Abstract
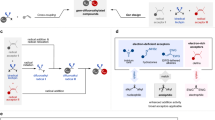
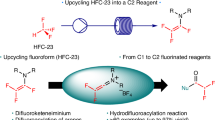
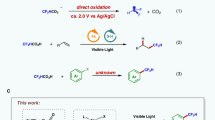
Discovery and application of reactive intermediates has prominently expedited the development of organic chemistry. In this respect, while ketene has been well recognized as a versatile intermediate for a wide range of transformations, practical application of difluoroketene remained virtually unrealized because of its highly reactive nature. Herein, we present a transition-metal-free approach for in situ generation of difluoroketene using difluorobromoacetylsilane as a precursor. The controlled release of chemically labile difluoroketene under mild conditions, achieved through desilylative β-elimination, establishes a foundation for successful development of its Belluš-Claisen rearrangement with allylic amines, Staudinger [2 + 2] cycloaddition with imines, [4 + 2] cycloaddition, as well as insertion into alcohols, amines, and thiols for their difluoroacetylation. A series of mechanistic experiments provide concrete support for the involvement of difluoroketene in these transformations.
Similar content being viewed by others
Data availability
The data generated in this study relating to the characterization of starting materials and products, general methods, optimization studies, experimental procedures, mechanistic studies, and NMR spectra are available in Supplementary Information. Data supporting the findings of this manuscript are also available from the corresponding author upon request.
References
Singh, M. S. Reactive Intermediates in Organic Chemistry (Wiley-VCH, 2014).
Allen, A. D. & Tidwell, T. T. Ketenes and other cumulenes as reactive intermediates. Chem. Rev. 113, 7287–7342 (2013).
Tidwell, T. T. The First Century of Ketenes (1905–2005): the birth of a versatile family of reactive intermediates. Angew. Chem. Int. Ed. 44, 5778–5785 (2005).
Paull, D. H., Weatherwax, A. & Lectka, T. Catalytic, asymmetric reactions of ketenes and ketene enolates. Tetrahedron 65, 6771–6803 (2009).
Gong, L., McAllister, M. A. & Tidwell, T. T. Substituent effects on ketene structure and stability: an ab initio study. J. Am. Chem. Soc. 113, 6021–6028 (1991).
Kötting, C., Sander, W., Senzlober, M. & Bürger, H. Oxidation of difluorovinylidene. Chem. Eur. J. 4, 1611–1615 (1998).
Dawson, D. F. & Holmes, J. L. Generation of F2C=C=O and F2C=C=O•+ from Ionized CF2=CFOCF3. First experimental observation of gas-phase neutral perfluoroketene. J. Phys. Chem. A 103, 5217–5220 (1999).
Habibi, M. H. Cycloaddition of dihaloketenes with trimethylsiloxycycloolefins and fluorination of the products. J. Fluorine Chem. 37, 177–181 (1987).
England, D. C. & Krespan, C. G. Fluoroketenes. Ⅱ. Difluoroketene. J. Org. Chem. 33, 816–819 (1968).
Zhang, C. et al. Biological utility of fluorinated compounds: from materials design to molecular imaging, therapeutics and environmental remediation. Chem. Rev. 122, 167–208 (2022).
Guo, Z. et al. Fluorine-containing functional group-based energetic materials. Chem. Rec. 23, e202300108 (2023).
Ogawa, Y., Tokunaga, E., Kobayashi, O., Hirai, K. & Shibata, N. Current contributions of organofluorine compounds to the agrochemical industry. iScience 23, 101467 (2020).
Hong, W. P., Lim, H. N. & Shin, I. Recent progress and perspectives in photo-induced organic reactions of acylsilanes. Org. Chem. Front. 10, 819–836 (2023).
Liu, J. et al. Progress in the preparation and application of Arylsilane. Curr. Org. Chem. 27, 28–37 (2023).
Komiyama, T., Minami, Y. & Hiyama, T. Recent advances in transition-metal-catalyzed synthetic transformations of organosilicon reagents. ACS Catal 7, 631–651 (2017).
Wang, F. et al. Chloride ion-catalyzed generation of difluorocarbene for efficient preparation of gem-difluorinated cyclopropenes and cyclopropanes. Chem. Commun. 47, 2411–2413 (2011).
