of avian brood parasites. Evolution of defences in hosts. Biol. Rev. 91, 569–577 (2016).
Thorogood, R., Spottiswoode, C. N., Portugal, S. J. & Gloag, R. The coevolutionary biology of brood parasitism: a call for integration. Philos. Trans. R. Soc. B Biol. Sci. 374, 20180190 (2019).
Lyon, B. E. & Eadie, J. McA. Conspecific brood parasitism in birds: a life-story perspective. Annu. Rev. Ecol. Evol. Syst. 39, 343–363 (2008).
Spottiswoode, C. N., Kilner, R. M. & Davies, N. B. Brood parasitism. In The Evolution of Parental Care 226–356. (Oxford University Press, Oxford, 2012).
Sless, T. J. L., Danforth, B. N. & Searle, J. B. Evolutionary origins and patterns of diversification in animal brood parasitism. Am. Nat. 202, 107–121 (2023).
Skelton, P. Freshwater Fishes of Southern Africa (Struik Nature, 2024).
Sato, T. A brood parasitic catfish of mouthbrooding cichlid fishes in Lake Tanganyika. Nature 323, 58–59 (1982).
Ronco, F. et al. Drivers and dynamics of a massive adaptive radiation in cichlid fishes. Nature 589, 76–81 (2021).
Blažek, R. et al. Success of cuckoo catfish brood parasitism reflects coevolutionary history and individual experience of their cichlid hosts. Sci. Adv. 4, eaar4380 (2018).
Wisenden, B. D. Alloparental care in fishes. Rev. Fish Biol. Fish. 9, 45–70 (1999).
Reichard, M. Cuckoo catfish. Curr. Biol. 29, R722–R723 (2019).
Zimmermann, H., Blažek, R., Polačik, M. & Reichard, M. Individual experience as a key to success for the cuckoo catfish brood parasitism. Nat. Commun. 13, 1723 (2022).
Cohen, M. S., Hawkins, M. B., Stock, D. W. & Cruz, A. Early life-history features associated with brood parasitism in the cuckoo catfish, Synodontis multipunctatus (Siluriformes: Mochokidae). Philos. Trans. R. Soc. B Biol. Sci. 374, 20180205 (2019).
Zimmerman, H., Tolman, D. & Reichard, M. Low incidence of cannibalism among brood parasitic cuckoo catfish embryos. Behav. Ecol. 34, 521–527 (2023).
Mouginot, P., Galipaud, M. & Reichard, M. The evolution of brood parasitism from host egg predation. Behav. Ecol. 35, arae043 (2024).
Day, J. J. & Wilkinson, M. On the origin of the Synodontis catfish species flock from Lake Tanganyika. Biol. Lett. 2, 548–552 (2006).
Koblmüller, S., Egger, B., Sturmbauer, C. & Sefc, K. M. Rapid radiation, ancient incomplete lineage sorting and ancient hybridization in the endemic Lake Tanganyika cichlid tribe Tropheini. Mol. Phylogenet. Evol. 55, 318–334 (2010).
Englmaier, G. K. et al. Revised taxonomy of Synodontis catfishes (Siluriformes: Mochokidae) from the Lake Tanganyika basin reveals lower species diversity than expected. Zool. J. Linn. Soc. 202, zlae130 (2024).
Wright, J. J. & Page, L. M. Taxonomic revision of Lake Tanganyikan Synodontis (Siluriformes: Mochokidae). Bull. Fla. Mus. Nat. Hist. 46, 57 (2006).
Peart, C. R., Bills, R., Newton, J., Near, T. J. & Day, J. J. Do sympatric catfish radiations in Lake Tanganyika show eco-morphological diversification? Evol. J. Linn. Soc. 3, kzae015 (2024).
Schluter, D. The Ecology of Adaptive Radiation (Oxford University Press, Oxford, 2000).
Stroud, J. T. & Losos, J. B. Ecological opportunity and adaptive radiation. Annu. Rev. Ecol. Evol. Syst. 47, 507–532 (2016).
Reichard, M. et al. Lack of host specialization despite selective host use in brood parasitic cuckoo catfish. Mol. Ecol. 32, 6070–6082 (2023).
