Abstract
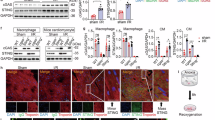
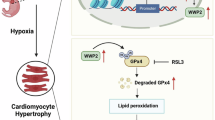
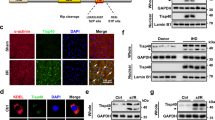
GPX4 is a crucial regulator of ferroptosis, yet its role in mitochondrial dysfunction during myocardial ischemia/reperfusion injury (MI/RI) is unclear. This study aims to clarify the effect and molecular mechanisms of GPX4 in MI/RI. We analyzed the spatiotemporal dynamics of GPX4 during MI/RI and observed high expression levels in border and normal areas but a significant reduction in the ischemic region utilizing spatial transcriptomics, spatial proteomics, and single-cell sequencing. Cardiomyocyte-derived GPX4 notably reduces myocardial damage and mitochondrial dysfunction in MI/RI while also alleviating long-term ventricular remodeling. Mechanistically, our findings reveal that GPX4, through its critical U46 active site, enhances the interaction between BNIP3 and USP20, decreasing ubiquitination at K131 of BNIP3. This process stabilizes BNIP3, promotes mitophagy, improves mitochondrial function, and ultimately preserves cardiac function. Our research defines the role of the GPX4/BNIP3/USP20 complex in MI/RI and uncovers a mechanism linking GPX4 to ferroptosis-related mitochondrial damage, providing valuable insights for advancing ferroptosis studies.
Similar content being viewed by others
Data availability
The proteomics data generated in this study have been deposited in the ProteomeXchange Consortium via the PRIDE partner repository under accession code PXD072382. Bulk RNA-seq data generated in this study have been deposited in the Genome Sequence Archive (GSA) under accession codes CRA035515 and CRA035410. Single-cell RNA sequencing data generated in this study have been deposited in the Genome Sequence Archive (GSA) under accession code CRA035546. All datasets are publicly accessible without restriction. Source data are provided with this paper.
References
Reed, G. W., Rossi, J. E. & Cannon, C. P. Acute myocardial infarction. Lancet 389, 197–210 (2017).
Alpert, J. S., Thygesen, K., Antman, E. & Bassand, J. P. Myocardial infarction redefined-a consensus document of The Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction. J. Am. Coll. Cardiol. 36, 959–969 (2000).
Heusch, G. Cardioprotection: chances and challenges of its translation to the clinic. Lancet 381, 166–175 (2013).
Hausenloy, D. J. & Yellon, D. M. Myocardial ischemia-reperfusion injury: a neglected therapeutic target. J. Clin. Invest 123, 92–100 (2013).
Heusch, G. Myocardial ischaemia-reperfusion injury and cardioprotection in perspective. Nat. Rev. Cardiol. 17, 773–789 (2020).
Xiang, Q., Yi, X., Zhu, X. H., Wei, X. & Jiang, D. S. Regulated cell death in myocardial ischemia-reperfusion injury. Trends Endocrinol. Metab. 35, 219–234 (2024).
Davidson, S. M. et al. Multitarget Strategies to Reduce Myocardial Ischemia/Reperfusion Injury: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 73, 89–99 (2019).
Karbassi, E. et al. Cardiomyocyte maturation: advances in knowledge and implications for regenerative medicine. Nat. Rev. Cardiol. 17, 341–359 (2020).
Lillo-Moya, J. et al. Targeting ferroptosis against ischemia/reperfusion cardiac injury. Antioxidants 10, 667 (2021).
Fang, X., Ardehali, H., Min, J. & Wang, F. The molecular and metabolic landscape of iron and ferroptosis in cardiovascular disease. Nat. Rev. Cardiol. 20, 7–23 (2023).
Xie, Y. et al. Ferroptosis: process and function. Cell Death Differ. 23, 369–379 (2016).
Cai, W. et al. Alox15/15-HpETE Aggravates Myocardial Ischemia-Reperfusion Injury by Promoting Cardiomyocyte Ferroptosis. Circulation 147, 1444–1460 (2023).
Wu, X., Li, Y., Zhang, S. & Zhou, X. Ferroptosis as a novel therapeutic target for cardiovascular disease. Theranostics 11, 3052–3059 (2021).
Liu, Y., Wan, Y., Jiang, Y., Zhang, L. & Cheng, W. GPX4: The hub of lipid oxidation, ferroptosis, disease and treatment. Biochim Biophys. Acta Rev. Cancer 1878, 188890 (2023).
