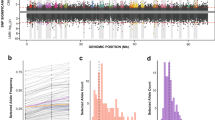
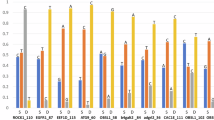
Abstract
Recombination has long been considered the primary mechanism to bring beneficial alleles together, which can increase the speed of adaptation from standing genetic variation. Recombination is fundamental to the transporter hypothesis proposed to explain precise parallel adaptation in Threespine Stickleback. We study an instance of freshwater adaptation in the Threespine Stickleback system using whole genome data from an evolutionary time-series to observe the genomic dynamics underlying rapid parallel adaptation. Here, we show that rapid adaptation to a freshwater environment depends on a few individuals with large haploblocks of freshwater-adaptive alleles (jackpot carriers) present among the anadromous founders at low frequencies. Biological kinship analyses indicates that mating among jackpot carriers and between jackpot carriers and non-jackpot individuals led to an increase in freshwater-adaptive alleles within the first few generations. This process allowed the population to overcome a substantial bottleneck likely caused by the low fitness of first-generation stickleback possessing a few freshwater-adaptive alleles born in the lake. Additionally, we find evidence that the genetic load that emerged from population growth after the bottleneck may have been reduced through an increase in homozygosity by inbreeding, ultimately purging deleterious alleles. Recombination likely played a limited role in this case of very rapid adaptation.
Similar content being viewed by others
Data availability
The whole genome data generated in this study have been deposited in the Sequence Read Archive (www.ncbi.nlm.nih.gov/sra) under accession code PRJNA1231081. Whole genomes from Rabbit Slough previously published in ref. 12 can be found at SRA under accession code PRJNA671690. All other whole genomes from ref. 12 can be found under accession code PRJNA247503. The data to generate the figures can be found in the Code Ocean capsule https://codeocean.com/capsule/9257557/tree (https://doi.org/10.24433/CO.2139373.v1).
Code availability
All custom scripts have been deposited to GitHub and can be accessed through the following link: https://github.com/a-kwakye/Rare-Jackpot-Individuals-Drive-Rapid-Adaptation-in-Threespine-Stickleback76 and Code Ocean at https://codeocean.com/capsule/9257557/tree (https://doi.org/10.24433/CO.2139373.v1).
References
Darwin Charles, R. On the origin of species by means of natural selection, or the preservation of favoured races in the struggle for life. (Murray, 1859).
Tellier, A., Hodgins, K., Stephan, W. & Stukenbrock, E. Rapid evolutionary adaptation: potential and constraints. Mol. Ecol. 33, e17350 (2024).
van Rijssel, J. C. et al. Fast adaptive responses in the oral jaw of Lake Victoria cichlids. Evolution 69, 179–189 (2015).
Bonnet, T. et al. Genetic variance in fitness indicates rapid contemporary adaptive evolution in wild animals. Science 376, 1012–1016 (2022).
Carroll, S. P., Hendry, A. P., Reznick, D. N. & Fox, C. W. Evolution on ecological time-scales. Funct. Ecol. 21, 387–393 (2007).
Barrett, R. D. H. & Schluter, D. Adaptation from standing genetic variation. Trends Ecol. Evol. 23, 38–44 (2008).
Pritchard, J. K., Pickrell, J. K. & Coop, G. The genetics of human adaptation: hard sweeps, soft sweeps, and polygenic adaptation. Curr. Biol. 20, R208–15 (2010).
Hermisson, J. & Pennings, P. S. Soft sweeps and beyond: understanding the patterns and probabilities of selection footprints under rapid adaptation. Methods Ecol. Evol. 8, 700–716 (2017).
Schrider, D. R. & Kern, A. D. Soft sweeps are the dominant mode of adaptation in the human genome. Mol. Biol. Evol. 34, 1863–1877 (2017).
Colosimo, P. F. et al. Widespread parallel evolution in sticklebacks by repeated fixation of Ectodysplasin alleles. Science 307, 1928–1933 (2005).
Reid, K., Bell, M. A. & Veeramah, K. R. Threespine stickleback: a model system for evolutionary genomics. Annu. Rev. Genomics Hum. Genet. 22, 357–383 (2021).
Roberts Kingman, G. A. et al. Predicting future from past: The genomic basis of recurrent and rapid stickleback evolution. Sci. Adv. 7, (2021).
