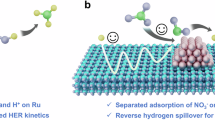
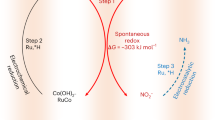
Abstract
Electrochemical nitrate reduction in alkaline media offers a sustainable route for ammonia synthesis at rates rivaling those of the Haber–Bosch process. However, its energy efficiency is limited by sluggish nitrate deoxidation and hydrogenation, compounded by challenges in proton supply via H2O dissociation. Here, we develop an enzyme-like substrate transport channel through a hierarchical arrangement of metallic Ag and Ru nanophases, to enable cascade nitrate-to-ammonia conversion and optimize underpotential-deposited hydrogen utilization. Operando characterization and theoretical calculations reveal that Ag–Ru electronic synergy regulates underpotential-deposited hydrogen coverage at Ru-centered active sites by coupling facilitated H2O dissociation with *OH-mediated site regeneration, thereby promoting nitrite relay conversion. Our underpotential-deposited hydrogen-assisted nitrate electroreduction system delivers a half-cell ammonia energy efficiency of 53.7% at 0.2 V versus RHE with near-unity Faradaic efficiency across a wide nitrate concentration range, and an ammonia partial current density of 2.2 A cm−2 at 0 V versus RHE. Pairing cathodic nitrate reduction with anodic H2 oxidation enables ammonia production costs below $1.15 kg−1 while maintaining sustained energy efficiency over 100 h at 200 mA cm−2.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are detailed in the Supporting information/Source data file. Source data are provided with this paper.
References
Zhang, X. et al. Managing nitrogen for sustainable development. Nature 528, 51–59 (2015).
Ye, D. P. & Tsang, S. C. E. Prospects and challenges of green ammonia synthesis. Nat. Synth. 2, 612–623 (2023).
Soloveichik, G. Electrochemical synthesis of ammonia as a potential alternative to the Haber–Bosch process. Nat. Catal. 2, 377–380 (2019).
Zhang, S. N. et al. Ampere-level reduction of pure nitrate by electron-deficient Ru with K+ ions repelling effect. Nat. Commun. 15, 10877 (2024).
Wang, Y. et al. Ammonia electrosynthesis from nitrate using a stable amorphous/crystalline dual-phase Cu catalyst. Nat. Commun. 16, 897 (2025).
van Langevelde, P. H. et al. Electrocatalytic nitrate reduction for sustainable ammonia production. Joule 5, 290–294 (2021).
Zhang, B. et al. Defect-induced triple synergistic modulation in copper for superior electrochemical ammonia production across broad nitrate concentrations. Nat. Commun. 15, 2816 (2024).
Chen, F. Y. et al. Efficient conversion of low-concentration nitrate sources into ammonia on a Ru-dispersed Cu nanowire electrocatalyst. Nat. Nanotechnol. 17, 759–767 (2022).
Liao, W. R. et al. Sustainable conversion of alkaline nitrate to ammonia at activities greater than 2 A cm−2. Nat. Commun. 15, 1264 (2024).
Chen, X. H. et al. Gradient-concentration RuCo electrocatalyst for efficient and stable electroreduction of nitrate into ammonia. Nat. Commun. 15, 6278 (2024).
Fang, J. Y. et al. Ampere-level current density ammonia electrochemical synthesis using CuCo nanosheets simulating nitrite reductase bifunctional nature. Nat. Commun. 13, 7899 (2022).
Wang, J. et al. Stepwise structural evolution toward robust carboranealkynyl-protected copper nanocluster catalysts for nitrate electroreduction. Sci. Adv. 10, eadn7556 (2024).
He, W. H. et al. Enhanced nitrate-to-ammonia efficiency over linear assemblies of copper-cobalt nanophases stabilized by redox polymers. Adv. Mater. 35, 202303050 (2023).
Wang, Y. et al. Enhanced nitrate-to-ammonia activity on copper–nickel alloys via tuning of intermediate adsorption. J. Am. Chem. Soc. 142, 5702–5708 (2020).
He, W. H. et al. Splicing the active phases of copper/cobalt-based catalysts achieves high-rate tandem electroreduction of nitrate to ammonia. Nat. Commun. 13, 1129 (2022).
Hu, Q. K. et al. Low power consumption ammonia electrosynthesis using hydrogen-nitrate flow electrolyzer. ACS Energy Lett. 9, 2303–2309 (2024).
Smith, C. et al. Current and future role of Haber–Bosch ammonia in a carbon-free energy landscape. Energy Environ. Sci. 13, 331–344 (2020).
Li, P. P. et al. A single-site iron catalyst with preoccupied active center that achieves selective ammonia electrosynthesis from nitrate. Energy Environ. Sci. 14, 3522–3531 (2021).
Chen, F. Y. et al. Electrochemical nitrate reduction to ammonia with cation shuttling in a solid electrolyte reactor. Nat. Catal. 7, 1032–1043 (2024).
de Groot, M. T. & Koper, M. T. M. The influence of nitrate concentration and acidity on the electrocatalytic reduction of nitrate on platinum. J. Electroanal. Chem. 562, 81–94 (2004).
de Vooys, A. C. A. et al. Electrocatalytic reduction of NO3− on palladium/copper electrodes. J. Mol. Catal. A Chem. 154, 203–215 (2000).
