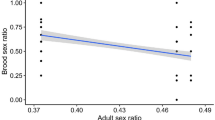
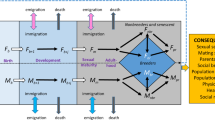
Abstract
The adult sex ratio plays a crucial role in shaping breeding systems and traits linked to sexual selection. Recent studies associate adult sex ratio with mate choice, pair-bonding, and parenting, as the rarer sex gains advantages in mate selection and parental investment. However, the causal relationships between the demographic factors that generate adult sex ratio bias and its broader implications remain debated. Skewed adult sex ratios can result from sex-biased mortality and maturation, which influence mating and parental behaviours. Conversely, the costs of these behaviours may further drive sex differences in mortality and maturation, reinforcing adult sex ratio biases. Here, we compile demographic and behavioural data from 261 bird species across 69 families to examine these associations within a phylogenetic framework. Our analyses confirm that demographic traits are associated with adult sex ratio and reveal links between adult sex ratio, pre-copulatory sexual selection, and parenting. Phylogenetic path analyses further demonstrate that sex differences in mortality and maturation drive adult sex ratio biases, which subsequently influence mating and parenting rather than the reverse. This study provides a comprehensive analysis of the interplay between demography, social environment, and breeding systems, highlighting adult sex ratio as a crucial link. Our findings underscore the need for further research on the demographic underpinnings of social behaviour and reproductive strategies.
Similar content being viewed by others
Data availability
The data used in this study are available in Figshare (https://doi.org/10.6084/m9.figshare.31490812)79. The Figshare repository includes the dataset used in the analyses and the phylogenetic trees used for phylogenetic mixed models and for generating Fig. 2. Data underlying Figs. 3 and 4 are provided in Table 1 and Supplementary Table 5. Source Data are provided for Fig. 5 and Supplementary Fig. 4. Source data are provided with this paper.
Code availability
The code used for data preparation, statistical analyses, and figure generation is available in Figshare (https://doi.org/10.6084/m9.figshare.31490812)79.
References
Schacht, R. et al. Adult sex ratios: causes of variation and implications for animal and human societies. Commun. Biol. 5, 1273 (2022).
Darwin, C. The Descent of Man, and Selection in Relation to Sex. Vol. 1. https://doi.org/10.1037/12293-000 (John Murray, 1871).
Székely, T., Weissing, F. J. & Komdeur, J. Adult sex ratio variation: implications for breeding system evolution. J. Evol. Biol. 27, 1500–1512 (2014).
Fresneau, N. et al. The evolution of sex roles: the importance of ecology and social environment. Proc. Natl. Acad. Sci. USA 121, e2321294121 (2024).
Fromhage, L. & Jennions, M. D. Coevolution of parental investment and sexually selected traits drives sex-role divergence. Nat. Commun. 7, 12517 (2016).
Long, X. & Weissing, F. J. Transient polymorphisms in parental care strategies drive divergence of sex roles. Nat. Commun. 14, 6805 (2023).
Kappeler, P. M. et al. Sex roles and sex ratios in animals. Biol. Rev. 98, 462–480 (2023).
Dyson, E. A. & Hurst, G. D. D. Persistence of an extreme sex-ratio bias in a natural population. Proc. Natl. Acad. Sci. USA 101, 6520–6523 (2004).
Kosztolányi, A., Barta, Z., Küpper, C. & Székely, T. Persistence of an extreme male-biased adult sex ratio in a natural population of polyandrous bird. J. Evol. Biol. 24, 1842–1846 (2011).
Ancona, S., Liker, A., Carmona-Isunza, M. C. & Székely, T. Sex differences in age-to-maturation relate to sexual selection and adult sex ratios in birds. Evol. Lett. 4, 44–53 (2020).
Promislow, D. E. L. Costs of sexual selection in natural populations of mammals. Proc. R. Soc. Lond. B Biol. Sci. 247, 203–210 (1992).
Promislow, D. E. L., Montgomerie, R. & Martin, T. E. Mortality costs of sexual dimorphism in birds. Proc. R. Soc. Lond. B Biol. Sci. 250, 143–150 (1992).
Eberhart-Phillips, L. J. et al. Sex-specific early survival drives adult sex ratio bias in snowy plovers and impacts mating system and population growth. Proc. Natl. Acad. Sci. USA. 114, E5474–E5481 (2017).