Jones, F. C. et al. The genomic basis of adaptive evolution in threespine sticklebacks. Nature 484, 55–61 (2012).
Klepaker, T. Morphological changes in a marine population of threespined stickleback, Gasterosteus aculeatus, recently isolated in fresh water. Can. J. Zool. 71, 1251–1258 (1993).
Bell, M. A., Aguirre, W. E. & Buck, N. J. Twelve years of contemporary armor evolution in a threespine stickleback population. Evolution 58, 814–824 (2004).
Schluter, D. & Conte, G. L. Genetics and ecological speciation. Proc. Natl. Acad. Sci. USA. 106, 9955–9962 (2009).
Bassham, S., Catchen, J., Lescak, E., von Hippel, F. A. & Cresko, W. A. Repeated selection of alternatively adapted haplotypes creates sweeping genomic remodeling in stickleback. Genetics 209, 921–939 (2018).
Galloway, J., Cresko, W. A. & Ralph, P. A Few Stickleback Suffice for the Transport of Alleles to New Lakes. G3 10, 505–514 (2020).
Venu, V. et al. Fine-scale contemporary recombination variation and its fitness consequences in adaptively diverging stickleback fish. Nat. Ecol. Evol. 8, 1337–1352 (2024).
Hohenlohe, P. A., Bassham, S., Currey, M. & Cresko, W. A. Extensive linkage disequilibrium and parallel adaptive divergence across threespine stickleback genomes. Philos. Trans. R. Soc. Lond. B Biol. Sci. 367, 395–408 (2012).
Bell, M. A. et al. Reintroduction of threespine stickleback into Cheney and Scout Lakes, Alaska. Evol. Ecol. Res. 17, 157–178 (2016).
Kurz, M. L., Heins, D. C., Bell, M. A. & von Hippel, F. A. Shifts in life-history traits of two introduced populations of threespine stickleback. Evol. Ecol. Res. 17, 225–242 (2016).
DeFaveri, J. & Merilä, J. Local adaptation to salinity in the three-spined stickleback? J. Evol. Biol. 27, 290–302 (2014).
Divino, J. N. et al. Osmoregulatory physiology and rapid evolution of salinity tolerance in threespine stickleback recently introduced to fresh water. Evol. Ecol. Res. 17, 179–201 (2016).
Aguirre, W. E. et al. Freshwater colonization, adaptation, and genomic divergence in Threespine Stickleback. Integr. Comp. Biol. 62, 388–405 (2022).
Baker, J. A., Heins, D. C., Foster, S. A. & King, R. W. An overview of life-history variation in female Threespine Stickleback. Behaviour 145, 579–602 (2008).
Korneliussen, T. S., Albrechtsen, A. & Nielsen, R. ANGSD: analysis of next generation sequencing data. BMC Bioinforma. 15, 356 (2014).
Monroy Kuhn, J. M., Jakobsson, M. & Günther, T. Estimating genetic kin relationships in prehistoric populations. PLoS One 13, e0195491 (2018).
Alaçamlı, E. et al. READv2: Advanced and user-friendly detection of biological relatedness in archaeogenomics. bioRxiv 2024.01.23.576660 (2024) https://doi.org/10.1101/2024.01.23.576660.
Hanghøj, K., Moltke, I., Andersen, P. A., Manica, A. & Korneliussen, T. S. Fast and accurate relatedness estimation from high-throughput sequencing data in the presence of inbreeding. Gigascience 8, (2019).
Bell, M. A. & Foster, S. A. Introduction to the evolutionary biology of the threespine stickleback. The evolutionary biology of the threespine (1994).
Crow, J. F. Genetic Loads and the Cost of Natural Selection. in Mathematical Topics in Population Genetics (ed. Kojima, K.-I.) 128–177 (Springer Berlin Heidelberg, 1970).
Bertorelle, G. et al. Genetic load: genomic estimates and applications in non-model animals. Nat. Rev. Genet. 23, 492–503 (2022).
Wang, J., Hill, W. G., Charlesworth, D. & Charlesworth, B. Dynamics of inbreeding depression due to deleterious mutations in small populations: mutation parameters and inbreeding rate. Genet. Res. 74, 165–178 (1999).
Charlesworth, D. & Willis, J. H. The genetics of inbreeding depression. Nat. Rev. Genet. 10, 783–796 (2009).
