Abstract
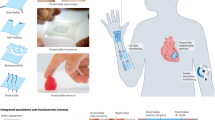
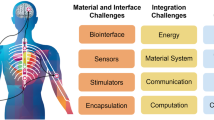
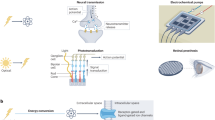
Recent advances in the development of in-body bioelectronic systems are providing new opportunities for the clinical management of various diseases and disorders. These emerging technologies are tailored to specific organs and are beginning to blend both diagnostic sensing and therapeutic actuation. The aim of these systems is to seamlessly integrate with the physiological environment, as illustrated by the diverse device strategies discussed throughout this article. Next generation modalities, such as optogenetics combining gene therapy with devices for photostimulation, are gaining popularity and offer advantages over existing therapeutic strategies. In this perspective, we explore the current state of technological developments, key challenges in the field and potential pathways for translating these innovations into clinical practice.
Similar content being viewed by others
References
Pavlov, V. A. & Tracey, K. J. Bioelectronic medicine: preclinical insights and clinical advances. Neuron 110, 3627–3644 (2022).
McRae, J. C., Jastrzebska-Perfect, P. & Traverso, G. Challenges and opportunities for ingestible electronics across timescales. Device 1, 100055 (2023).
Mariello, M. Reliability and stability of Bioelectronic medicine: a critical and pedagogical perspective. Bioelectron. Med. 11, 16 (2025).
Schalk, G. et al. Translation of neurotechnologies. Nat. Rev. Bioeng. 2, 637–652 (2024).
Zhang, Y. et al. Advances in bioresorbable materials and electronics. Chem. Rev. 123, 11722–11773 (2023).
Min, S. et al. Wearable blood pressure sensors for cardiovascular monitoring and machine learning algorithms for blood pressure estimation. Nat. Rev. Cardiol. 22, 629–648 (2025).
Madhvapathy, S. R. et al. Implantable bioelectronics and wearable sensors for kidney health and disease. Nat. Rev. Nephrol. 21, 443–463 (2025).
Pandey, A. & Sahoo, S. Progress on medical implant: a review and prospects. J. Bionic Eng. 20, 470–494 (2023).
Chiang, C.-H. et al. Development of a neural interface for high-definition, long-term recording in rodents and nonhuman primates. Sci. Transl. Med. 12, eaay4682 (2020).
Kim, T. I. et al. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science https://doi.org/10.1126/science.1232437 (2013).
Koo, J. et al. Wireless bioresorbable electronic system enables sustained nonpharmacological neuroregenerative therapy. Nat. Med 24, 1830–1836 (2018).
Kopeček, J. Swell gels. Nature 417, 389–391 (2002).
Yuk, H., Wu, J. & Zhao, X. Hydrogel interfaces for merging humans and machines. Nat. Rev. Mater. 7, 935–952 (2022).
Yuk, H., Lu, B. & Zhao, X. Hydrogel bioelectronics. Chem. Soc. Rev. 48, 1642–1667 (2019).
Kleber, C., Lienkamp, K., Rühe, J. & Asplund, M. Electrochemically controlled drug release from a conducting polymer hydrogel (PDMAAp/PEDOT) for local therapy and bioelectronics. Adv. Healthc. Mater. 8, 1801488 (2019).
Chapman, C. A. R., Cuttaz, E. A., Goding, J. A. & Green, R. A. Actively controlled local drug delivery using conductive polymer-based devices. Appl. Phys. Lett. 116, 010501 (2020).
Dong, C. et al. Electrochemically actuated microelectrodes for minimally invasive peripheral nerve interfaces. Nat. Mater. https://doi.org/10.1038/s41563-024-01886-0 (2024).
Torricelli, F. et al. Electrolyte-gated transistors for enhanced performance bioelectronics. Nat. Rev. Methods Prim. 1, 1–24 (2021).
Saleh, A., Koklu, A., Uguz, I., Pappa, A.-M. & Inal, S. Bioelectronic interfaces of organic electrochemical transistors. Nat. Rev. Bioeng. https://doi.org/10.1038/s44222-024-00180-7 (2024).
Wang, Y. et al. Designing organic mixed conductors for electrochemical transistor applications. Nat. Rev. Mater. 9, 249–265 (2024).
Liu, D. et al. Increasing the dimensionality of transistors with hydrogels. Science 390, 824–830 (2025).