Viertler, A., Salzburger, W. & Ronco, F. Comparative scale morphology in the adaptive radiation of cichlid fishes (Perciformes: Cichlidae) from Lake Tanganyika. Biol. J. Linn. Soc. 134, 541–556 (2021).
Jawad, L. A. Comparative morphology of scales of four teleost fishes from Sudan and Yemen. J. Nat. Hist. 39, 2643–2660 (2005).
Muschick, M., Indermaur, A. & Salzburger, W. Convergent evolution within an adaptive radiation of cichlid fishes. Curr. Biol. 22, 2362–2368 (2012).
Nair, P., Miller, C. M. & Fuiman, L. A. Tracing exploitation of egg boons: an experimental study using fatty acids and stable isotopes. J. Exp. Biol. 226, jeb246247 (2023).
Hunter, J. R. & Kimbrell, C. A. Egg cannibalism in the Northern anchovy, Engraulis mordax. Fish. Bull. 78, 811–816 (1980).
Yongo, E., Iteba, J. & Agember, S. Review of food and feeding habits of some Synodontis fishes in African freshwaters. Oceanogr. Fish. Open Access J. 10, 27–31 (2019).
Konings, A. Tanganyika Cichlids in Their Natural Habitat (Cichlid Press, El Paso, 2015).
Stoddard, M. C. & Hauber, M. E. Colour, vision and coevolution in avian brood parasitism. Philos. Trans. R. Soc. B Biol. Sci. 372, 20160339 (2017).
Santos, M. E. et al. The evolution of cichlid fish egg-spots is linked with a cis-regulatory change. Nat. Commun. 5, 5149 (2014).
Atukorala, A. D. S. & Franz-Odendaal, T. A. Spatial and temporal events in tooth development of Astyanax mexicanus. Mech. Dev. 134, 42–54 (2014).
Debiais-Thibaud, M. et al. Development of oral and pharyngeal teeth in the medaka (Oryzias latipes): comparison of morphology and expression of eve1 gene. J. Exp. Zoolog. B Mol. Dev. Evol. 308B, 693–708 (2007).
Koblmüller, S., Sturmbauer, C., Verheyen, E., Meyer, A. & Salzburger, W. Mitochondrial phylogeny and phylogeography of East African squeaker catfishes (Siluriformes: Synodontis). BMC Evol. Biol. 6, 49 (2006).
Cohen, M. S., Hawkins, M. B., Knox-Hayes, J., Vinton, A. C. & Cruz, A. A laboratory study of host use by the cuckoo catfish Synodontis multipunctatus. Environ. Biol. Fishes 101, 1417–1425 (2018).
Title, P. O. et al. The macroevolutionary singularity of snakes. Science 383, 918–923 (2024).
Polačik, M., Reichard, M., Smith, C. & Blažek, R. Parasitic cuckoo catfish exploit parental responses to stray offspring. Philos. Trans. R. Soc. B Biol. Sci. 374, 20180412 (2019).
Kattan, G. H. Shiny cowbirds follow the ‘shotgun’ strategy of brood parasitism. Anim. Behav. 53, 647–654 (1997).
Payne, R. B. The evolution of clutch size and reproductive rates in parasitic cuckoos. Evolution 28, 169–181 (1974).
Langmore, N. E. et al. Coevolution with hosts underpins speciation in brood-parasitic cuckoos. Science 384, 1030–1036 (2024).
McClelland, S. C. et al. Highly virulent avian brood-parasitic species show elevated embryonic metabolic rates at specific incubation stages compared to less virulent and non-parasitic species. Biol. Lett. 20, 20240411 (2024).
Spottiswoode, C. N. & Koorevaar, J. A stab in the dark: chick killing by brood parasitic honeyguides. Biol. Lett. 8, 241–244 (2012).
Reichard, M. & Zimmermann, H. Dataset and analytical code for ‘The ecological and developmental foundations of brood parasitism in a catfish’. Figshare. https://doi.org/10.6084/m9.figshare.28646141 (2026).
Oksanen, J. vegan: community ecology package. R package version 2, 5–6 (2019).