Seibt, T. M., Proneth, B. & Conrad, M. Role of GPX4 in ferroptosis and its pharmacological implication. Free Radic. Biol. Med. 133, 144–152 (2019).
Ursini, F. & Maiorino, M. Lipid peroxidation and ferroptosis: The role of GSH and GPx4. Free Radic. Biol. Med 152, 175–185 (2020).
Qu, Z. et al. The positive feedback loop of the NAT10/Mybbp1a/p53 axis promotes cardiomyocyte ferroptosis to exacerbate cardiac I/R injury. Redox Biol. 72, 103145 (2024).
Ma, X. H. et al. ALOX15-launched PUFA-phospholipids peroxidation increases the susceptibility of ferroptosis in ischemia-induced myocardial damage. Signal Transduct. Target Ther. 7, 288 (2022).
Xu, S. et al. Naringenin alleviates myocardial ischemia/reperfusion injury by regulating the nuclear factor-erythroid factor 2-related factor 2 (Nrf2) /System xc-/ glutathione peroxidase 4 (GPX4) axis to inhibit ferroptosis. Bioengineered 12, 10924–10934 (2021).
Lu, H., Xiao, H., Dai, M., Xue, Y. & Zhao, R. Britanin relieves ferroptosis-mediated myocardial ischaemia/reperfusion damage by upregulating GPX4 through activation of AMPK/GSK3β/Nrf2 signalling. Pharm. Biol. 60, 38–45 (2022).
Ramachandra, C. J. A., Hernandez-Resendiz, S., Crespo-Avilan, G. E., Lin, Y. H. & Hausenloy, D. J. Mitochondria in acute myocardial infarction and cardioprotection. EBioMedicine 57, 102884 (2020).
Hernandez-Resendiz, S., Prunier, F., Girao, H., Dorn, G. & Hausenloy, D. J. Targeting mitochondrial fusion and fission proteins for cardioprotection. J. Cell Mol. Med 24, 6571–6585 (2020).
Onishi, M., Yamano, K., Sato, M., Matsuda, N. & Okamoto, K. Molecular mechanisms and physiological functions of mitophagy. EMBO J. 40, e104705 (2021).
Titus, A. S., Sung, E. A., Zablocki, D. & Sadoshima, J. Mitophagy for cardioprotection. Basic Res. Cardiol. 118, 42 (2023).
Kubli, D. A. et al. Parkin protein deficiency exacerbates cardiac injury and reduces survival following myocardial infarction. J. Biol. Chem. 288, 915–926 (2013).
Zhang, J. & Ney, P. A. Role of BNIP3 and NIX in cell death, autophagy, and mitophagy. Cell Death Differ. 16, 939–946 (2009).
Li, E. et al. BMAL1 regulates mitochondrial fission and mitophagy through mitochondrial protein BNIP3 and is critical in the development of dilated cardiomyopathy. Protein Cell 11, 661–679 (2020).
Hoshino, A. et al. p53-TIGAR axis attenuates mitophagy to exacerbate cardiac damage after ischemia. J. Mol. Cell Cardiol. 52, 175–184 (2012).
Yamashita, S. I. et al. Mitophagy mediated by BNIP3 and NIX protects against ferroptosis by downregulating mitochondrial reactive oxygen species. Cell Death Differ. 31, 651–661 (2024).
Kuppe, C. et al. Spatial multi-omic map of human myocardial infarction. Nature 608, 766–777 (2022).
Rao, S. V. et al. 2025 ACC/AHA/ACEP/NAEMSP/SCAI Guideline for the Management of Patients With Acute Coronary Syndromes: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 151, e771–e862 (2025).
Liu, L. et al. Deubiquitinase OTUD5 as a Novel Protector against 4-HNE-Triggered Ferroptosis in Myocardial Ischemia/Reperfusion Injury. Adv. Sci. 10, e2301852 (2023).
Xu, X. et al. The mechanisms of ferroptosis and its role in atherosclerosis. Biomed. Pharmacother. 171, 116112 (2024).
Xie, S. S. et al. Endothelial cell ferroptosis mediates monocrotaline-induced pulmonary hypertension in rats by modulating NLRP3 inflammasome activation. Sci. Rep. 12, 3056 (2022).