Strakosas, X. et al. Metabolite-induced in vivo fabrication of substrate-free organic bioelectronics. Science 379, 795–802 (2023).
Zhang, A. et al. Genetically targeted chemical assembly of polymers specifically localized extracellularly to surface membranes of living neurons. Sci. Adv. 9, eadi1870 (2023).
Minev, I. R. Electronic tissue technologies for seamless biointerfaces. J. Polym. Sci. 61, 1707–1712 (2023).
Mantione, D. et al. Poly(3,4-ethylenedioxythiophene):GlycosAminoGlycan aqueous dispersions: toward electrically conductive bioactive materials for neural interfaces. Macromol. Biosci. 16, 1227–1238 (2016).
Saez, J. et al. A 3D bioelectrical interface to assess colorectal cancer progression in vitro. Mater. Today Chem. 24, 100990 (2022).
Da Silva, A. C., Wang, J. & Minev, I. R. Electro-assisted printing of soft hydrogels via controlled electrochemical reactions. Nat. Commun. 13, 1353 (2022).
Zhao, Z. et al. Super-resolution imaging of highly curved membrane structures in giant vesicles encapsulating molecular condensates. Adv. Mater. 34, 2106633 (2022).
Terenzi, L. et al. Engineering lipid-based pop-up conductive interfaces with PEDOT:PSS and light-responsive azopolymer films. Adv. Healthc. Mater. 13, 2303812 (2024).
Li, Q. et al. A highly conductive n -type conjugated polymer synthesized in water. J. Am. Chem. Soc. 146, 15860–15868 (2024).
Wu, Y. et al. High-conductivity PBFDO-based self-adhesive hydrogel for low-hysteresis flexible sensing applications. Polymer 336, 128876 (2025).
Tao, K., Makam, P., Aizen, R. & Gazit, E. Self-assembling peptide semiconductors. Science 358, eaam9756 (2017).
Liu, Y. et al. Soft and elastic hydrogel-based microelectronics for localized low-voltage neuromodulation. Nat. Biomed. Eng. 3, 58–68 (2019).
Cheng, S., Zhu, R. & Xu, X. Hydrogels for next generation neural interfaces. Commun. Mater. 5, 99 (2024).
Nie, L. et al. Fabrication and desired properties of conductive hydrogel dressings for wound healing. RSC Adv. 13, 8502–8522 (2023).
Zhang, Z. et al. Soft bioelectronics for therapeutics. ACS Nano 17, 17634–17667 (2023).
Wu, Z. et al. Interstitial fluid-based wearable biosensors for minimally invasive healthcare and biomedical applications. Commun. Mater. 5, 1–15 (2024).
Zhao, L. et al. Emerging sensing and modeling technologies for wearable and cuffless blood pressure monitoring. NPJ Digit Med 6, 93 (2023).
Mansour, M., Saeed Darweesh, M. & Soltan, A. Wearable devices for glucose monitoring: a review of state-of-the-art technologies and emerging trends. Alex. Eng. J. 89, 224–243 (2024).
Madhvapathy, S. R. et al. Implantable bioelectronic systems for early detection of kidney transplant rejection. Science 381, 1105–1112 (2023).
Madhvapathy, S. R. et al. Miniaturized implantable temperature sensors for the long-term monitoring of chronic intestinal inflammation. Nat. Biomed. Eng. https://doi.org/10.1038/s41551-024-01183-w (2024).
Kwon, K. et al. A battery-less wireless implant for the continuous monitoring of vascular pressure, flow rate and temperature. Nat. Biomed. Eng. 7, 1215–1228 (2023).
Yin, L. et al. Dissolvable metals for transient electronics. Adv. Funct. Mater. 24, 645–658 (2014).
Lendlein, A., Schmidt, A. M. & Langer, R. AB-polymer networks based on oligo(epsilon-caprolactone) segments showing shape-memory properties. Proc. Natl. Acad. Sci. USA 98, 842–847 (2001).
Halacheva, S. S. et al. Injectable biocompatible and biodegradable pH-responsive hollow particle gels containing poly(acrylic acid): the effect of copolymer composition on gel properties. Biomacromolecules 15, 1814–1827 (2014).