Li, L., Wang, F., Ni, C. & Hu, J. Synthesis of gem-Difluorocyclopropa(e)nes and O-, S-, N-, and P-Difluoromethylated Compounds with TMSCF2Br. Angew. Chem. Int. Ed. 52, 12390–12394 (2013).
Wang, F., Li, L., Ni, C. & Hu, J. Deoxygenative gem-difluoroolefination of carbonyl compounds with (chlorodifluoromethyl)trimethylsilane and triphenylphosphine. Beilstein J. Org. Chem. 10, 344–351 (2014).
Himeshima, Y., Sonoda, T. & Kobayashi, H. Fluoride-induced 1,2-elimination of o-trimethylsilylphenyl triflate to benzyne under mild conditions. Chem. Lett. 62, 1211–1214 (1983).
Shi, J., Li, L. & Li, Y. o‑Silylaryl Triflates: a journey of Kobayashi Aryne Precursors. Chem. Rev. 121, 3892–4044 (2021).
Sarmah, M., Sharma, A. & Gogoi, P. Exploration of Kobayashi’s aryne precursor: a potent reactive platform for the synthesis of polycyclic aromatic hydrocarbons. Org. Biomol. Chem. 19, 722–737 (2021).
Zhu, C., Sun, M.-M., Chen, K., Liu, H. & Feng, C. Selective C–F bond allylation of trifluoromethylalkenes. Angew. Chem. Int. Ed. 60, 20237–20242 (2021).
Liu, H., Ge, L., Wang, D.-X., Chen, N. & Feng, C. Photoredox-coupled F-nucleophilic addition: allylation of gem-difluoroalkenes. Angew. Chem. Int. Ed. 58, 3918–3922 (2019).
Wang, C.-Q., Ye, L., Feng, C. & Loh, T.-P. C−F bond cleavage enabled redox-neutral [4+1] annulation via C−H bond activation. J. Am. Chem. Soc. 139, 1762–1765 (2017).
Tian, P. et al. F- nucleophilic-addition-induced allylic alkylation. J. Am. Chem. Soc. 138, 15869–15872 (2016).
Tang, L., Liu, Z.-Y., She, W. & Feng, C. Selective single C–F bond arylation of trifluoromethylalkene derivatives. Chem. Sci. 10, 8701–8705 (2019).
Malherhe, R. & Belluš, D. A new type of Claisen rearrangement involving 1,3-dipolar intermediates. Helv. Chim. Acta 61, 3096–3099 (1978).
Gonda, J. The Belluš–Claisen rearrangement. Angew. Chem. Int. Ed. 43, 3516–3524 (2004).
Feng, C. et al. Preparation method of difluoroketene and method of converting into difluoroacetate and difluoroacetamide compound. CN Patent CN117843519 A (2024).
Feng, C. et al. Method for preparing α,α-difluorobutyrolactam using difluorobromoacetyl silicon compound and imine. CN Patent CN117843543 A (2024).
Feng, C. et al. Preparation of allyl-difluoroamide compounds. CN Patent CN119661475 A (2025).
Zhou, G. et al. Visible-light-induced [2 + 2] cyclization of alkynes with bromodifluoroacetylsilanes: facile access to gem-difluorocyclobutenones. J. Am. Chem. Soc. 147, 15955–15962 (2025).
Higashiya, S. et al. Synthesis of mono- and difluoroacetyltrialkylsilanes and the corresponding enol silyl ethers. J. Org. Chem. 69, 6323–6328 (2004).
Fu, N. & Tidwell, T. T. Preparation of β-lactams by [2+2] cycloaddition of ketenes and imines. Tetrahedron 64, 10465–10496 (2008).
Cassú, D. Computational insight into the scope of the Staudinger cycloaddition reaction for the preparation of β-lactams. ChemistrySelect 5, 3278–3282 (2020).
Grygorenko, O. O., Melnykov, K. P., Holovach, S. & Demchuk, O. Fluorinated cycloalkyl building blocks for drug discovery. ChemMedChem 17, e202200365 (2022).
Mondal, R., Agbaria, M. & Nairoukh, Z. Fluorinated rings: conformation and application. Chem. Eur. J. 27, 7193–7213 (2021).