Schütte, J. P. et al. Platelet-Derived MicroRNAs Regulate Cardiac Remodeling After Myocardial Ischemia. Circ. Res 132, e96–e113 (2023).
Bahit, M. C., Kochar, A. & Granger, C. B. Post-Myocardial Infarction Heart Failure. JACC Heart Fail 6, 179–186 (2018).
Tadokoro, T. et al. Mitochondria-dependent ferroptosis plays a pivotal role in doxorubicin cardiotoxicity. JCI Insight 5, e132747 (2020).
Lesnefsky, E. J., Chen, Q., Tandler, B. & Hoppel, C. L. Mitochondrial dysfunction and myocardial ischemia-reperfusion: implications for novel therapies. Annu. Rev. Pharm. Toxicol. 57, 535–565 (2017).
Zhang, Q. et al. Inhibiting CD36 palmitoylation improves cardiac function post-infarction by regulating lipid metabolic homeostasis and autophagy. Nat. Commun. 16, 6602 (2025).
Gustafsson, ÅB. & Dorn, G. W. 2nd. Evolving and expanding the roles of mitophagy as a homeostatic and pathogenic process. Physiol. Rev. 99, 853–892 (2019).
Cao, Y. et al. A mitochondrial SCF-FBXL4 ubiquitin E3 ligase complex degrades BNIP3 and NIX to restrain mitophagy and prevent mitochondrial disease. EMBO J. 42, e113033 (2023).
Popovic, D., Vucic, D. & Dikic, I. Ubiquitination in disease pathogenesis and treatment. Nat. Med. 20, 1242–1253 (2014).
Liu, J., Tang, D. & Kang, R. Targeting GPX4 in ferroptosis and cancer: chemical strategies and challenges. Trends Pharm. Sci. 45, 666–670 (2024).
Liu, H. et al. Small-molecule allosteric inhibitors of GPX4. Cell Chem. Biol. 29, 1680–1693.e1689 (2022).
Yang, W. S. et al. Peroxidation of polyunsaturated fatty acids by lipoxygenases drives ferroptosis. Proc. Natl. Acad. Sci. USA 113, E4966–E4975 (2016).
Jang, S. et al. Elucidating the contribution of mitochondrial glutathione to ferroptosis in cardiomyocytes. Redox Biol. 45, 102021 (2021).
Sun, Y. et al. A mitophagy sensor PPTC7 controls BNIP3 and NIX degradation to regulate mitochondrial mass. Mol. Cell 84, 327–344.e329 (2024).
Zhong, L. et al. Novel GSDMD inhibitor GI-Y1 protects heart against pyroptosis and ischemia/reperfusion injury by blocking pyroptotic pore formation. Basic Res. Cardiol. 118, 40 (2023).
Acknowledgements
We would like to express our sincere gratitude to Mengxin Zhang and Wenting Wang from the scientific research center of Wenzhou Medical University for their invaluable assistance with the immunofluorescence experiment. Additionally, we appreciate Lingli Hou and Yanni Dong from the Scientific Research Center of Wenzhou Medical University for their help in echocardiography. At last, we thank Zhuqi Huang for valuable assistance in uploading the raw mass spectrometry data. This work was supported by the Natural Science Foundation of Zhejiang Province (LQ21H020009 and LMS25H020006 to B.-Z.Y.), Zhejiang Provincial postdoctoral research project (ZJ2024075 to B.-Z.Y.), the Medical and Health Science and Technology Project of Zhejiang Province (2025KY995 to B.-Z.Y.), the National Science Foundation of China (82570321 to B.-Z.Y., 82170242 and 81570454 to D.-L.Y.) and the State Key Laboratory of Transvascular Implantation Devices (012024015 to D.-L.Y.).
Author information
Authors and Affiliations
Contributions
L.-F.Z., Z.-F.C., Y.-C.Z., X.-X.F., Y.-X.Z., F.Y., R.-H.Z., K.-K.Y., D.-R.C., J.-H.L., and X.C. carried out the experiments. B.-Z.Y. contributed to the design of the experiments. B.-Z.Y., and L.-F.Z. participated in the drafting of the article. Z.-Q.H., C.C., D.-L.Y., and W.-J.H. contributed to data collection and analysis. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhong, L., Cheng, Z., Zhang, Y. et al. Cardiomyocyte-derived GPX4 stabilizes BNIP3 to facilitate mitophagy and mitigate myocardial ischemia/reperfusion injury. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71232-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71232-2