Changez, M., Koul, V., Krishna, B., Dinda, A. K. & Choudhary, V. Studies on biodegradation and release of gentamicin sulphate from interpenetrating network hydrogels based on poly(acrylic acid) and gelatin: in vitro and in vivo. Biomaterials 25, 139–146 (2004).
Bruining, M. J. et al. Biodegradable three-dimensional networks of poly(dimethylamino ethyl methacrylate). Synthesis, characterization and in vitro studies of structural degradation and cytotoxicity. Biomaterials 21, 595–604 (2000).
Liu, J. et al. Bioresorbable shape-adaptive structures for ultrasonic monitoring of deep-tissue homeostasis. Science 383, 1096–1103 (2024).
Li, S. et al. Bioresorbable, wireless, passive sensors for continuous pH measurements and early detection of gastric leakage. Sci. Adv. 10, eadj0268 (2024).
Kim, J. et al. A wireless, implantable bioelectronic system for monitoring urinary bladder function following surgical recovery. Proc. Natl. Acad. Sci. USA 121, e2400868121 (2024).
Choi, Y. S. et al. Fully implantable and bioresorbable cardiac pacemakers without leads or batteries. Nat. Biotechnol. 39, 1228–1238 (2021).
Agarwal, K., Jegadeesan, R., Guo, Y.-X. & Thakor, N. V. Wireless power transfer strategies for implantable bioelectronics. IEEE Rev. Biomed. Eng. 10, 136–161 (2017).
Jafari, M., Marquez, G., Dechiraju, H., Gomez, M. & Rolandi, M. Merging machine learning and bioelectronics for closed-loop control of biological systems and homeostasis. Cell Rep. Phys. Sci. 4, 101535 (2023).
Sunwoo, S.-H. et al. Soft bioelectronics for the management of cardiovascular diseases. Nat. Rev. Bioeng. 2, 8–24 (2024).
Dennison, M. S. et al. Biodegradable electronic materials for promoting sustainability in next-generation electronics—a comprehensive review. Discov. Mater. 5, 182 (2025).
Corsi, M., Bellotti, E., Surdo, S. & Barillaro, G. Implantable bioresorbable electronic systems for sustainable precision medicine. Nat. Rev. Electr. Eng. 2, 572–583 (2025).
Bendale, G. S. et al. Nerve tape is an effective tool for small nerve repair. Plast. Reconstruct. Surg. Glob. Open 13, e7043 (2025).
Gunnam, S., Chopra, S., Khanna, A., Shukla, V. K. & Sharma, N. Regulatory guidelines for bioresorbable medical devices. in Bioresorbable Materials and Bioactive Surface Coatings https://doi.org/10.1016/B978-0-443-31606-7.00014-4 (Elsevier, 2025).
Ahn, H.-Y. et al. Bioresorbable, wireless dual stimulator for peripheral nerve regeneration. Nat. Commun. 16, 4752 (2025).
Laksono, S. Bioresorbable vascular scaffolds: should we use them again? Int. J. Cardiovasc. Pract. 8, e141366 (2023).
Zhang, Y. et al. Millimetre-scale bioresorbable optoelectronic systems for electrotherapy. Nature 640, 77–86 (2025).
Bae, J.-Y. et al. A biodegradable and self-deployable electronic tent electrode for brain cortex interfacing. Nat. Electron 7, 815–828 (2024).
Lee, G. et al. A bioresorbable peripheral nerve stimulator for electronic pain block. Sci. Adv. 8, eabp9169 (2022).
Kaelberer, M. M. et al. A gut-brain neural circuit for nutrient sensory transduction. Science 361, eaat5236 (2018).
Mackay, R. S. & Jacobson, B. Endoradiosonde. Nature 179, 1239–1240 (1957).
Iddan, G., Meron, G., Glukhovsky, A. & Swain, P. Wireless capsule endoscopy. Nature 405, 417–417 (2000).
Liu, K. et al. Gastrointestinal neuromuscular interfaces: bioelectronic device design for gastrointestinal motility. Device 3, 100867 (2025).
Kirtane, A. R. et al. A once-a-month oral contraceptive. Sci. Transl. Med. 11, eaay2602 (2019).
Ramadi, K. B. et al. Bioinspired, ingestible electroceutical capsules for hunger-regulating hormone modulation. Sci. Robot. 8, eade9676 (2023).
Srinivasan, S. S. et al. An ingestible self-propelling device for intestinal reanimation. Sci. Robot. 9, eadh8170 (2024).