Nietlisbach, P., Muff, S., Reid, J. M., Whitlock, M. C. & Keller, L. F. Nonequivalent lethal equivalents: Models and inbreeding metrics for unbiased estimation of inbreeding load. Evol. Appl. 12, 266–279 (2019).
Haller, B. C. & Messer, P. W. SLiM 4: multispecies eco-evolutionary modeling. Am. Nat. 201, E127–E139 (2023).
Marchinko, K. B. & Schluter, D. Parallel evolution by correlated response: lateral plate reduction in threespine stickleback. Evolution 61, 1084–1090 (2007).
Gibbons, T. C., Rudman, S. M. & Schulte, P. M. Responses to simulated winter conditions differ between threespine stickleback ecotypes. Mol. Ecol. 25, 764–775 (2016).
Lescak, E. A. et al. Evolution of stickleback in 50 years on earthquake-uplifted islands. Proc. Natl. Acad. Sci. USA. 112, E7204–12 (2015).
Pressley, P. H. Parental effort and the evolution of nest-guarding tactics in the threespine stickleback, Gasterosteus aculeatus l. Evolution 35, 282–295 (1981).
Bell, M. Evolution of phenotypic diversity in Gasterosteus aculeatus superspecies on the Pacific coast of North America. Syst. Biol. 25, 211–227 (1976).
Withler, R. E. & Mcphail, J. D. Genetic variability in freshwater and anaromous sticklebacks (Gasterosteus aculeatus) of southern British Columbia. Can. J. Zool. 63, 528–533 (1985).
Taylor, E. B. & McPhail, J. D. Historical contingency and ecological determinism interact to prime speciation in sticklebacks, Gasterosteus. Proc. Biol. Sci. 267, 2375–2384 (2000).
Hedrick, P. W. Purging inbreeding depression and the probability of extinction: full-sib mating. Heredity 73, 363–372 (1994).
Lynch, M., Conery, J. & Burger, R. Mutation accumulation and the extinction of small populations. Am. Nat. 146, 489–518 (1995).
Palkopoulou, E. et al. Complete genomes reveal signatures of demographic and genetic declines in the woolly mammoth. Curr. Biol. 25, 1395–1400 (2015).
Barrett, S. C. H. & Charlesworth, D. Effects of a change in the level of inbreeding on the genetic load. Nature 352, 522–524 (1991).
Grossen, C., Guillaume, F., Keller, L. F. & Croll, D. Purging of highly deleterious mutations through severe bottlenecks in Alpine ibex. Nat. Commun. 11, 1001 (2020).
Croft, L., Matheson, P., Butterworth, N. J. & McGaughran, A. Fitness consequences of population bottlenecks in an invasive blowfly. Mol. Ecol. e17492 (2024).
Jay, P. et al. Mutation load at a mimicry supergene sheds new light on the evolution of inversion polymorphisms. Nat. Genet. 53, 288–293 (2021).
Harringmeyer, O. S. & Hoekstra, H. E. Chromosomal inversion polymorphisms shape the genomic landscape of deer mice. Nat. Ecol. Evol. 6, 1965–1979 (2022).
Nickel, J., Laine, J. & Foote, A. D. Complex patterns of hitchhiking mutation load among stickleback populations. bioRxiv (2025) https://doi.org/10.1101/2025.07.24.666323.
Cresko, W. A. et al. Parallel genetic basis for repeated evolution of armor loss in Alaskan threespine stickleback populations. Proc. Natl. Acad. Sci. USA. 101, 6050–6055 (2004).
Xie, K. T. et al. DNA fragility in the parallel evolution of pelvic reduction in stickleback fish. Science 363, 81–84 (2019).
Baker, J. A., Foster, S. A., Heins, D. C., Bell, M. A. & King, R. W. Variation in female life history traits among Alaskan populations of the threespine stickleback, Gasterosteus aculeatus. Biol. J. Linn. Soc. 63, 141–159 (1998).
Heins, D. C., Singer, S. S. & Baker, J. A. Virulence of the cestode Schistocephalus solidus and reproduction in infected threespine stickleback, Gasterosteus aculeatus. Can. J. Zool. 77, 1967–1974 (1999).
Heins, D. C. & Brown-Peterson, N. J. The reproductive biology of small fishes and the clutch concept: Combining macroscopic and histological approaches. Aquac. Fish. Fish. 2, 253–264 (2022).