Thaisrivongs, S., Schostarez, H. J., Pals, D. T. & Turner, S. R. alpha, alph-Difluoro-beta-aminodeoxystatine-containing renin-inhibitory peptides. J. Med. Chem. 30, 1837–1842 (1987).
Joyeau, R., Molines, Labia, H. R. & Wakselman, M. N-Aryl 3-halogenated azetidin-2-ones and benzocarbacephems, inhibitors of beta-lactamases. J. Med. Chem. 31, 370–374 (1988).
Braun, M. The asymmetric difluoro-reformatsky reaction. Eur. J. Org. Chem. 2021, 1825–1836 (2021).
Shi, J., Linden, A. & Heimgartner, H. Reactions of acid chlorides/ketenes with 2-substituted 4,5-dihydro-4,4-dimethyl-1,3-thiazoles: formation of penam derivatives. Helv. Chim. Acta 96, 1462–1481 (2013).
Barba, V., Hernández, C., Rojas-Lima, S., Farfán, N. & Santillan, R. Preparation of N-aryl-substituted spiro-β-lactams via Staudinger cycloaddition. Can. J. Chem. 77, 2025–2032 (1999).
Bari, S. S., Trehan, I. R., Sharma, A. K. & Manhas, M. S. A facile synthesis of Azetidine-2,4-diones. Synthesis 1992, 439–442 (1992).
Vedejs, E. & Buchanan, R. A. Macrolide synthesis via dichloroketene ring expansion. J. Org. Chem. 49, 1840–1841 (1984).
Katagiri, N. & Kato, T. Studies on ketene and its derivatives. Part 119. Reaction of haloketenes with 2-arylideneaminopyridines. J. Heterocycl. Chem. 21, 407–412 (1984).
Meißner, G., Kretschmar, K., Braun, T. & Kemnitz, E. Consecutive transformations of tetrafluoropropenes: hydrogermylation and catalytic C−F activation steps at a Lewis acidic aluminum fluoride. Angew. Chem. Int. Ed. 56, 16338–16341 (2017).
Cossío, F. P., Arrieta, A. & Sierra, M. A. The mechanism of the ketene−imine (Staudinger) Reaction in its centennial: Still an unsolved problem?. Acc. Chem. Res. 41, 925–936 (2008).
Ocampo, R., Dolbier, W. R. Jr & Paredes, R. Synthesis of 1,1-difluoroalkenes via α,α-difluoro-β-lactones. J. Fluorine Chem. 88, 41–50 (1998).
Otaka, A. et al. SmI2-mediated reduction of γ,γ-difluoro-α,β-enoates with application to the synthesis of functionalized (Z)-fluoroalkene-type dipeptide isosteres. J. Org. Chem. 69, 1634–1645 (2004).
Tarui, A. et al. One-pot ring-opening peptide synthesis using α,α-difluoro-β-lactams. Synthesis 52, 3657–3666 (2020).
Van Brabandt, W. & De Kimpe, N. Synthesis of 3,3-Difluoroazetidines. Synlett 2006, 2039–2042 (2006).
Cely-Veloza, W., Kato, M. J. & Coy-Barrera, E. Quinolizidine-type alkaloids: chemodiversity, occurrence, and bioactivity. ACS Omega 8, 27862–27893 (2023).
Acknowledgements
The authors gratefully acknowledge the financial support of the National Natural Science Foundation of China (grant no. 22271151 (C.F.), 22571158 (C.F.), 22301133 (C.Z.)), the State Key Laboratory of Fluorine and Nitrogen Chemistry and Advanced Materials (grant no. 2026PT0009 (C.F.)), the State Key Laboratory of Materials-Oriented Chemical Engineering (grant no. SKL-MCE-25A17 (C.F.)).
Author information
Authors and Affiliations
Contributions
C.F. conceived the project and directed the investigations. S.X., C.Z., and L.F. performed the reaction development and condition optimization. S.X. and L.F. performed the synthetic experiments, mechanistic study, and analyzed the experimental data with contributions from H.G., H.Z., and Y.L. C.Z., X.C., Y.Z., and C.F. wrote and revised the manuscript with input from all authors. W.J. contributed to the revision of the manuscript. K.G. and C.F. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Jianbo Wang and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xi, S., Zhang, C., Fu, L. et al. Unlocking the reactivity of difluoroketene and its synthetic applications. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71202-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71202-8