Abramson, A. et al. An ingestible self-orienting system for oral delivery of macromolecules. Science 363, 611–615 (2019).
Abramson, A. et al. A luminal unfolding microneedle injector for oral delivery of macromolecules. Nat. Med. 25, 1512–1518 (2019).
Srinivasan, S. S. et al. RoboCap: robotic mucus-clearing capsule for enhanced drug delivery in the gastrointestinal tract. Sci. Robot. 7, eabp9066 (2022).
Levy, J. A., Straker, M. A., Stine, J. M., Stephen, J. A. C. & Ghodssi, R. Microinjectors for controlled cross-barrier drug delivery in the gastrointestinal tract. ACS Appl. Mater. Interfaces 17, 36420–36432 (2025).
Abramson, A. et al. Ingestible transiently anchoring electronics for microstimulation and conductive signaling. Sci. Adv. 6, eaaz0127 (2020).
Srinivasan, S. S. et al. A vibrating ingestible bioelectronic stimulator modulates gastric stretch receptors for illusory satiety. Sci. Adv. 9, eadj3003 (2023).
Rao, S. S. C., Quigley, E. M. M., Chey, W. D., Sharma, A. & Lembo, A. J. Randomized placebo-controlled phase 3 trial of vibrating capsule for chronic constipation. Gastroenterology 164, 1202–1210.e6 (2023).
Carey, J. B. A simplified gastrointestinal biopsy capsule. Gastroenterology 46, 550–557 (1964).
Traverso, G. et al. First-in-human trial of an ingestible vitals-monitoring pill. Device 1, 100125 (2023).
You, S. S. et al. An ingestible device for gastric electrophysiology. Nat. Electron 7, 497–508 (2024).
Jia, N. Z. et al. Shape memory-based gastric motility 3D mapping. Device 1, 100010 (2023).
Kalantar-Zadeh, K. et al. A human pilot trial of ingestible electronic capsules capable of sensing different gases in the gut. Nat. Electron 1, 79–87 (2018).
Min, J. et al. Continuous biochemical profiling of the gastrointestinal tract using an integrated smart capsule. Nat. Electron https://doi.org/10.1038/s41928-025-01407-0 (2025).
Rezaei Nejad, H. et al. Ingestible osmotic pill for in vivo sampling of gut microbiomes. Adv. Intell. Syst. 1, 1900053 (2019).
Chen, L., Gruzinskyte, L., Jørgensen, S. L., Boisen, A. & Srivastava, S. K. An ingestible self-polymerizing system for targeted sampling of gut Microbiota and Biomarkers. ACS Nano 14, 12072–12081 (2020).
Mohammed, H. et al. Passive intestinal microbiome sampling using an ingestible device with tortuous lattices. Device https://doi.org/10.1016/j.device.2025.100904 (2025).
Shafieinouri, M. et al. Gut-brain nexus: mapping multimodal links to neurodegeneration at biobank scale. Sci. Adv. 11, eadu2937 (2025).
Payne, S. C., Furness, J. B. & Stebbing, M. J. Bioelectric neuromodulation for gastrointestinal disorders: effectiveness and mechanisms. Nat. Rev. Gastroenterol. Hepatol. 16, 89–105 (2019).
Sharma, S. et al. Location-aware ingestible microdevices for wireless monitoring of gastrointestinal dynamics. Nat. Electron 6, 242–256 (2023).
Sharova, A. S., Melloni, F., Lanzani, G., Bettinger, C. J. & Caironi, M. Edible electronics: the vision and the challenge. Adv. Mater. Technol. 6, 2000757 (2021).
Sun, Y. et al. Magnetically driven capsules with multimodal response and multifunctionality for biomedical applications. Nat. Commun. 15, 1839 (2024).
Gora, M. J. et al. Tethered capsule endomicroscopy enables less invasive imaging of gastrointestinal tract microstructure. Nat. Med. 19, 238–240 (2013).
Gregersen, H. et al. Fecobionics in proctology: review and perspectives. Surg. Open Digest. Adv. 12, 100117 (2023).
Xue, J. et al. Scalable balloon catheter assisted contact enhancement of 3D electrode array for colon electrophysiological recording. Sens. Actuators B: Chem. 424, 136955 (2025).
Srinivasan, S. S. et al. Luminal electrophysiological neuroprofiling system for gastrointestinal neuromuscular diseases. Device 2, 100400 (2024).