Wang, Y., Naumann, U., Wright, S. T. & Warton, D. I. mvabund– an R package for model-based analysis of multivariate abundance data. Methods Ecol. Evol. 3, 471–474 (2012).
Hogsden, K. L. & McHugh, P. A. Preservatives and sample preparation in stable isotope analysis of New Zealand freshwater invertebrates. N. Z. J. Mar. Freshw. Res. 51, 455–464 (2017).
Kiljunen, M. et al. A revised model for lipid-normalizing δ13 C values from aquatic organisms, with implications for isotope mixing models. J. Appl. Ecol. 43, 1213–1222 (2006).
McCutchan, J. H. Jr, Lewis, W. M. Jr, Kendall, C. & McGrath, C. C. Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 102, 378–390 (2003).
Post, D. M. Using Stable Isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83, 703–718 (2002).
Martinez Arabizu, P. pairwiseAdonis: Pairwise Multilevel Comparison using Adonis (R package version 0.4, 1, 2020).
Dyková, I. et al. Oogenesis, spermatogenesis and spermiation structures in Lake Tanganyika Synodontis species (Mochokidae, Telostei: Siluriformes). J. Vertebr. Biol. 73, 24023 (2024).
Brown-Peterson, N. J., Wyanski, D. M., Saborido-Rey, F., Macewicz, B. J. & Lowerre-Barbieri, S. K. A standardized terminology for describing reproductive development in fishes. Mar. Coast. Fish. 3, 52–70 (2011).
Sipos, M. J. et al. Evaluation of cGnRH IIa for induction spawning of two ornamental Synodontis species. Aquaculture 511, 734226 (2019).
Troscianko, J. & Stevens, M. Image calibration and analysis toolbox – a free software suite for objectively measuring reflectance, colour and pattern. Methods Ecol. Evol. 6, 1320–1331 (2015).
Brooks, M. E. glmmTMB} Balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J. 9, 378–400 (2017).
Hartig, F. DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models (R Package Version 0.4. 4, 2021).
Lenth R. V., Piaskowski J. emmeans: Estimated Marginal Means, aka Least-squares Means (R package version 2.0.1, 2025).
Revell, L. Phytools 2.0: an updated R ecosystem for phylogenetic comparative methods (and other things). PeerJ 12, e16505 (2024).
Harmon, L. J., Weir, J. T., Brock, C. D., Glor, R. E. & Challenger, W. GEIGER: investigating evolutionary radiations. Bioinformatics 24, 129–131 (2008).
Walker, M. & Kimmel, C. A two-color acid-free cartilage and bone stain for zebrafish larvae. Biotech. Histochem. 82, 23–28 (2007).
Acknowledgements
We thank the Department of Fisheries in Mpulungu, especially Lwabanya Mabo, for facilitating research in Zambia, and Tanzanian Fisheries Research Institute in Kigoma, especially Deogratias P. Mulokozi, for facilitating research in Tanzania. The manuscript benefited from comments from Carl Smith, Lukáš Kratochvíl, Miranda Sherlock and four anonymous referees. This research was funded by the Czech Science Foundation (21-00788X to M.R.) and, in part, by the Austrian Science Fund (FWF) (doi.org/10.55776/J4584 to H.Z). For the purpose of open access, the authors have applied a CC BY public copyright license to any Author Accepted Manuscript version arising from this submission.
Author information
Authors and Affiliations
Contributions
Conceptualization: M.R., S.K., H.Z. Methodology: M.R., H.Z., R.B., K.P., J.Ž., R.C. Investigation: M.R., H.Z., R.B., T.S., M.P., G.E., V.B., K.P., J.Ž., L.K., I.D., S.K. Visualization: M.R., H.Z., T.S., G.E. Funding acquisition: M.R., H.Z. Project administration: M.R. Supervision: M.R. Writing – original draft: M.R., H.Z. Writing – review and editing: M.R, H.Z., R.B., T.S., M.P., G.E., V.B., K.P., J.Ž., L.K., I.D., R.C., S.K.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Meghan Chenoweth and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Reichard, M., Blažek, R., Polačik, M. et al. The ecological and developmental foundations of brood parasitism in a catfish. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71179-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71179-4