Székely, T., Liker, A., Freckleton, R. P., Fichtel, C. & Kappeler, P. M. Sex-biased survival predicts adult sex ratio variation in wild birds. Proc. R. Soc. Lond. B Biol. Sci. 281, 20140342 (2014).
Bleu, J., Bessa-Gomes, C. & Laloi, D. Evolution of female choosiness and mating frequency: Effects of mating cost, density and sex ratio. Anim. Behav. 83, 131–136 (2012).
Eberhart-Phillips, L. J. et al. Demographic causes of adult sex ratio variation and their consequences for parental cooperation. Nat. Commun. 9, 1651 (2018).
Ditchkoff, S. S., Welch, E. R., Lochmiller, R. L., Masters, R. E. & Starry, W. R. Age-specific causes of mortality among male white-tailed deer support mate-competition theory. J. Wildl. Manag. 65, 552 (2001).
Engqvist, L., Cordes, N. & Reinhold, K. Evolution of risk-taking during conspicuous mating displays. Evolution 69, 395–406 (2015).
Liker, A. & Székely, T. Mortality costs of sexual selection and parental care in natural populations of birds. Evolution 59, 890–897 (2005).
Johnson, J. B. Predator exposure alters female mate choice in the green swordtail. Behav. Ecol. 14, 619–625 (2003).
Berger, J. & Gompper, M. E. Sex ratios in extant ungulates: products of contemporary predation or past life histories?. J. Mammal. 80, 1084–1113 (1999).
Stockley, P. & Bro-Jørgensen, J. Female competition and its evolutionary consequences in mammals. Biol. Rev. 86, 341–366 (2011).
Lemaître, J.-F. et al. Sex differences in adult lifespan and aging rates of mortality across wild mammals. Proc. Natl. Acad. Sci. Usa. 117, 8546–8553 (2020).
Santos, E. S. A. & Nakagawa, S. The costs of parental care: a meta-analysis of the trade-off between parental effort and survival in birds. J. Evol. Biol. 25, 1911–1917 (2012).
Hadfield, J. D. MCMC methods for multi-response generalized linear mixed models: the MCMCglmm R package. J. Stat. Softw. 33, 1–22 (2010).
Prum, R. O. et al. A comprehensive phylogeny of birds (Aves) using targeted next-generation DNA sequencing. Nature 526, 569–573 (2015).
Santos, J. C. Fast molecular evolution associated with high active metabolic rates in poison frogs. Mol. Biol. Evol. 29, 2001–2018 (2012).
Bentler, P. M. Comparative fit indexes in structural models. Psychol. Bull. 107, 238–246 (1990).
West, S. G., Taylor, A. B. & Wu, W. Model fit and model selection in structural equation modeling. in Handbook of Structural Equation Modeling (ed. Hoyle, R. H.) 209–231 (Guilford, 2012).
van der Bijl, W. phylopath: Easy phylogenetic path analysis in R. PeerJ 6, e4718 (2018).
Benito, M. M. & González Solís, J. Sex ratio, sex-specific chick mortality and sexual size dimorphism in birds. J. Evol. Biol. 20, 1522–1530 (2007).
Pipoly, I. et al. Variation in adult sex ratios in tetrapods is linked to sex chromosomes through mortality differences between males and females. PLoS Biol. 23, e3003156 (2025).
Houston, A. I. & McNamara, J. M. John Maynard Smith and the importance of consistency in evolutionary game theory. Biol. Philos. 20, 933–950 (2006).
Clutton-Brock, T. H. Sex ratio variation in birds. Ibis 128, 317–329 (1986).
Briedis, M. et al. A full annual perspective on sex-biased migration timing in long-distance migratory birds. Proc. R. Soc. Lond. B Biol. Sci. 286, 20182821 (2019).
Kokko, H., Gunnarsson, T. G., Morrell, L. J. & Gill, J. A. Why do female migratory birds arrive later than males?. J. Anim. Ecol. 75, 1293–1303 (2006).
Végvári, Z. et al. Sex-biased breeding dispersal is predicted by social environment in birds. Ecol. Evol. 8, 6483–6491 (2018).