Willett, F. R. et al. A high-performance speech neuroprosthesis. Nature 620, 1031–1036 (2023).
Metzger, S. L. et al. A high-performance neuroprosthesis for speech decoding and avatar control. Nature 620, 1037–1046 (2023).
Kacker, K. et al. Motor activity in gamma and high gamma bands recorded with a Stentrode from the human motor cortex in two people with ALS. J. Neural Eng. 22, 026036 (2025).
Gittis, A. H. & Sillitoe, R. V. Circuit-specific deep brain stimulation provides insights into movement control. Annu. Rev. Neurosci. https://doi.org/10.1146/annurev-neuro-092823-104810 (2024).
Cho, N. et al. Hypothalamic deep brain stimulation augments walking after spinal cord injury. Nat. Med. https://doi.org/10.1038/s41591-024-03306-x (2024).
Austelle, C. W., Cox, S. S., Wills, K. E. & Badran, B. W. Vagus nerve stimulation (VNS): recent advances and future directions. Clin. Auton. Res 34, 529–547 (2024).
Kathe, C. et al. The neurons that restore walking after paralysis. Nature 611, 540–547 (2022).
Wagner, F. B. et al. Targeted neurotechnology restores walking in humans with spinal cord injury. Nature https://doi.org/10.1038/s41586-018-0649-2 (2018).
Soloukey, S. et al. The dorsal root ganglion as a novel neuromodulatory target to evoke strong and reproducible motor responses in chronic motor complete spinal cord injury: a case series of five patients. Neuromodulation 24, 779–793 (2021).
Luz, A., Rupp, R., Ahmadi, R. & Weidner, N. Beyond treatment of chronic pain: a scoping review about epidural electrical spinal cord stimulation to restore sensorimotor and autonomic function after spinal cord injury. Neurol. Res. Pract. 5, 14 (2023).
D’Anna, E. et al. A closed-loop hand prosthesis with simultaneous intraneural tactile and position feedback. Sci. Robot 4, eaau8892 (2019).
Boretius, T. et al. A transverse intrafascicular multichannel electrode (TIME) to interface with the peripheral nerve. Biosens. Bioelectron. 26, 62–69 (2010).
Raspopovic, S. et al. Restoring natural sensory feedback in real-time bidirectional hand prostheses. Sci. Transl. Med 6, 222ra19 (2014).
Iberite, F. et al. Restoration of natural thermal sensation in upper-limb amputees. Science 380, 731–735 (2023).
Lorach, H. et al. Walking naturally after spinal cord injury using a brain–spine interface. Nature 618, 126–133 (2023).
Sellers, K. K. et al. Closed-loop neurostimulation for the treatment of psychiatric disorders. Neuropsychopharmacology 49, 163–178 (2024).
Scangos, K. W. et al. Closed-loop neuromodulation in an individual with treatment-resistant depression. Nat. Med. 27, 1696–1700 (2021).
Formento, E. et al. Electrical spinal cord stimulation must preserve proprioception to enable locomotion in humans with spinal cord injury. Nat. Neurosci. 21, 1728–1741 (2018).
Lee, S. Y. et al. Cell-class-specific electric field entrainment of neural activity. Neuron 112, 2614–2630 (2024).
Emiliani, V. et al. Optogenetics for light control of biological systems. Nat. Rev. Methods Prim. 2, 55 (2022).
Asrican, B. et al. Next-generation transgenic mice for optogenetic analysis of neural circuits. Front. Neural Circuits 7, 160 (2013).
Drew, L. Restoring vision with optogenetics. Nature 639, S7–S9 (2025).
Freise, J. Regulatory aspects of optogenetic research and therapy for retinitis pigmentosa under EU law. Front. Med. Technol. 7, 1548927 (2025).
Sahel, J.-A. et al. Partial recovery of visual function in a blind patient after optogenetic therapy. Nat. Med 27, 1223–1229 (2021).
Mickle, A. D. et al. A wireless closed-loop system for optogenetic peripheral neuromodulation. Nature 565, 361–365 (2019).
Soliman, H. & Gourcerol, G. Gastric electrical stimulation: role and clinical impact on chronic nausea and vomiting. Front. Neurosci. 16, 909149 (2022).
Rakhilin, N. et al. Simultaneous optical and electrical in vivo analysis of the enteric nervous system. Nat. Commun. 7, 11800 (2016).