Lindgreen, S. AdapterRemoval: easy cleaning of next-generation sequencing reads. BMC Res. Notes 5, 337 (2012).
Peichel, C. L., Sullivan, S. T., Liachko, I. & White, M. A. Improvement of the Threespine Stickleback genome using a Hi-C-based proximity-guided assembly. J. Hered. 108, 693–700 (2017).
Danecek, P. et al. Twelve years of SAMtools and BCFtools. Gigascience 10, (2021).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Van Der Auwera, G. A. et al. From FastQ data to high-confidence variant calls: the genome analysis toolkit best practices pipeline. Curr. Protoc. Bioinformatics 43, (2013).
DePristo, M. A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498 (2011).
Browning, S. R. & Browning, B. L. Rapid and accurate haplotype phasing and missing-data inference for whole-genome association studies by use of localized haplotype clustering. Am. J. Hum. Genet. 81, 1084–1097 (2007).
Shanfelter, A. F., Archambeault, S. L. & White, M. A. Divergent fine-scale recombination landscapes between a freshwater and marine population of threespine stickleback fish. Genome Biol. Evol. 11, 1573–1585 (2019).
Chan, A. H., Jenkins, P. A. & Song, Y. S. Genome-wide fine-scale recombination rate variation in Drosophila melanogaster. PLoS Genet. 8, e1003090 (2012).
Nielsen, R., Korneliussen, T., Albrechtsen, A., Li, Y. & Wang, J. SNP calling, genotype calling, and sample allele frequency estimation from New-Generation Sequencing data. PLoS One 7, e37558 (2012).
Gutenkunst, R. N., Hernandez, R. D., Williamson, S. H. & Bustamante, C. D. Inferring the joint demographic history of multiple populations from multidimensional SNP frequency data. PLoS Genet. 5, e1000695 (2009).
Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 123, 585–595 (1989).
Kim, S. Y. et al. Estimation of allele frequency and association mapping using next-generation sequencing data. BMC Bioinforma. 12, 231 (2011).
Lynch, M., Bost, D., Wilson, S., Maruki, T. & Harrison, S. Population-genetic inference from pooled-sequencing data. Genome Biol. Evol. 6, 1210–1218 (2014).
Cotterman, C. W. A calculus for statistico-genetics. (1940).
Haller, B. C., Ralph, P. L. & Messer, P. W. SLiM 5: Eco-evolutionary simulations across multiple chromosomes and full genomes. bioRxiv (2025) https://doi.org/10.1101/2025.08.07.669155.
McPhail, J. D. Speciation and the evolution of reproductive isolation in the sticklebacks (Gasterosteus) of south-western British Columbia. in The Evolutionary Biology of the Threespine Stickleback 399–437 (Oxford University Press, Oxford, 1994).
Kwakye, A. A-kwakye/Rare-Jackpot-Individuals-Drive-Rapid-Adaptation-in-Threespine-Stickleback: Rare-Jackpot-Individuals-Drive-Rapid-Adaptation-in-Threespine-Stickleback. (Zenodo, 2026). https://doi.org/10.5281/ZENODO.18345063.
Acknowledgements
This work was supported by NIH R01GM124330 to K.R.V. and M.A.B., NSF grants DEB-0322818 and DEB-0919184 to M.A.B. and F. J. Rohlf, and the Newcomb College Institute of Tulane University to D.C.H. We would like to thank the many students who helped found the Scout Lake population and to capture stickleback from it. In particular, we thank W. E. Aguirre and P. J. Park, who were instrumental in the fieldwork. We thank members of the Veeramah Lab for comments and discussions during the preparation of this manuscript, especially Thomas Bertino and Rachael Herman for useful discussions on simulations.
Author information
Authors and Affiliations
Contributions
A.K: Formal analysis, DNA quantification, library preparation, genomic data processing; K.R: experimental design, DNA extraction, DNA quantification, library preparation, construction of reference panel for imputation, genomic data processing; M.W: crosses from Mile Lake; D.C.H: sampling, life-history data collection and processing; M.A.B: experimental design, population founding, most sampling, tissue sampling, and morphological data; K.R.V: experimental design, project supervision. A.K wrote the initial draft with input from all authors. All authors contributed to production of the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Bohao Fang, Xidong Mu, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kwakye, A., Reid, K., Wund, M.A. et al. Rare jackpot individuals drive rapid adaptation in Threespine Stickleback. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71236-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71236-y