Martinez, J. et al. State-of-the-art and perspectives of the catalytic and electrocatalytic reduction of aqueous nitrates. Appl. Catal. B Environ. 207, 42–59 (2017).
Wang, Y. T. et al. Nitrate electroreduction: mechanism insight, in situ characterization, performance evaluation, and challenges. Chem. Soc. Rev. 50, 6720–6733 (2021).
Gao, Q. et al. Breaking adsorption-energy scaling limitations of electrocatalytic nitrate reduction on intermetallic CuPd nanocubes by machine-learned insights. Nat. Commun. 13, 2338 (2022).
Liu, Y. et al. Efficient tandem electroreduction of nitrate into ammonia through coupling Cu single atoms with adjacent Co3O4. Nat. Commun. 15, 3619 (2024).
Han, S. H. et al. Ultralow overpotential nitrate reduction to ammonia via a three-step relay mechanism. Nat. Catal. 6, 402–414 (2023).
Zhu, S. Q. et al. The role of ruthenium in improving the kinetics of hydrogen oxidation and evolution reactions of platinum. Nat. Catal. 4, 711–718 (2021).
Ledezma-Yanez, I. et al. Interfacial water reorganization as a pH-dependent descriptor of the hydrogen evolution rate on platinum electrodes. Nat. Energy 2, 17031 (2017).
Wan, C. Z. et al. Amorphous nickel hydroxide shell tailors local chemical environment on platinum surface for alkaline hydrogen evolution reaction. Nat. Mater. 22, 1022–1029 (2023).
Huang, Q. et al. Single-zinc vacancy unlocks high-rate H2O2 electrosynthesis from mixed dioxygen beyond Le Chatelier principle. Nat. Commun. 16, 1539 (2025).
Zheng, S. S. et al. The loss of interfacial water-adsorbate hydrogen bond connectivity position surface-active hydrogen as a crucial intermediate to enhance nitrate reduction reaction. J. Am. Chem. Soc. 146, 26965–26974 (2024).
Li, H. et al. Steering from electrochemical denitrification to ammonia synthesis. Nat. Commun. 14, 112 (2023).
Li, Y. et al. Reversible hydrogen acceptor-donor enables relay mechanism for nitrate-to-ammonia electrocatalysis. Angew. Chem. Int. Ed. 64, e202417631 (2024).
Li, Y. F. et al. Improved nitrate-to-ammonia electrocatalysis through hydrogen poisoning effects. Angew. Chem. Int. Ed. 63, e202411068 (2024).
Liang, S. Z. et al. H* species regulation by Mn-Co(OH)2 for efficient nitrate electro-reduction in neutral solution. Angew. Chem. Int. Ed. 63, e202400206 (2024).
Wang, Y. et al. Phase-regulated active hydrogen behavior on molybdenum disulfide for electrochemical nitrate-to-ammonia conversion. Angew. Chem. Int. Ed. 63, e202315109 (2023).
Zhou, J. et al. Regulating active hydrogen adsorbed on grain boundary defects of nano-nickel for boosting ammonia electrosynthesis from nitrate. Energy Environ. Sci. 16, 2611–2620 (2023).
Liu, M. J. et al. Catalytic performance and near-surface X-ray characterization of titanium hydride electrodes for the electrochemical nitrate reduction reaction. J. Am. Chem. Soc. 144, 5759–5744 (2022).
Fan, K. et al. Active hydrogen boosts electrochemical nitrate reduction to ammonia. Nat. Commun. 13, 7958 (2022).
Liu, H. L. et al. Active hydrogen-controlled CO2/N2/NOx electroreduction: from mechanism understanding to catalyst design. Innov. Mater. 2, 100058 (2024).
Carvalho, O. Q. et al. Role of electronic structure on nitrate reduction to ammonium: a periodic journey. J. Am. Chem. Soc. 144, 14809–14818 (2022).
Jerkiewicz, G. Electrochemical hydrogen adsorption and absorption. Part 1: under-potential deposition of hydrogen. Electrocatalysis 1, 179–199 (2010).
McCrum, I. T. & Koper, M. T. M. The role of adsorbed hydroxide in hydrogen evolution reaction kinetics on modified platinum. Nat. Energy 5, 891–899 (2020).
Liu, H. et al. Low-coordination rhodium catalysts for an efficient electrochemical nitrate reduction to ammonia. ACS Catal. 13, 1513–1521 (2023).
Rousseau, B. J. G. et al. Theoretical analysis of hydrogen underpotential deposition on Pt(111) under alkaline conditions. J. Phys. Chem. C 128, 12109–12120 (2024).
Benedetti, T. M. et al. Electrocatalytic nanoparticles that mimic the three-dimensional geometric architecture of enzymes: nanozymes. J. Am. Chem. Soc. 140, 13449–13455 (2018).