Hibberd, T. J. et al. Optogenetic induction of colonic motility in mice. Gastroenterology 155, 514–528.e6 (2018).
Sahasrabudhe, A. et al. Multifunctional microelectronic fibers enable wireless modulation of gut and brain neural circuits. Nat. Biotechnol. 42, 892–904 (2024).
Efimov, A. I. et al. Remote optogenetic control of the enteric nervous system and brain-gut axis in freely-behaving mice enabled by a wireless, battery-free optoelectronic device. Biosens. Bioelectron. 258, 116298 (2024).
Ouyang, W. et al. A wireless and battery-less implant for multimodal closed-loop neuromodulation in small animals. Nat. Biomed. Eng. 7, 1252–1269 (2023).
Zhou, N. & Ma, L. Smart bioelectronics and biomedical devices. Bio-des. Manuf. 5, 1–5 (2022).
Goh, G. D. et al. Machine learning for bioelectronics on wearable and implantable devices: challenges and potential. Tissue Eng. Part A 29, 20–46 (2023).
Jafari, M. et al. Feedback control of bioelectronic devices using machine learning. IEEE Control Syst. Lett. 5, 1133–1138 (2021).
Rochford, A. E., Carnicer-Lombarte, A., Curto, V. F., Malliaras, G. G. & Barone, D. G. When bio meets technology: biohybrid neural interfaces. Adv. Mater. 32, e1903182 (2020).
Stieglitz, T., Ruf, H. H., Gross, M., Schuettler, M. & Meyer, J.-U. A biohybrid system to interface peripheral nerves after traumatic lesions: design of a high channel sieve electrode. Biosens. Bioelectron. 17, 685–696 (2002).
Araki, Y., Solomon, B. A., Basile, R. M. & Chick, W. L. Biohybrid artificial pancreas. Long-term insulin secretion by devices seeded with canine islets. Diabetes 34, 850–854 (1985).
Urbanchek, M. G. et al. Development of a regenerative peripheral nerve interface for control of a neuroprosthetic limb. Biomed. Res Int 2016, 5726730 (2016).
Irwin, Z. T. et al. Chronic recording of hand prosthesis control signals via a regenerative peripheral nerve interface in a rhesus macaque. J. Neural Eng. 13, 046007 (2016).
Brown, J. et al. Optogenetic stimulation of a cortical biohybrid implant guides goal directed behavior. Preprint at https://doi.org/10.1101/2024.11.22.624907 (2024).
Azemi, E., Gobbel, G. T. & Cui, X. T. Seeding neural progenitor cells on silicon-based neural probes. J. Neurosurg. 113, 673–681 (2010).
Purcell, E. K., Seymour, J. P., Yandamuri, S. & Kipke, D. R. In vivo evaluation of a neural stem cell-seeded prosthesis. J. Neural Eng. 6, 026005 (2009).
Cullen, D. K. et al. Microtissue engineered constructs with living axons for targeted nervous system reconstruction. Tissue Eng. Part A 18, 2280–2289 (2012).
Maimon, B. E. et al. Optogenetic peripheral nerve immunogenicity. Sci. Rep. 8, 14076 (2018).
Williams, J. J., Watson, A. M., Vazquez, A. L. & Schwartz, A. B. Viral-mediated optogenetic stimulation of peripheral motor nerves in non-human primates. Front Neurosci. 13, 759 (2019).
Arjomandnejad, M., Dasgupta, I., Flotte, T. R. & Keeler, A. M. Immunogenicity of recombinant adeno-associated virus (AAV) vectors for gene transfer. BioDrugs 37, 311–329 (2023).
McMurphy, T. B. et al. AAV-mediated co-expression of an immunogenic transgene plus PD-L1 enables sustained expression through immunological evasion. Sci. Rep. 14, 28853 (2024).
Berry, M. H. et al. Restoration of high-sensitivity and adapting vision with a cone opsin. Nat. Commun. 10, 1221 (2019).
Yamashita, T. Discovery and design of photocyclic animal opsins: potential application to gene therapy from non-visual opsin research. Eye Vis. 12, 42 (2025).
Shen, Y., Campbell, R. E., Côté, D. C. & Paquet, M.-E. Challenges for therapeutic applications of opsin-based optogenetic tools in humans. Front. Neural Circuits 14, 41 (2020).