Payevsky, V. A. Sex-biased survival and philopatry in birds: Do they interact?. Biol. Bull. 43, 804–818 (2016).
Fandos, G. et al. Standardised empirical dispersal kernels emphasise the pervasiveness of long-distance dispersal in European birds. J. Anim. Ecol. 92, 158–170 (2023).
Trivers, R. L. Parental Investment and Sexual Selection. In Sexual Selection and the Descent of Man (ed. Campbell, B.) 136–179 (Aldine, 1972).
Kokko, H. & Jennions, M. D. Parental investment, sexual selection and sex ratios. J. Evol. Biol. 21, 919–948 (2008).
Jennions, M. D. & Fromhage, L. Not all sex ratios are equal: The Fisher condition, parental care and sexual selection. Philos. Trans. R. Soc. Lond. B Biol. Sci. 372, 20160312 (2017).
Weir, L. K., Grant, J. W. A. & Hutchings, J. A. The influence of pperational sex ratio on the intensity of competition for mates. Am. Nat. 177, 167–176 (2011).
Liker, A. et al. Evolution of large males is associated with female-skewed adult sex ratios in amniotes. Evolution 75, 1636–1649 (2021).
Grant, P. R. & Grant, B. R. Adult sex ratio influences mate choice in Darwin’s finches. Proc. Natl. Acad. Sci. USA 116, 12373–12382 (2019).
Liker, A., Freckleton, R. P. & Székely, T. Divorce and infidelity are associated with skewed adult sex ratios in birds. Curr. Biol. 24, 880–884 (2014).
Valcu, M., Valcu, C. & Kempenaers, B. Extra-pair paternity and sexual dimorphism in birds. J. Evol. Biol. 36, 764–779 (2023).
Søraker, J. S., Wright, J., Hanslin, F. Ø & Pepke, M. Le. The evolution of extra-pair paternity and paternal care in birds. Behav. Ecol. 34, 780–789 (2023).
Dunn, P. O. & Whittingham, L. A. Search costs influence the spatial distribution, but not the level, of extra-pair mating in tree swallows. Behav. Ecol. Sociobiol. 61, 449–454 (2006).
Matysioková, B. & Remeš, V. Faithful females receive more help: The extent of male parental care during incubation in relation to extra-pair paternity in songbirds. J. Evol. Biol. 26, 155–162 (2013).
Poiani, A. & Wilks, C. Sexually transmitted diseases: a possible cost of promiscuity in birds? Auk 117, 1061–1065 (2000).
Sheldon, B. C. Male phenotype, fertility, and the pursuit of extra-pair copulations by female birds. Proc. R. Soc. Lond. B Biol. Sci. 257, 25–30 (1994).
Shipley, B. Cause and Correlation in Biology: A User’s Guide to Path Analysis, Structural Equations, and Causal Inference with R. https://doi.org/10.1017/CBO9781139979573 (Cambridge University Press, 2016).
Song, Z., Liker, A., Liu, Y. & Székely, T. Evolution of social organization: phylogenetic analyses of ecology and sexual selection in weavers. Am. Nat. 200, 250–263 (2022).
Brown, CharlesR. & Brown, M. Group size and ectoparasitism affect daily survival probability in a colonial bird. Behav. Ecol. Sociobiol. 56, 498–511 (2004).
Guindre-Parker, S. & Rubenstein, D. R. Survival benefits of group living in a fluctuating environment. Am. Nat. 195, 1027–1036 (2020).
Greenwood, P. J. Mating systems, philopatry and dispersal in birds and mammals. Anim. Behav. 28, 1140–1162 (1980).
Toïgo, C. & Gaillard, J. Causes of sex-biased adult survival in ungulates: Sexual size dimorphism, mating tactic or environment harshness?. Oikos 101, 376–384 (2003).
Clutton-Brock, T. Review Lecture: mammalian mating systems. Proc. R. Soc. Lond. B Biol. Sci. 236, 339–372 (1989).
Fairbairn, D. J., Blanckenhorn, W. U. & Székely, T. Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism. https://doi.org/10.1093/acprof:oso/9780199208784.001.0001 (Oxford University Press, 2007).
Pipoly, I. et al. The genetic sex-determination system predicts adult sex ratios in tetrapods. Nature 527, 91–94 (2015).