O’Mara, P. B. et al. Cascade reactions in nanozymes: spatially separated active sites inside Ag-core–porous-Cu-shell nanoparticles for multistep carbon dioxide reduction to higher organic molecules. J. Am. Chem. Soc. 141, 14093–14097 (2019).
Karakaya, I. & Thompson, W. T. The Ag−Ru (silver-ruthenium) system. Bull. Alloy Ph. Diagr. 7, 365–368 (1986).
Zamora Zeledón, J. A. et al. Tuning the electronic structure of Ag-Pd alloys to enhance performance for alkaline oxygen reduction. Nat. Commun. 12, 620 (2021).
Gao, Q. et al. Synthesis of core/shell nanocrystals with ordered intermetallic single-atom alloy layers for nitrate electroreduction to ammonia. Nat. Synth. 2, 624–634 (2023).
Zhang, S. et al. Fe/Cu diatomic catalysts for electrochemical nitrate reduction to ammonia. Nat. Commun. 14, 3634 (2023).
Liu, Y. L. et al. Shear-strained Pd single-atom electrocatalysts for nitrate reduction to ammonia. Angew. Chem. Int. Ed. 63, e202411396 (2024).
Wan, Y. C. et al. Interfacial water regulation for nitrate electroreduction to ammonia at ultralow overpotentials. Adv. Mater. 37, 2417696 (2025).
Zhang, Z. X. et al. Graphdiyne enabled nitrogen vacancy formation in copper nitride for efficient ammonia synthesis. J. Am. Chem. Soc. 146, 14898–14904 (2024).
Shi, X. Y. et al. Synergistic effect of Ni/Ni(OH)2 core-shell catalyst boosts tandem nitrate reduction for ampere-level ammonia production. Angew. Chem. Int. Ed. 63, e202406750 (2024).
Song, Z. M. et al. Efficient electroreduction of nitrate into ammonia at ultralow concentrations via an enrichment effect. Adv. Mater. 34, 2204306 (2022).
Dai, J. et al. Spin polarized Fe1–Ti pairs for highly efficient electroreduction nitrate to ammonia. Nat. Commun. 15, 88 (2024).
Liu, H. et al. Electrocatalytic nitrate reduction on oxide-derived silver with tunable selectivity to nitrite and ammonia. ACS Catal. 11, 8431–8442 (2021).
Janik, M. J. et al. On the presence of surface bound hydroxyl species on polycrystalline Pt electrodes in the “hydrogen potential region” (0–0.4 V-RHE). J. Catal. 367, 332–337 (2018).
Intikhab, S. et al. Adsorbed hydroxide does not participate in the volmer step of alkaline hydrogen electrocatalysis. ACS Catal. 7, 8314–8319 (2017).
Li, Y. et al. Hydrogen spillover mechanism at the metal-metal interface in electrocatalytic hydrogenation. Angew. Chem. Int. Ed. 63, e202407810 (2024).
Damian, A. & Omanovic, S. Ni and NiMo hydrogen evolution electrocatalysts electrodeposited in a polyaniline matrix. J. Power Sources 158, 464–476 (2006).
Šimpraga, R. et al. In situ determination of the ‘real are factor’ in H2 evolution electrocatalysis at porous Ni–Fe composite electrodes. J. Electroanal. Chem. 424, 141–151 (1997).
Krstajić, N. V. et al. The determination of kinetics parameters of the hydrogen evolution on Ti–Ni alloys by ac impedance. Electrochim. Acta 42, 323–330 (1997).
Zhu, S. et al. pH-dependent hydrogen and water binding energies on platinum surfaces as directly probed through surface-enhanced infrared absorption spectroscopy. J. Am. Chem. Soc. 142, 8748–8754 (2020).
Liu, E. S. et al. Unifying the hydrogen evolution and oxidation reactions kinetics in base by identifying the catalytic roles of hydroxyl-water-cation adducts. J. Am. Chem. Soc. 141, 3232–3239 (2019).
Wei, R. L. et al. Ammonia oxidation on iridium electrode in alkaline media: an in situ ATR-SEIRAS study. J. Electroanal. Chem. 896, 115254 (2021).
Huang, Y. M. et al. Pulsed electroreduction of low-concentration nitrate to ammonia. Nat. Commun. 14, 7368 (2023).
Rosca, V. et al. Hydroxylamine electrochemistry at polycrystalline platinum in acidic media: a voltammetric, DEMS and FTIR study. J. Electroanal. Chem. 566, 53–62 (2004).
Kresse, G. & Furthmiiller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Grimme, S. et al. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Henkelman, G. & Jónsson, H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J. Chem. Phys. 113, 9978–9985 (2000).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (nos. 22374141, 22134006, and U2241287).
Author information
Authors and Affiliations
Contributions
L.L., W.H. supervised this project. W.H., L.Z. concevied the project, analyzed the data and wrote the manuscript. L.Z. conducted the experiments. H.G. performed the theoretical calculations. R.L., X.L., and Y.F. participated in the discussion for the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Dong-Hee Lim and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, L., Liu, R., Liang, X. et al. Exploiting underpotential deposited hydrogen enables energy-efficient nitrate electroreduction to ammonia. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71299-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71299-x