Hickey, D. G. et al. Chimeric human opsins as optogenetic light sensitisers. J. Exp. Biol. 224, jeb240580 (2021).
Chen, R. et al. Deep brain optogenetics without intracranial surgery. Nat. Biotechnol. 39, 161–164 (2021).
Gong, X. et al. An ultra-sensitive step-function opsin for minimally invasive optogenetic stimulation in mice and macaques. Neuron 107, 38–51.e8 (2020).
Shin, H. et al. Transcranial optogenetic brain modulator for precise bimodal neuromodulation in multiple brain regions. Nat. Commun. 15, 10423 (2024).
Chen, S. et al. Near-infrared deep brain stimulation via upconversion nanoparticle-mediated optogenetics. Science 359, 679–684 (2018).
Prasad, S. et al. Immune responses and immunosuppressive strategies for adeno-associated virus-based gene therapy for treatment of central nervous system disorders: current knowledge and approaches. Hum. Gene Ther. 33, 1228–1245 (2022).
Senior, M. Fresh from the biotech pipeline: record-breaking FDA approvals. Nat. Biotechnol. 42, 355–361 (2024).
Tosolini, A. P. & Smith, G. M. Editorial: gene therapy for the central and peripheral nervous system. Front. Mol. Neurosci. 11, 54 (2018).
Russell, S. et al. Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) in patients with RPE65 -mediated inherited retinal dystrophy: a randomised, controlled, open-label, phase 3 trial. Lancet 390, 849–860 (2017).
Mendell, J. R. et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N. Engl. J. Med 377, 1713–1722 (2017).
Mullard, A. FDA approves l-amino acid decarboxylase deficiency gene therapy. Nat. Rev. Drug Discov. 24, 6–6 (2024).
Skinnider, M. A. et al. Cell type prioritization in single-cell data. Nat. Biotechnol. 39, 30–34 (2021).
Qian, Y. et al. Programmable RNA sensing for cell monitoring and manipulation. Nature 610, 713–721 (2022).
Ben-Simon, Y. et al. A suite of enhancer AAVs and transgenic mouse lines for genetic access to cortical cell types. Cell 188, 3045–3064.e23 (2025).
Graybuck, L. T. et al. Enhancer viruses for combinatorial cell subclass-specific labeling. Neuron 109, 1449–1464.e13 (2021).
Kathe, C. et al. Wireless closed-loop optogenetics across the entire dorsoventral spinal cord in mice. Nat. Biotechnol. 40, 198–208 (2021).
Liu, X. et al. Fatigue-resistant hydrogel optical fibers enable peripheral nerve optogenetics during locomotion. Nat. Methods 20, 1802–1809 (2023).
Banerjee, H., Bartolomei, N., Song, J. & Sorin, F. Soft, stretchable optical fibers via thermal drawing. EPJ Web Conf. 287, 10003 (2023).
Cao, Y. et al. Flexible and stretchable polymer optical fibers for chronic brain and vagus nerve optogenetic stimulations in free-behaving animals. BMC Biol. 19, 252 (2021).
Chen, H.-H. et al. Optogenetic activation of peripheral somatosensory neurons in transgenic mice as a neuropathic pain model for assessing the therapeutic efficacy of analgesics. ACS Pharm. Transl. Sci. 7, 236–248 (2024).
White, M., Mackay, M. & Whittaker, R. G. Taking optogenetics into the human brain: opportunities and challenges in clinical trial design. OAJCT 12, 33–41 (2020).
Hsueh, B. et al. Cardiogenic control of affective behavioural state. Nature 615, 292–299 (2023).
Sakai, D., Tomita, H. & Maeda, A. Optogenetic therapy for visual restoration. Int. J. Mol. Sci. 23, 15041 (2022).
Busskamp, V., Roska, B. & Sahel, J.-A. Optogenetic vision restoration. Cold Spring Harb. Perspect. Med 14, a041660 (2024).
Huet, A., Mager, T., Gossler, C. & Moser, T. Toward optogenetic hearing restoration. Annu. Rev. Neurosci. 47, 103–121 (2024).
Duan, X. et al. Suppression of epileptic seizures by transcranial activation of K+-selective channelrhodopsin. Nat. Commun. 16, 559 (2025).
Ledri, M., Andersson, M., Wickham, J. & Kokaia, M. Optogenetics for controlling seizure circuits for translational approaches. Neurobiol. Dis. 184, 106234 (2023).