Donald, P. F. Adult sex ratios in wild bird populations. Ibis 149, 671–692 (2007).
Stoffel, M. A., Nakagawa, S. & Schielzeth, H. rptR: repeatability estimation and variance decomposition by generalized linear mixed-effects models. Methods Ecol. Evol. 8, 1639–1644 (2017).
Gonzalez-Voyer, A. et al. Sex roles in birds: phylogenetic analyses of the influence of climate, life histories and social environment. Ecol. Lett. 25, 647–660 (2022).
Revelle, W. psych: Procedures for personality and psychological research. Northwestern University, Evanston, Illinois. R package version 2.5.6, https://CRAN.R-project.org/package=psych (2017).
Liker, A., Freckleton, R. P. & Székely, T. The evolution of sex roles in birds is related to adult sex ratio. Nat. Commun. 4, 1587 (2013).
Liker, A., Freckleton, R. P., Remeš, V. & Székely, T. Sex differences in parental care: gametic investment, sexual selection, and social environment. Evolution 69, 2862–2875 (2015).
Valcu, C., Valcu, M. & Kempenaers, B. The macroecology of extra-pair paternity in birds. Mol. Ecol. 30, 4884–4898 (2021).
Nakagawa, S. & Freckleton, R. P. Missing inaction: the dangers of ignoring missing data. Trends Ecol. Evol. 23, 592–596 (2008).
Mokos, J., Scheuring, I., Liker, A., Freckleton, R. P. & Székely, T. Degree of anisogamy is unrelated to the intensity of sexual selection. Sci. Rep. 11, 19424 (2021).
Goolsby, E. W., Bruggeman, J. & Ané, C. Rphylopars: fast multivariate phylogenetic comparative methods for missing data and within-species variation. Methods Ecol. Evol. 8, 22–27 (2017).
Team, R. C. R.: A Language and Environment for Statistical Computing. https://www.R-project.org/ (2013).
Paradis, E. Analysis of Phylogenetics and Evolution with R. https://doi.org/10.1007/978-1-4614-1743-9 (Springer New York, 2012)
Lefcheck, J. S. <scp>piecewiseSEM</scp>: Piecewise structural equation modelling in <scp>r</scp> for ecology, evolution, and systematics. Methods Ecol. Evol. 7, 573–579 (2016).
Rosseel, Y. lavaan: an R Package for Structural Equation Modeling. J. Stat. Softw. 48 (2012).
Cooney, C. Jetz avian phylogenies mapped to Prum backbone. The University of Sheffield. Dataset (2022).
Hardenberg, A. von & Gonzalez-Voyer, A. Disentangling evolutionary cause-effect relationships with phylogenetic confirmatory path analysis. Evolution 67, 378–387 (2013).
Shipley, B. The AIC model selection method applied to path analytic models compared using a d-separation test. Ecology 94, 560–564 (2013).
Song, Z., Liker, A., Liu, Y., Freckleton, R. & Székely, T. Data for ‘Demographic causes and social consequences of adult sex ratio variation’. figshare https://doi.org/10.6084/m9.figshare.31490812 (2026).
Acknowledgements
We thank Bálint Kovács for extracting some of the data used in the analyses. This work was supported by the National Research, Development and Innovation Office of Hungary to TS (HUN-REN–Debrecen University Reproductive Strategies Research Group, Ref. 1102207, ADVANCED 150852, HU-RIZONT-2024-00109). RPF and TS were supported by the Hungarian Academy of Sciences Guest Professorship scheme 2024-47. AL received funding from the HUN-REN TKI Hungarian Research Network (HUN-REN–PE Evolutionary Ecology Research Group, Ref. 16007) and was also supported by the National Research, Development and Innovation Office of Hungary (ADVANCED 150703).
Author information
Authors and Affiliations
Contributions
T.S. and Z.S. developed the concept of the study and wrote the first draft, Z.S. and A.L. prepared the data, Z.S. analysed the data, supported by A.L. and R.P.F., Y.L. provided feedback on the study design and manuscript, and all authors contributed to revising the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Achaz von Hardenberg, Mariana Carro and the other anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Song, Z., Liker, A., Liu, Y. et al. Demographic causes and social consequences of adult sex ratio variation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71230-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71230-4