Fakhoury, M. Optogenetics: a revolutionary approach for the study of depression. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 106, 110094 (2021).
Iyer, S. M. et al. Optogenetic and chemogenetic strategies for sustained inhibition of pain. Sci. Rep. 6, 30570 (2016).
Iyer, S. M. et al. Virally mediated optogenetic excitation and inhibition of pain in freely moving nontransgenic mice. Nat. Biotechnol. 32, 274–278 (2014).
Bi, Y., Wang, Y., Gong, L., Ren, Y. & Guo, Y. Insight into the application of optogenetics for regulating motor-related circuits in Parkinson’s disease and novel therapeutic target exploration. Parkinsonism Relat. Disord. https://doi.org/10.1016/j.parkreldis.2025.107988 (2025).
Hyeon, B. et al. Integrating artificial intelligence and optogenetics for Parkinson’s disease diagnosis and therapeutics in male mice. Nat. Commun. 16, 7797 (2025).
White, M. & Whittaker, R. G. Post-trial considerations for an early phase optogenetic trial in the human brain. OAJCT ume 14, 1–9 (2022).
Lüscher, C. et al. Roadmap for direct and indirect translation of optogenetics into discoveries and therapies for humans. Nat. Neurosci. 28, 2415–2431 (2025).
Lucas, A., Revell, A. & Davis, K. A. Artificial intelligence in epilepsy — applications and pathways to the clinic. Nat. Rev. Neurol. 20, 319–336 (2024).
Thenaisie, Y. et al. Principles of gait encoding in the subthalamic nucleus of people with Parkinson’s disease. Sci. Transl. Med. 14, eabo1800 (2022).
Cheung, C. Y. et al. A deep learning model for detection of Alzheimer’s disease based on retinal photographs: a retrospective, multicentre case-control study. Lancet Digit. Health 4, e806–e815 (2022).
Card, N. S. et al. An accurate and rapidly calibrating speech neuroprosthesis. N. Engl. J. Med. 391, 609–618 (2024).
Lee, K. et al. A brain-controlled exoskeleton with cascaded event-related desynchronization classifiers. Robot. Autonomous Syst. 90, 15–23 (2017).
Hochberg, L. R. et al. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 485, 372–375 (2012).
Losanno, E., Mender, M., Chestek, C., Shokur, S. & Micera, S. Neurotechnologies to restore hand functions. Nat. Rev. Bioeng. 1, 390–407 (2023).
Perni, S., Lehmann, L. S. & Bitterman, D. S. Patients should be informed when AI systems are used in clinical trials. Nat. Med 29, 1890–1891 (2023).
Finlayson, S. G. et al. The clinician and dataset shift in artificial intelligence. N. Engl. J. Med. 385, 283–286 (2021).
Acknowledgements
S.C., T.H.H. and K.L. are supported by the Wyss Center Geneva Lighthouse partnership. T.H.H. is supported by the Swiss National Science Foundation (grant number: 10006212). C.K. is supported by the Swiss National Science Foundation (grant number: 218058), Novartis Foundation of Medical-Biological Research (24B094) and the International Foundation for Research in Paraplegia (P205). M.J.G. is supported by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (grant agreement No 853378). K.L. is supported by Innosuisse Project 103.116 IP-LS. I.M. acknowledges funding from the European Research Council (Consolidator Grant GELECTRO, 101125081). Views and opinions expressed are however those of the authors only and do not necessarily reflect those of the European Union and neither the European Union nor the granting authority can be held responsible for them. J.R. and M.W. are supported by the Querrey Simpson Institute for Bioelectronics.
Author information
Authors and Affiliations
Contributions
C.K. and T.H.H. conceived the manuscript and supervised the work. All authors contributed to writing, reviewing and editing the manuscript. M.W., J.R., C.T., I.M., K.L., C.K. and T.H.H. prepared the figures. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.R. is co-founder of the company Neurolux, Inc, which provides devices to support optogenetic studies in small animal models. M.J.G. is an inventor on a pending patent application (No. PCT/IB2025/056531) filed by the Wyss Center for Bio and Neuro Engineering that covers method for functional evaluation of the gut. All other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ceto, S., Elshove, S.A., Wu, M. et al. Transforming healthcare through in-body bioelectronic systems. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71188-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71188-3